Advanced Diaryl Pyrazole Synthesis: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
Advanced Diaryl Pyrazole Synthesis: Enhancing Purity and Scalability for Global Pharmaceutical Supply Chains
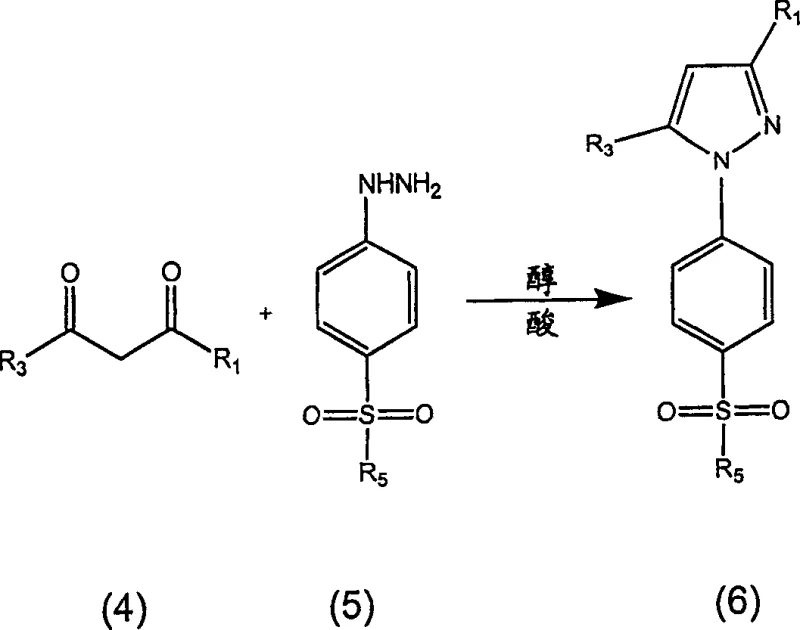
The pharmaceutical industry continuously seeks robust manufacturing pathways for high-value anti-inflammatory agents, particularly within the class of COX-2 inhibitors. A pivotal advancement in this domain is detailed in patent CN1671669A, which discloses a novel process for the qualitative preparation of 3-haloalkyl-1H-pyrazoles suitable for efficient, high payload commercial application. This technology specifically addresses the longstanding challenges associated with the synthesis of celecoxib and its analogs, offering a route that significantly mitigates the formation of regioisomeric and hydroxy regioisomeric by-products. For R&D directors and process chemists, the core innovation lies in the precise manipulation of reaction kinetics through solvent selection, water content control, and acid catalysis. By shifting away from traditional amide-based solvent systems and multi-step isolation protocols, this method enables the production of crystalline reaction products comprising at least about 98 weight percent of 1-(4-sulfonylphenyl)pyrazole. The implications for supply chain stability are profound, as the process allows for increased batch sizes and reduced cycle times while maintaining stringent purity specifications required for active pharmaceutical ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of pyrazoles via the condensation of diketones and hydrazines has been plagued by scalability issues and impurity profiles that complicate downstream purification. Prior art methods, such as those described in WO 00/42021, often rely on amide solvents which result in the formation of pyrazole solvates. These solvates necessitate additional processing steps, including isolation and recrystallization from secondary solvent systems like isopropanol and water, to yield the unsolvated active pharmaceutical ingredient. Furthermore, earlier scalable two-step processes, while effective for initial production, suffer from the generation of approximately 2-5 weight percent of regioisomer and hydroxy regioisomer by-products under commercial conditions. These impurities are structurally similar to the target molecule, making separation via crystallization difficult and often requiring multiple recrystallization cycles to achieve pharmacopeial purity. Each additional crystallization step inherently reduces overall yield due to product loss in the mother liquor and extends the production cycle time, thereby negatively impacting productivity and increasing the cost of goods sold. The reliance on solvents like tetrahydrofuran (THF) or methyl tert-butyl ether (MTBE) in some legacy processes also introduces potential safety and environmental concerns regarding peroxide formation and volatility.
