Scalable Production of 4,4'-Dinitrobibenzyl via Advanced Oxidative Coupling Technology
Introduction to Advanced Oxidative Coupling Technology
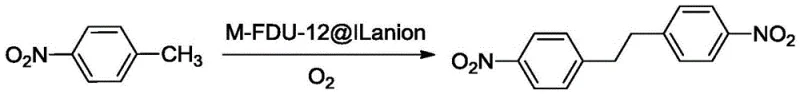
The chemical industry is constantly seeking more sustainable and efficient pathways for producing key intermediates like 4,4'-dinitrobibenzyl, a vital building block for pharmaceuticals, agrochemicals, and functional materials. A groundbreaking approach detailed in patent CN113636939A introduces a novel preparation method that utilizes p-nitrotoluene as the starting material and molecular oxygen as the oxidant. This technology represents a significant leap forward from traditional methods by employing a transition metal-doped FDU-12 mesoporous molecular sieve supported guanidine salt ionic liquid as a highly selective heterogeneous catalyst. The process operates under mild conditions, typically between 20-70°C, and achieves exceptional conversion rates while minimizing environmental impact through the use of ethanol as a green solvent.
This innovative route addresses critical pain points in fine chemical manufacturing, specifically the need for high purity and reduced waste generation. By leveraging the unique properties of the supported ionic liquid catalyst, the reaction proceeds via a selective oxidative dehydrogenation coupling mechanism. This ensures that the desired 4,4'-isomer is formed with high specificity, avoiding the complex mixtures often seen in non-selective radical reactions. The ability to easily separate the solid catalyst from the liquid product phase by simple hot filtration further enhances the practicality of this method for industrial applications, offering a robust solution for reliable pharma intermediate supplier networks seeking greener alternatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,4'-dinitrobibenzyl has relied on three primary methodologies, each fraught with significant operational and environmental drawbacks. The reduction coupling method, which typically employs p-nitrobenzyl halides and transition metal catalysts, often involves toxic solvents and generates substantial amounts of halogenated waste that requires costly disposal. Alternatively, the diphenylethane nitration method uses concentrated nitric acid or mixed acids, leading to severe corrosion issues, low selectivity, and the production of large volumes of hazardous waste acid that complicates downstream processing. Furthermore, electrochemical reduction methods, while avoiding some chemical oxidants, suffer from high energy consumption, complex cell designs, and difficulties in scaling up safely due to safety control challenges inherent in electrochemical processes.
Another common approach involves oxidative coupling using hydrogen peroxide; while greener than nitric acid, hydrogen peroxide presents stability issues and higher industrial costs compared to molecular oxygen. Existing oxygen oxidation methods reported in prior art have struggled with low conversion rates, poor yields, and the necessity for large amounts of waste alkali for neutralization, resulting in significant environmental pollution. These conventional pathways often require harsh reaction conditions, expensive reagents, and generate impurity profiles that are difficult to manage, ultimately driving up the cost of goods sold and creating supply chain vulnerabilities for manufacturers of high-purity OLED material or polymer additive precursors.
The Novel Approach
In stark contrast, the novel method disclosed in the patent utilizes a sophisticated heterogeneous catalytic system that transforms p-nitrotoluene directly into 4,4'-dinitrobibenzyl using air or pure oxygen. This oxidative dehydrogenation coupling reaction is catalyzed by a specialized material where transition metals such as Iridium, Manganese, or Titanium are doped into an FDU-12 mesoporous framework and functionalized with guanidinium ionic liquids. This design maximizes the active surface area and creates a microenvironment that favors the selective formation of the carbon-carbon bond between two p-nitrotoluene molecules. The use of ethanol as a solvent not only dissolves the reactants effectively but also aligns with green chemistry principles, being less toxic and easier to recover than chlorinated or aromatic solvents used in older methods.
The operational simplicity of this new approach cannot be overstated; the reaction proceeds efficiently at temperatures ranging from 40-60°C, significantly reducing energy consumption compared to high-temperature nitration or reduction processes. Post-reaction work-up is streamlined through a hot filtration step that cleanly separates the solid catalyst from the product solution, allowing the catalyst to be dried and reused immediately. This eliminates the need for complex extraction procedures or neutralization steps involving large quantities of base, thereby drastically simplifying the manufacturing workflow. For procurement teams, this translates to a process that is not only chemically superior but also logistically simpler, reducing the burden on waste treatment facilities and enhancing the overall sustainability profile of the supply chain.
Mechanistic Insights into Transition Metal-Doped FDU-12 Catalysis
The core of this technological breakthrough lies in the intricate design of the M-FDU-12@ILanion catalyst, which synergistically combines the structural stability of mesoporous silica with the catalytic activity of transition metals and ionic liquids. The FDU-12 support provides a highly ordered mesoporous structure with large pore sizes, facilitating the diffusion of reactant molecules to the active sites while preventing the aggregation of metal species. The guanidinium ionic liquid moiety, covalently anchored to the silica surface via a propyl linker, acts as a phase-transfer mediator and stabilizes the transition state during the oxidative coupling. The presence of transition metals like Ir, Mn, or Ti within the framework serves as the primary redox center, activating molecular oxygen to generate reactive oxygen species that abstract hydrogen atoms from the methyl group of p-nitrotoluene.
