Advanced Diaryl Pyrazole Synthesis: High-Purity Manufacturing and Commercial Scalability
Advanced Diaryl Pyrazole Synthesis: High-Purity Manufacturing and Commercial Scalability
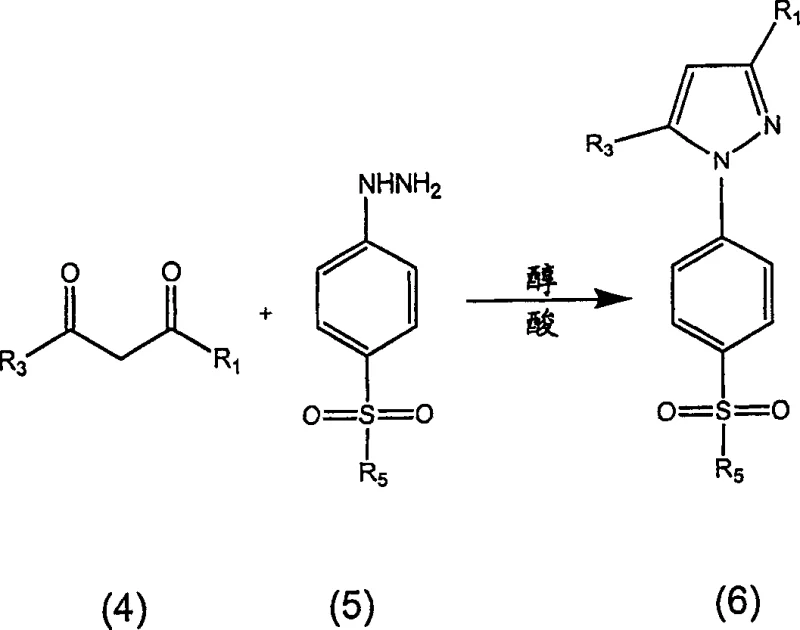
The pharmaceutical industry continuously seeks robust manufacturing processes for critical anti-inflammatory agents, particularly within the class of COX-2 inhibitors. Patent CN1671669A introduces a transformative methodology for the qualitative preparation of 3-haloalkyl-1H-pyrazoles, specifically targeting the efficient production of celecoxib and its analogs. This technology addresses long-standing challenges in impurity profiles, specifically the formation of regioisomers and hydroxy regioisomers that plague traditional condensation routes. By leveraging a sophisticated understanding of reaction kinetics and solvent interactions, this process enables the production of crystalline reaction products comprising at least about 98 weight percent of the desired 1-(4-sulfonylphenyl) pyrazole. For R&D directors and procurement specialists, this represents a significant opportunity to enhance supply chain reliability and reduce the cost burden associated with complex purification sequences in API intermediate manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of pyrazoles via the condensation of diketones and hydrazines has been fraught with scalability and purity issues. Prior art methods, such as those described in WO 00/42021, often rely on amide solvents which result in the formation of solvates that require complex isolation and recrystallization steps from mixed solvent systems like isopropanol and water. Furthermore, earlier scalable processes described in US patents 5,892,053 and 5,910,597, while effective for initial production, suffer from the generation of approximately 2-5 weight percent of regioisomer and hydroxy regioisomer by-products under commercial conditions. These impurities are chemically similar to the target molecule, making separation difficult and typically necessitating multiple crystallization steps. The second crystallization not only extends the production cycle time, negatively impacting productivity, but also reduces overall yield as some product remains unrecovered in the mother liquor. Additionally, the use of solvents like THF or MTBE in previous iterations poses potential safety and environmental concerns that modern green chemistry initiatives strive to eliminate.
The Novel Approach
The innovative process disclosed in the patent data overcomes these limitations by utilizing a single solvent system, preferably comprising lower alcohols such as isopropanol or tert-butanol, which eliminates the need for problematic amides or ethers. A key breakthrough is the realization that controlling the water content in the 1,3-dione source material to less than about 30 equivalents based on 1 equivalent of 1,3-dione drastically reduces the formation of hydration by-products. The method employs a strong acid catalyst, preferably trifluoroacetic acid (TFA) with a pKa below 3, to maintain an acidic environment that kinetically favors the formation of the desired pyrazole over competing by-product pathways. This approach allows for high payload operations with celecoxib loadings of at least 10% to 20% (w/w), significantly increasing batch sizes in existing production plants. The result is a streamlined workflow where high-purity celecoxib can be isolated via a single direct crystallization, offering substantial advantages in both operational efficiency and environmental compliance.
