Advanced Synthesis of Redox-Active Tetrathiafulvalene Diols for Next-Generation Polyurethanes
The landscape of functional polymer chemistry is witnessing a significant shift towards incorporating electroactive units into standard resin backbones, a trend exemplified by the innovations detailed in patent CN112812096B. This intellectual property introduces a robust methodology for synthesizing tetrathiafulvalene (TTF) diol compounds, which serve as critical precursors for redox-active polyurethanes. Unlike conventional approaches that often struggle with the instability of organosulfur intermediates, this patent outlines a streamlined pathway starting from methyl propiolate and carbon disulfide. The core innovation lies in the efficient construction of the TTF core followed by a versatile transesterification step that allows for the modular attachment of various glycol chains. 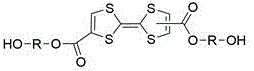 This structural flexibility is paramount for R&D directors aiming to tune the electronic properties of the final polymer matrix. By enabling the precise integration of the TTF unit, manufacturers can produce materials with reversible oxidation states, opening new avenues for applications in molecular wires, sensors, and smart coatings. The patent emphasizes not just the novelty of the molecule, but the practical viability of its production, addressing a long-standing gap between academic curiosity and industrial applicability in the realm of electroactive organic materials.
This structural flexibility is paramount for R&D directors aiming to tune the electronic properties of the final polymer matrix. By enabling the precise integration of the TTF unit, manufacturers can produce materials with reversible oxidation states, opening new avenues for applications in molecular wires, sensors, and smart coatings. The patent emphasizes not just the novelty of the molecule, but the practical viability of its production, addressing a long-standing gap between academic curiosity and industrial applicability in the realm of electroactive organic materials.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the functionalization of tetrathiafulvalene derivatives has been plagued by synthetic complexity and poor atom economy. Traditional routes often require harsh lithiation conditions or the use of sensitive organometallic reagents that demand rigorous exclusion of moisture and oxygen, significantly driving up operational costs and safety risks. Furthermore, introducing hydroxyl groups directly onto the TTF skeleton frequently involves multi-step protection and deprotection sequences, which drastically reduce overall yield and generate substantial chemical waste. For procurement managers, these inefficiencies translate into volatile supply chains and unpredictable pricing for high-purity intermediates. The reliance on exotic catalysts and difficult-to-source precursors creates bottlenecks that hinder the commercial scale-up of complex organosulfur compounds. Additionally, the purification of TTF derivatives synthesized via older methods often proves challenging due to the formation of closely related sulfur-containing byproducts that are difficult to separate, compromising the electronic performance of the final polyurethane material.
The Novel Approach
In stark contrast, the methodology disclosed in CN112812096B leverages a clever two-stage strategy that bypasses these historical hurdles through the use of stable ester intermediates. The process begins with the formation of tetrathiafulvalene dimethyl ester, a robust intermediate that can be isolated and stored, decoupling the core synthesis from the functionalization step. The subsequent transesterification reaction utilizes concentrated sulfuric acid as a catalyst, a ubiquitous and inexpensive reagent, to swap the methyl ester groups for desired glycol chains such as ethylene glycol or tetraethylene glycol. This approach dramatically simplifies the workflow, eliminating the need for cryogenic conditions in the second step and allowing the reaction to proceed at moderate temperatures between 115°C and 125°C. For supply chain heads, this translates to a process that relies on commodity chemicals rather than specialized reagents, ensuring greater continuity of supply. The ability to directly introduce diol functionality without protecting groups represents a paradigm shift in efficiency, reducing both the time and solvent volume required to produce kilogram quantities of these high-value intermediates.
