Advanced Manufacturing of 4-Amino-1-Naphthol Ethers for High-Value Pharmaceutical Intermediates
Advanced Manufacturing of 4-Amino-1-Naphthol Ethers for High-Value Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks more efficient pathways to synthesize complex heterocyclic intermediates, particularly those serving as precursors for active pharmaceutical ingredients (APIs). Patent CN1547568A introduces a transformative methodology for preparing 4-amino-1-naphthol ether compounds, addressing critical bottlenecks in traditional synthetic routes. This innovation shifts the paradigm from expensive, multi-step protection strategies to a streamlined process originating from readily available 1-naphthol derivatives. By leveraging a novel sequence of alkylation, selective acylation, and hydrolysis, this technology enables the production of high-purity intermediates essential for modern drug discovery and development. For R&D directors and procurement specialists, understanding this mechanistic shift is vital for optimizing supply chains and reducing the cost of goods sold (COGS) in competitive therapeutic areas.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-amino-1-naphthol ethers has been plagued by economic and operational inefficiencies inherent to starting materials like 4-amino-1-naphthol hydrochloride. Conventional routes, such as those disclosed in WO 00/43384, necessitate a cumbersome three-step sequence involving the initial protection of the amine functionality, followed by the alkylation of the hydroxyl group, and finally, the cleavage of the protecting group. This approach suffers from significant drawbacks, primarily the high cost of both the starting amino-naphthol and the specialized protecting group reagents required. Furthermore, each additional synthetic step introduces potential yield losses and complicates the impurity profile, creating substantial challenges for purification at a commercial scale. The reliance on protected derivatives also extends the overall processing time and increases the environmental footprint due to the generation of additional waste streams associated with protection and deprotection cycles.
The Novel Approach
In stark contrast, the methodology described in CN1547568A offers a streamlined alternative that begins with inexpensive 1-naphthol compounds, effectively bypassing the need for amine protection entirely. The process initiates with the direct alkylation of the naphthol hydroxyl group to form a 1-naphthol ether, followed by a highly selective conversion to the 4-acylamino derivative, and concludes with a simple hydrolytic cleavage to reveal the free amine. 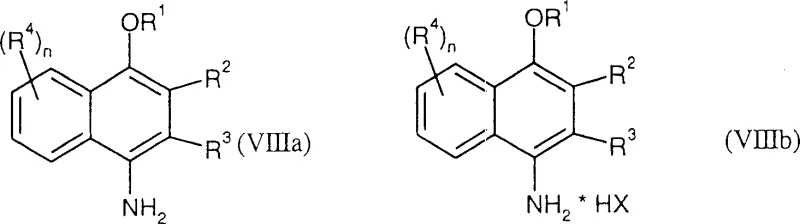 This strategic inversion of the synthetic order allows manufacturers to utilize commodity feedstocks while achieving superior regioselectivity at the 4-position of the naphthalene ring. The result is a robust pathway that significantly reduces raw material costs and simplifies the operational workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates. By eliminating the protection-deprotection burden, this approach enhances overall process mass intensity (PMI) and delivers a more sustainable manufacturing solution.
This strategic inversion of the synthetic order allows manufacturers to utilize commodity feedstocks while achieving superior regioselectivity at the 4-position of the naphthalene ring. The result is a robust pathway that significantly reduces raw material costs and simplifies the operational workflow, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates. By eliminating the protection-deprotection burden, this approach enhances overall process mass intensity (PMI) and delivers a more sustainable manufacturing solution.
Mechanistic Insights into Polyphosphoric Acid Mediated Acylation
The core of this technological breakthrough lies in the second step, where the 1-naphthol ether is converted into the corresponding 4-acylamino-1-naphthol ether using a specific combination of hydroxylamine salts, carboxylic acids, and polyphosphoric acid (PPA). This reaction environment facilitates an electrophilic aromatic substitution that is remarkably selective for the 4-position, even in the presence of other potential reactive sites on the naphthalene scaffold. The use of polyphosphoric acid, preferably with a content of 116% based on orthophosphoric acid, acts as both a solvent and a potent dehydrating catalyst, driving the equilibrium towards the formation of the acylamino species.  The reaction typically proceeds at temperatures between 70°C and 120°C, ensuring complete conversion while minimizing thermal degradation of the sensitive naphthyl ether backbone. This mechanistic precision is crucial for R&D teams aiming to minimize downstream purification burdens, as high regioselectivity directly correlates with a cleaner crude product profile.
The reaction typically proceeds at temperatures between 70°C and 120°C, ensuring complete conversion while minimizing thermal degradation of the sensitive naphthyl ether backbone. This mechanistic precision is crucial for R&D teams aiming to minimize downstream purification burdens, as high regioselectivity directly correlates with a cleaner crude product profile.
Furthermore, the control of impurities is intrinsically built into the reaction design through the careful selection of reagents and conditions. The use of acetic acid or trifluoroacetic acid as the acyl source, combined with hydroxylamine hydrochloride, generates the active acylating species in situ without introducing extraneous metal contaminants often associated with transition metal catalysis. The subsequent workup involves quenching the reaction mixture with ice and adjusting the pH to precipitate the product, a technique that effectively separates the desired 4-acylamino compound from unreacted starting materials and acidic byproducts. This precise control over the reaction milieu ensures that the final 4-amino-1-naphthol ether derivatives meet stringent purity specifications, often exceeding 99% as demonstrated in the patent examples. Such high purity is paramount for pharmaceutical intermediates, where trace impurities can impact the safety and efficacy of the final drug substance.