Mechanistic Insights into Acid-Catalyzed Cyclization and Impurity Control
A deep mechanistic understanding reveals why this specific combination of reagents and conditions yields superior results. The 1,3-diketone reactant exists in equilibrium between three different tautomeric species, as illustrated in the patent data. Crucially, water acts as a competitive nucleophile that reacts preferentially with the carbonyl carbon bearing the more electronegative substituent, such as the trifluoromethyl group in the case of celecoxib precursors. When water attacks this site, it forms a diketone hydrate which effectively blocks that carbonyl center. Consequently, the hydrazine nucleophile is forced to attack the remaining available carbonyl center (the phenyl-substituted side), leading to the formation of the undesired regioisomer. By strictly limiting the water content in the diketone source prior to the introduction of the hydrazine, the formation of this blocking hydrate is minimized, thereby preserving the correct reactivity profile for high-selectivity cyclization.
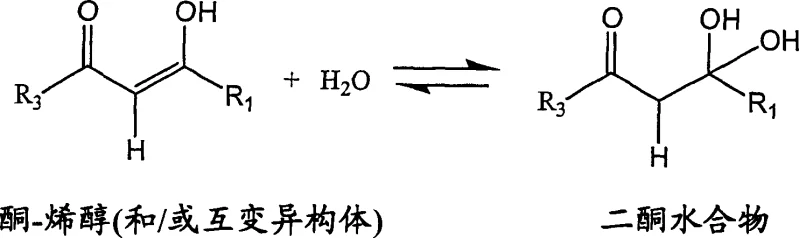
Furthermore, the concentration of dissolved hydrazine plays a pivotal role in determining the impurity profile. Kinetic studies suggest that the formation of the desired celecoxib is approximately first-order with respect to hydrazine concentration, whereas the formation of the hydroxy regioisomer appears to be second-order. This implies that high local concentrations of hydrazine disproportionately favor by-product generation. The novel process mitigates this by controlling the rate of addition or utilizing hydrazine salts with limited solubility, ensuring that the molar equivalent ratio of dissolved hydrazine to diketone remains below about 2:1 throughout the reaction. Additionally, the use of strong acids like TFA not only solubilizes the hydrazine salt but also suppresses the rate of by-product formation inversely proportional to acid concentration, while leaving the main reaction rate relatively unaffected. This dual mechanism of water exclusion and kinetic control via acid catalysis ensures the production of high-purity intermediates suitable for direct pharmaceutical application.
How to Synthesize Celecoxib Efficiently
The synthesis of celecoxib via this improved route involves the condensation of a metal salt of a 1,3-diketone, such as the sodium salt of 4-methylphenyl-1,1,1-trifluoro-2,4-butanedione, with a 4-sulfamoylphenylhydrazine salt in an alcoholic solvent. The process is characterized by the in situ generation or addition of a strong acid to facilitate the reaction and control impurity levels. Detailed operational parameters regarding temperature control, addition rates, and crystallization protocols are essential for replicating the high yields and purity described in the patent literature. The following guide outlines the standardized synthesis steps derived from the preferred embodiments of the technology.
- Prepare a reaction mixture containing a 1,3-dione source (such as a metal salt of 4-methylphenyl-1,1,1-trifluoro-2,4-butanedione) in an alcoholic solvent like isopropanol.
- Combine the diketone source with a 4-sulfonylphenylhydrazine salt (e.g., hydrochloride) in the presence of a strong acid with a pKa below 3, such as trifluoroacetic acid.