The Novel Approach
The methodology outlined in the present invention fundamentally re-engineers the condensation reaction to favor the desired kinetic pathway while suppressing competing side reactions. Instead of isolating the diketone intermediate, the process utilizes a substantially anhydrous single solvent system, preferably comprising lower alcohols such as isopropanol or tert-butanol. A critical differentiator is the controlled introduction of the hydrazine source, specifically 4-sulfamoylphenylhydrazine (4-SAPH), in the presence of a strong acid with a pKa of less than about 3. This acidic environment not only facilitates the dissolution of the hydrazine salt but also kinetically inhibits the formation of regioisomers. The process allows for reactions to be carried out at high concentrations, enabling operation at high load—up to about 20% weight by weight—which drastically increases production capacity within existing reactor volumes. By eliminating the need for amide solvents and reducing the number of crystallization steps to a single direct crystallization, the novel approach achieves a substantial reduction in solvent usage and waste generation. This streamlined workflow translates directly into operational efficiency, allowing manufacturers to increase batch sizes by up to about 75% and reduce cycle times significantly compared to prior art processes.
Mechanistic Insights into Acid-Catalyzed Cyclization and Impurity Control
Understanding the mechanistic underpinnings of this synthesis is crucial for R&D teams aiming to replicate or optimize the process. The formation of 3-haloalkyl-1H-pyrazoles involves the condensation of a 1,3-diketone with a hydrazine. However, the selectivity of this reaction is highly sensitive to the electronic environment of the diketone. The diketone exists in equilibrium between keto and enol tautomeric forms. In the presence of water, a competitive hydration reaction occurs where water molecules attack the carbonyl carbon. Crucially, water preferentially reacts with the carbonyl group attached to the more electronegative substituent, such as the trifluoromethyl group in celecoxib precursors. This hydration creates a gem-diol or hydrate species that effectively blocks that specific carbonyl center from reacting with the hydrazine. Consequently, the hydrazine is forced to attack the remaining available carbonyl group (the one attached to the phenyl ring), leading to the formation of the undesired regioisomer. This mechanistic insight explains why controlling water content in the diketone source material is paramount; keeping the water content to less than about 30 equivalents based on the diketone minimizes hydrate formation and preserves the correct regioselectivity.
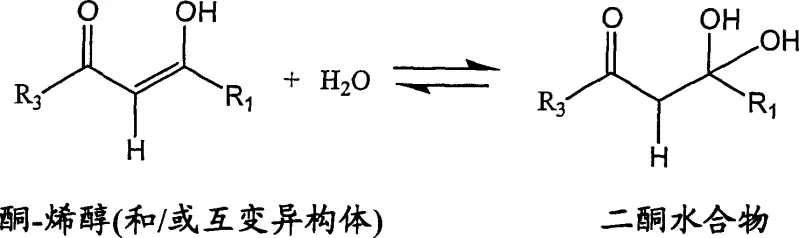
Furthermore, the concentration of dissolved hydrazine in the reaction mixture plays a pivotal role in impurity profile management. Kinetic studies suggest that the formation of the desired celecoxib is approximately first-order with respect to hydrazine concentration, whereas the formation of the hydroxy regioisomer by-product appears to be second-order. This implies that high local concentrations of hydrazine disproportionately accelerate by-product formation. The patented process addresses this by controlling the rate of addition of the hydrazine source or by utilizing a hydrazine salt with limited solubility that dissolves in situ at a rate matching the consumption by the reaction. Additionally, the use of strong acids like trifluoroacetic acid (TFA) serves to maintain a low pH environment (below 2.5), which further suppresses the rate of by-product formation relative to the main product. The combination of low water activity, controlled hydrazine concentration, and strong acid catalysis creates a kinetic window where the desired cyclization pathway is overwhelmingly favored, resulting in crude products with exceptionally low levels of regioisomers, often less than 0.5%.
How to Synthesize High-Purity Celecoxib Efficiently
The implementation of this synthesis route requires careful attention to reagent quality and addition protocols to maximize the benefits of the kinetic control mechanisms described above. The process is designed to be robust enough for industrial scale-up while maintaining the precision necessary for high-purity output. Operators must ensure that the diketone source, typically prepared as a metal salt in an alcoholic solvent, is kept substantially anhydrous prior to the introduction of the hydrazine component. The reaction is typically conducted at temperatures ranging from 35°C to 70°C, with a preference for 50°C to 60°C to balance reaction rate and selectivity. The following guide outlines the standardized approach for executing this transformation effectively.
- Prepare a reaction mixture containing a 1,3-dione source (such as a metal salt of 4-methylphenyl-1,1,1-trifluoro-2,4-butanedione) in an alcoholic solvent like isopropanol, ensuring water content is strictly controlled.