How to Synthesize 4-Amino-1-Naphthol Ether Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for translating this chemistry from the laboratory bench to pilot and commercial production scales. The process is divided into three distinct operational phases: the initial etherification of 1-naphthol, the PPA-mediated acylation, and the final hydrolytic deprotection. Each step utilizes standard chemical engineering unit operations, such as stirred tank reactors for the alkylation and controlled heating systems for the acylation phase. The detailed standardized synthesis steps below provide a technical overview of the critical parameters, including stoichiometry, temperature profiles, and workup procedures necessary to replicate the high yields and purities reported in the intellectual property. Adhering to these guidelines ensures consistent quality and reproducibility, which are essential for regulatory compliance in the pharmaceutical supply chain.
- Step 1: Alkylation of 1-naphthol with reactive alkyl compounds (e.g., N-(2-chloroethyl)morpholine) using NaOH in ethanol at 40-80°C.
- Step 2: Conversion to 4-acylamino derivative using hydroxylamine salt, carboxylic acid, and polyphosphoric acid at 70-120°C.
- Step 3: Acidic hydrolysis of the acyl group using concentrated HCl at 95°C to yield the final amine salt or free base.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers compelling economic and logistical advantages over legacy methods. The primary driver of cost reduction is the substitution of expensive protected amino-naphthol starting materials with commodity-grade 1-naphthol, which is widely available and priced significantly lower in the global chemical market. Additionally, the elimination of protection and deprotection reagents removes a substantial portion of the raw material bill, while simultaneously reducing the volume of solvents and consumables required for purification. This streamlined material usage translates directly into a lower cost of goods sold (COGS), allowing pharmaceutical companies to improve their margins or pass savings on to healthcare systems. The process relies on common solvents like ethanol and water, further mitigating supply chain risks associated with specialized or hazardous solvent availability.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived from the fundamental simplification of the synthetic tree. By removing the need for amine protecting groups, manufacturers avoid the purchase of costly reagents and the associated waste disposal fees. The use of polyphosphoric acid, while requiring careful handling, is a bulk chemical that offers high efficiency per unit cost compared to specialized coupling agents. Furthermore, the high yields reported in the patent examples, such as the 93.8% yield in the alkylation step and quantitative conversion in the hydrolysis step, maximize the throughput of the manufacturing facility. This efficiency means that less raw material is wasted, and the overall capacity of the plant is utilized more effectively, leading to substantial cost savings in pharmaceutical intermediate manufacturing without compromising on quality standards.
- Enhanced Supply Chain Reliability: From a supply chain perspective, the reliance on ubiquitous raw materials like 1-naphthol, sodium hydroxide, and acetic acid ensures a high degree of supply security. Unlike processes dependent on custom-synthesized building blocks that may have single-source suppliers, the inputs for this method are produced by multiple vendors globally, reducing the risk of shortages or price volatility. The robustness of the reaction conditions, which operate at ambient pressure and moderate temperatures, also means that the process can be executed in a wide range of manufacturing facilities without requiring specialized high-pressure equipment. This flexibility allows for diversified sourcing strategies and easier technology transfer between different contract development and manufacturing organizations (CDMOs), ensuring continuity of supply for critical drug programs.
- Scalability and Environmental Compliance: The scalability of this process is supported by its use of well-understood chemical transformations that do not involve exotic catalysts or extreme conditions. The exothermic nature of the reactions is manageable with standard cooling systems, and the workup procedures involve straightforward filtration and crystallization steps that are easily scaled from kilograms to metric tons. Environmentally, the process benefits from the avoidance of heavy metal catalysts, which simplifies the removal of trace metals to meet strict regulatory limits for APIs. The aqueous workups and the potential for solvent recovery, particularly of ethanol, contribute to a greener manufacturing profile. This alignment with green chemistry principles not only reduces the environmental footprint but also simplifies regulatory filings and environmental permitting, facilitating faster time-to-market for new pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios. The answers reflect the practical realities of scaling this chemistry, from reagent selection to impurity management.
Q: How does this process improve regioselectivity compared to conventional methods?
A: By utilizing polyphosphoric acid mediated acylation on the pre-formed ether, the process achieves high selectivity for the 4-position, avoiding the complex protection-deprotection sequences required when starting from amino-naphthols.
Q: What is the expected purity profile for commercial scale production?
A: The patent data demonstrates purities exceeding 99% (HPLC) for the acylated intermediate and 99.4% for the final free amine, indicating a robust impurity profile suitable for pharmaceutical applications.
Q: Are the raw materials readily available for large-scale manufacturing?
A: Yes, the process relies on commodity chemicals such as 1-naphthol, sodium hydroxide, ethanol, and acetic acid, ensuring a stable and cost-effective supply chain without reliance on exotic reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Amino-1-Naphthol Ether Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of life-saving medicines. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial manufacturing is seamless. We are committed to delivering high-purity 4-amino-1-naphthol ether intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our facility is equipped to handle the specific requirements of polyphosphoric acid chemistry and multi-step organic synthesis, guaranteeing a reliable supply of this valuable pharmaceutical intermediate for your drug development pipeline.
We invite you to collaborate with us to leverage this advanced technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing expertise can optimize your supply chain and reduce overall production costs. Let us be your partner in bringing high-quality chemical solutions to the global market.