- Maintain the dissolved hydrazine to diketone molar ratio below 2: 1 during reaction to minimize regioisomer formation, followed by crystallization via pH adjustment and cooling.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and logistical benefits driven by process intensification and waste reduction. The ability to operate at significantly higher loadings means that the same volume of reactor capacity can produce a much larger mass of finished product, effectively expanding manufacturing capability without capital expenditure on new equipment. The elimination of multiple crystallization steps not only reduces the consumption of solvents and energy but also shortens the overall cycle time, allowing for faster turnaround on purchase orders. Furthermore, the shift away from hazardous solvents like THF and amides simplifies waste disposal protocols and reduces the regulatory burden associated with volatile organic compound (VOC) emissions, aligning with increasingly stringent global environmental standards.
- Cost Reduction in Manufacturing: The process achieves cost optimization primarily through the elimination of expensive and difficult-to-remove solvents, as well as the reduction of unit operations. By avoiding the need for multiple recrystallizations to remove regioisomers, the facility saves significantly on solvent purchase, recovery, and disposal costs. The high-yield nature of the reaction, coupled with the ability to isolate product in a single step, minimizes material loss typically associated with purification stages. Additionally, the use of commodity alcohols like isopropanol instead of specialized amide solvents further drives down raw material expenses, creating a more resilient cost structure for the final API intermediate.
- Enhanced Supply Chain Reliability: The robustness of this chemical route enhances supply security by reducing the complexity of the manufacturing process. Fewer processing steps mean fewer points of failure and less variability in batch-to-batch quality. The process produces celecoxib with large particle sizes, typically ranging from 40 μm to 100 μm, which significantly improves the efficiency of solid-liquid separation operations like filtration and centrifugation. This physical characteristic prevents filter clogging and ensures consistent drying rates, reducing the risk of production delays caused by equipment bottlenecks. Consequently, suppliers can maintain tighter delivery schedules and respond more agilely to fluctuations in market demand.
- Scalability and Environmental Compliance: From a sustainability perspective, the process offers a greener alternative to legacy methods by utilizing safer solvent systems and generating less chemical waste. The high concentration operation reduces the total volume of solvent required per kilogram of product, directly lowering the carbon footprint of the manufacturing process. The avoidance of chlorinated solvents and amides simplifies the effluent treatment process, making it easier for manufacturing sites to comply with local environmental regulations. This environmental compatibility ensures long-term operational continuity and reduces the risk of shutdowns due to regulatory non-compliance, securing the supply chain for downstream pharmaceutical partners.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this diaryl pyrazole synthesis technology. These answers are derived directly from the experimental data and mechanistic explanations provided in the patent documentation, offering clarity on critical process parameters.
Q: Why is water content critical in the synthesis of 3-haloalkyl-1H-pyrazoles?
A: Water reacts competitively with the diketone reactant, specifically attacking the carbonyl carbon bearing the more electronegative group (such as the trifluoromethyl group). This hydration forms a blocking group that forces the hydrazine to react at the incorrect carbonyl center, significantly increasing the formation of difficult-to-remove regioisomeric by-products.
Q: What is the role of strong acids like trifluoroacetic acid in this process?
A: Strong acids with a pKa below approximately 3 serve multiple functions: they solubilize the hydrazine salt in the organic solvent, neutralize the basic diketone solution, and kinetically suppress the formation of by-products. Kinetic studies indicate that while pyrazole formation is relatively unaffected by acid strength, by-product formation rates decrease significantly as acid strength increases.
Q: How does this process improve purification efficiency compared to conventional methods?
A: By minimizing the formation of regioisomers through strict control of water and hydrazine concentration, the process allows for the isolation of high-purity product (>98%) via a single direct crystallization step. Conventional methods often require multiple recrystallizations to remove regioisomers, which reduces overall yield and increases processing time.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Celecoxib Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of purity and consistency in the production of pharmaceutical intermediates like celecoxib. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated kinetic controls required for this synthesis are maintained at an industrial level. We utilize stringent purity specifications and rigorous QC labs to verify that every batch meets the exacting standards required for API manufacturing, including the minimization of regioisomeric impurities to undetectable levels. Our commitment to quality assurance guarantees that our clients receive materials that facilitate smooth downstream processing and final drug product registration.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, ensuring that your project moves forward with the most efficient and reliable chemical strategy available in the market.