- Combine the diketone solution with a source of 4-sulfonylphenylhydrazine (e.g., a halide salt) in the presence of a strong acid with a pKa below 3, such as trifluoroacetic acid.
- Maintain the reaction temperature between 35°C and 70°C to favor pyrazole formation over hydration byproducts, followed by a single crystallization step to isolate the product with greater than 98% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis technology offers tangible benefits that extend beyond mere chemical yield. The shift to a single-solvent system based on lower alcohols eliminates the dependency on expensive and potentially hazardous amide solvents or ethers, simplifying solvent recovery and recycling infrastructure. This change alone contributes to significant cost reduction in API intermediate manufacturing by lowering raw material costs and reducing the complexity of waste treatment protocols. Moreover, the ability to operate at high payload concentrations means that manufacturers can produce the same quantity of product in smaller reactor volumes or significantly increase throughput in existing facilities without capital expenditure on new equipment. This flexibility enhances supply chain reliability, allowing suppliers to respond more agilely to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of multiple crystallization steps and solvent swaps drastically reduces energy consumption and labor hours associated with purification. By achieving high purity through a single direct crystallization from the reaction mixture, the process minimizes product loss typically incurred during repeated recrystallizations. Furthermore, the use of commodity alcohols like isopropanol instead of specialized amide solvents lowers the overall material cost base. The reduction in solvent volume per unit of product also decreases the load on solvent recovery distillation columns, leading to lower utility costs for steam and cooling water.
- Enhanced Supply Chain Reliability: The robustness of the reaction conditions, particularly the tolerance for controlled water levels in the hydrazine feed (while keeping the diketone dry), simplifies raw material specifications and logistics. Suppliers do not need to source ultra-dry hydrazine salts, which can be costly and difficult to handle, as the process accommodates aqueous hydrazine sources provided they are added correctly. This flexibility broadens the supplier base for key starting materials, reducing the risk of supply disruptions. Additionally, the improved particle size distribution of the final product, typically ranging from 40 μm to 100 μm, ensures excellent flow characteristics and filtration rates, preventing bottlenecks in the isolation and drying stages of production.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as mixing, heating, and filtration, without requiring exotic catalysts or extreme pressure conditions. The reduction in solvent usage and the avoidance of chlorinated or amide solvents align with green chemistry principles, facilitating easier compliance with increasingly stringent environmental regulations. The generation of less hazardous waste streams simplifies disposal and lowers the environmental footprint of the manufacturing site. This sustainability advantage is increasingly valued by downstream pharmaceutical customers who are auditing their supply chains for environmental, social, and governance (ESG) performance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this diaryl pyrazole synthesis pathway. These insights are derived directly from the experimental data and mechanistic studies presented in the patent literature, providing clarity on critical process parameters.
Q: Why is water content critical in the synthesis of 3-haloalkyl-1H-pyrazoles?
A: Water reacts competitively with the diketone starting material, specifically attacking the carbonyl group attached to the electronegative haloalkyl group (like CF3). This forms a hydrate that blocks the correct reaction site, forcing the hydrazine to attack the wrong carbonyl and generating difficult-to-remove regioisomer impurities.
Q: What is the role of strong acids like trifluoroacetic acid in this process?
A: Strong acids with a pKa below 3 serve multiple functions: they solubilize the hydrazine salt in the organic solvent, neutralize the basic diketone salt, and kinetically suppress the formation of byproducts. Kinetic studies indicate that stronger acids significantly reduce the rate of regioisomer formation without negatively impacting the main product yield.
Q: How does this process improve purification efficiency compared to conventional methods?
A: By minimizing the formation of regioisomers through strict water control and acid catalysis, the crude product contains significantly fewer impurities. This allows for isolation via a single direct crystallization step from an alcoholic solvent system, eliminating the need for multiple recrystallizations or complex solvent swaps required in older amide-based processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Celecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the supply of pharmaceutical intermediates. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated kinetic controls required for this pyrazole synthesis are maintained at every scale. We utilize state-of-the-art rigorous QC labs to verify stringent purity specifications, guaranteeing that our celecoxib intermediates meet the exacting standards required for global regulatory filings. Our commitment to process excellence means we can deliver high-purity materials with minimal regioisomer content, supporting your drug development and commercial manufacturing timelines with confidence.
We invite you to engage with our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of how our optimized synthesis routes can reduce your overall cost of goods. We are prepared to provide specific COA data and route feasibility assessments to demonstrate our capability to serve as a long-term strategic partner for your supply chain.