Mechanistic Insights into Phosphine-Mediated Cyclization and Transesterification
The success of this synthetic route hinges on the precise control of two distinct mechanistic phases, beginning with the construction of the TTF core itself. In the first stage, tributylphosphine acts as a nucleophilic catalyst to initiate the coupling of methyl propiolate and carbon disulfide. This reaction, conducted in tetrahydrofuran at temperatures ranging from -40°C to -60°C, facilitates the formation of a dithiole ring system which subsequently dimerizes to form the tetrathiafulvalene skeleton. The low temperature is critical here to manage the exothermicity and prevent polymerization side reactions, ensuring high selectivity for the dimethyl ester product. Following the isolation of this intermediate, the mechanism shifts to a classic acid-catalyzed ester exchange. 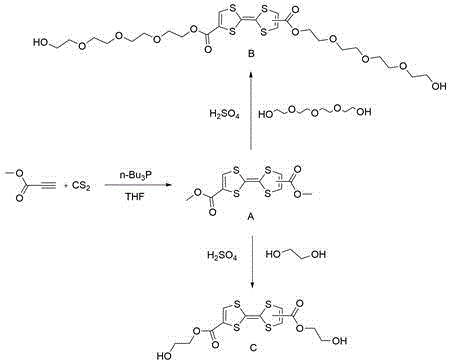 As illustrated in the reaction scheme, the sulfuric acid protonates the carbonyl oxygen of the TTF dimethyl ester, increasing its electrophilicity and making it susceptible to nucleophilic attack by the hydroxyl groups of the glycol solvent. This transesterification is driven to completion by the excess of glycol solvent, which acts as both reactant and medium. The mechanistic elegance lies in the stability of the TTF core under these acidic conditions; unlike many other organosulfur systems that might degrade or oxidize prematurely, the TTF unit remains intact, preserving its crucial pi-conjugated system. This preservation is essential for maintaining the redox activity required for the final polyurethane application, ensuring that the electronic properties are intrinsic to the polymer backbone rather than being degraded during synthesis.
As illustrated in the reaction scheme, the sulfuric acid protonates the carbonyl oxygen of the TTF dimethyl ester, increasing its electrophilicity and making it susceptible to nucleophilic attack by the hydroxyl groups of the glycol solvent. This transesterification is driven to completion by the excess of glycol solvent, which acts as both reactant and medium. The mechanistic elegance lies in the stability of the TTF core under these acidic conditions; unlike many other organosulfur systems that might degrade or oxidize prematurely, the TTF unit remains intact, preserving its crucial pi-conjugated system. This preservation is essential for maintaining the redox activity required for the final polyurethane application, ensuring that the electronic properties are intrinsic to the polymer backbone rather than being degraded during synthesis.
From an impurity control perspective, this mechanism offers distinct advantages that are vital for producing high-purity OLED material or electronic chemical precursors. The initial phosphine-mediated step generates a solid precipitate that can be filtered, effectively removing soluble phosphine oxides and unreacted starting materials before the second stage even begins. In the transesterification phase, the primary byproduct is methanol, which is volatile and easily removed during the concentration step, simplifying the downstream purification burden. The use of silica gel column chromatography as the final purification step is highly effective because the polarity difference between the starting dimethyl ester and the final diol product is significant due to the introduction of multiple hydroxyl groups. This allows for the separation of any mono-substituted intermediates or unreacted starting materials, ensuring that the final product meets the stringent purity specifications required for electronic applications. The result is a clean impurity profile that minimizes the risk of charge trapping sites in the final polymer, a common failure mode in organic electronics.
How to Synthesize Tetrathiafulvalene Diol Efficiently
Implementing this synthesis requires careful attention to the stoichiometry and thermal profiles outlined in the patent data to maximize yield and reproducibility. The process is designed to be operationally simple, utilizing standard laboratory glassware and common solvents, which facilitates easy technology transfer from R&D to pilot plant scales. The initial cyclization step demands strict temperature control to ensure the formation of the correct dithiole intermediate, while the subsequent ester exchange benefits from prolonged heating to drive the equilibrium towards the diol product. Detailed standardized operating procedures for each reaction stage, including specific workup protocols for the removal of sulfuric acid and residual solvents, are essential for consistent batch-to-batch quality. Adhering to these optimized parameters ensures that the unique redox properties of the TTF unit are fully preserved in the final diol structure, ready for polymerization.
- Synthesize tetrathiafulvalene dimethyl ester by reacting methyl propiolate with carbon disulfide and tributylphosphine in THF at low temperatures.
- Perform transesterification by reacting the dimethyl ester intermediate with ethylene glycol or tetraethylene glycol using sulfuric acid catalysis.
- Purify the final tetrathiafulvalene diol product via silica gel column chromatography to ensure high purity for polymer applications.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement professionals evaluating the viability of this technology, the economic implications of the CN112812096B process are profound, primarily driven by the simplification of the supply chain and the reduction of processing steps. The reliance on methyl propiolate and carbon disulfide as starting materials leverages existing global chemical infrastructure, meaning these inputs are readily available from multiple suppliers, mitigating the risk of single-source dependency. Furthermore, the elimination of expensive transition metal catalysts or rare ligands, which are often required in alternative cross-coupling strategies for TTF functionalization, results in a drastic reduction in raw material costs. The process also avoids the use of cryogenic reagents in the second step, significantly lowering energy consumption associated with cooling and heating cycles. These factors combine to create a manufacturing profile that is not only cost-effective but also resilient to market fluctuations in specialty chemical pricing.
- Cost Reduction in Manufacturing: The transesterification strategy fundamentally alters the cost structure by replacing complex multi-step functionalization with a single-pot reaction using commodity glycols. By utilizing sulfuric acid as the catalyst instead of precious metals, the process eliminates the need for costly metal scavenging steps and the associated waste disposal fees. The high atom economy of the reaction means that a larger proportion of the input mass ends up in the final product, reducing the cost per kilogram of the active pharmaceutical ingredient or functional additive. Additionally, the ability to use the glycol as the solvent removes the need for purchasing and recovering large volumes of dedicated reaction solvents, further driving down operational expenditures.
- Enhanced Supply Chain Reliability: The robustness of the synthetic route ensures a stable supply of these critical intermediates, which is essential for manufacturers of high-performance polyurethanes. Since the key reagents are bulk chemicals with established production capacities globally, the risk of supply disruption due to geopolitical issues or manufacturer shutdowns is significantly minimized. The simplicity of the purification process, relying on standard silica chromatography rather than preparative HPLC or crystallization from exotic solvent systems, means that production throughput can be increased rapidly to meet surging demand without requiring capital-intensive equipment upgrades. This agility allows suppliers to respond quickly to market needs, ensuring continuous availability for downstream polymer producers.
- Scalability and Environmental Compliance: From an environmental and safety standpoint, this process offers a cleaner alternative to traditional organosulfur synthesis. The avoidance of pyrophoric reagents and the use of relatively benign solvents like dichloromethane and THF, which have well-established recycling protocols, simplifies compliance with increasingly strict environmental regulations. The generation of methanol as the primary byproduct allows for easy recovery and reuse, contributing to a circular economy approach within the manufacturing facility. The scalability is further enhanced by the fact that the reaction does not generate significant amounts of inorganic salts or heavy metal waste, reducing the burden on wastewater treatment plants and lowering the overall environmental footprint of the production facility.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these tetrathiafulvalene derivatives, based on the specific embodiments and data provided in the patent documentation. Understanding these details is crucial for technical teams assessing the feasibility of integrating these monomers into their existing polymer formulations. The answers reflect the practical realities of scaling this chemistry and the performance characteristics observed in the experimental examples. These insights provide a clear picture of the value proposition offered by this novel synthetic methodology.
Q: What are the primary advantages of this transesterification method for TTF diols?
A: The method described in patent CN112812096B offers a significantly simplified workflow compared to traditional multi-step functionalizations. By utilizing a direct ester exchange reaction with commercially available glycols, the process eliminates the need for complex protecting group strategies and reduces the number of isolation steps, leading to improved overall yield and easier purification via standard column chromatography.
Q: Can these TTF diol intermediates be scaled for industrial polyurethane production?
A: Yes, the synthetic route is highly amenable to scale-up. The starting materials, such as methyl propiolate and carbon disulfide, are bulk commodity chemicals, and the reaction conditions utilize standard solvents like THF and dichloromethane. The absence of exotic catalysts or extreme pressure requirements makes the transition from laboratory gram-scale to metric-ton production feasible for specialized chemical manufacturers.
Q: What specific redox properties do these compounds exhibit?
A: The resulting tetrathiafulvalene diol compounds retain the characteristic reversible redox behavior of the TTF core. Cyclic voltammetry data indicates distinct oxidation and reduction peaks, confirming their ability to undergo two-electron charge transfer reactions. This makes them ideal candidates for creating electroactive polymers, molecular switches, and organic electrode materials where controlled electron transport is critical.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrathiafulvalene Diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of redox-active polymers and are uniquely positioned to support your development of next-generation electronic materials. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We understand that the integrity of the TTF core is non-negotiable for your application, which is why our stringent purity specifications and rigorous QC labs are designed to detect and eliminate even trace impurities that could compromise electronic performance. Our commitment to quality assurance means that every batch of tetrathiafulvalene diol we deliver meets the highest standards of consistency and reliability required by the global electronics and specialty polymer industries.
We invite you to collaborate with us to unlock the full potential of this technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how our optimized process can improve your bottom line. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in organosulfur chemistry can become a strategic asset for your supply chain.