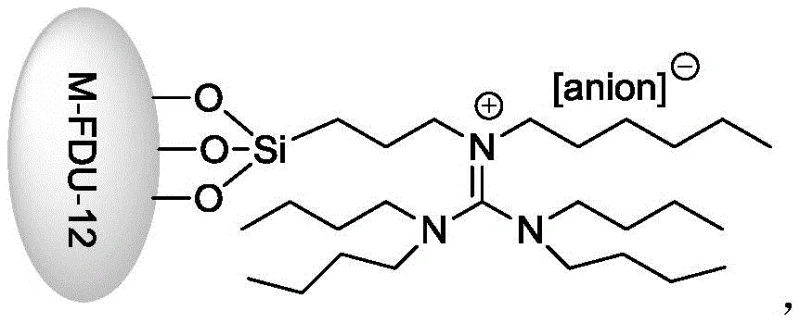
This activation leads to the formation of benzyl radicals which subsequently couple to form the bibenzyl skeleton. The specific anion associated with the ionic liquid, such as CoCl3, CuCl2, or CeCl4, plays a crucial role in modulating the electronic environment of the active site, thereby tuning the selectivity towards the 4,4'-isomer and suppressing over-oxidation to aldehydes or acids. The heterogeneous nature of the catalyst ensures that the reaction occurs primarily on the surface or within the pores, allowing for precise control over the reaction kinetics. This mechanistic precision results in high product purity, often exceeding 99%, as confirmed by HPLC analysis in the patent examples, which is critical for applications requiring stringent impurity specifications in pharmaceutical intermediates.
Furthermore, the robustness of the covalent bonding between the ionic liquid and the silica support prevents leaching of the active species into the reaction medium, a common failure mode in liquid-phase catalysis. This stability is evidenced by the catalyst's ability to maintain high activity over multiple recycling runs, with experimental data showing consistent yields even after six consecutive cycles. The thermal stability of the supported ionic liquid up to 200°C ensures that the catalyst can withstand the reaction conditions and the drying processes required for regeneration without degradation. For R&D directors, understanding this mechanism highlights the potential for adapting this catalytic platform to other oxidative coupling reactions, offering a versatile tool for developing new synthetic routes for complex organic molecules.
How to Synthesize 4,4'-Dinitrobibenzyl Efficiently
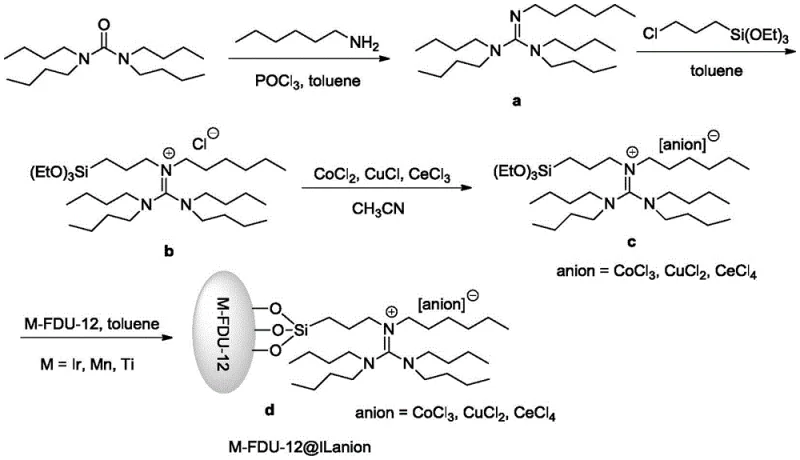
Implementing this synthesis route requires the prior preparation of the specialized catalyst, followed by the oxidative coupling reaction under controlled oxygen flow. The catalyst synthesis involves a multi-step organic modification of the FDU-12 support, starting with the formation of a guanidine precursor from tetrabutyl urea and phosphorus oxychloride, followed by silylation and metal complexation. Once the catalyst is prepared, the main reaction is straightforward: p-nitrotoluene is dissolved in ethanol, the catalyst is added, and oxygen is bubbled through the mixture at moderate temperatures. The process is designed to be scalable, with the patent demonstrating success on a 0.1 mol scale which can be readily extrapolated to pilot and commercial plant scales with appropriate engineering controls for gas-liquid mass transfer.
- Prepare the transition metal-doped FDU-12 mesoporous molecular sieve supported guanidine salt ionic liquid catalyst by reacting tetrabutyl urea, phosphorus oxychloride, and n-hexylamine, followed by silanization and metal salt complexation.
- Charge a reactor with p-nitrotoluene, ethanol solvent, and the prepared heterogeneous catalyst (1-15% mass ratio), then stir the mixture while introducing oxygen gas at a controlled flow rate.
- Maintain the reaction temperature between 40-60°C for 1-4 hours, then separate the catalyst phase from the product phase by hot filtration, cool the filtrate to crystallize the pure product, and recover the catalyst for reuse.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented technology offers compelling advantages that directly address the priorities of procurement managers and supply chain heads, particularly regarding cost efficiency and operational reliability. The shift from stoichiometric oxidants like nitric acid or hydrogen peroxide to molecular oxygen represents a fundamental reduction in raw material costs, as oxygen is abundant and inexpensive. Additionally, the elimination of corrosive acids and the associated need for specialized corrosion-resistant equipment (such as glass-lined reactors) lowers capital expenditure requirements for new production lines. The use of ethanol as a solvent further reduces costs compared to more expensive or regulated solvents, and its ease of recovery through distillation allows for closed-loop solvent management systems that minimize waste and purchase volume.
- Cost Reduction in Manufacturing: The heterogeneous nature of the catalyst allows for its recovery and reuse multiple times without significant loss of activity, drastically reducing the cost per kilogram of catalyst consumed. By eliminating the need for expensive transition metal complexes that are lost in homogeneous systems, the overall material cost is significantly lowered. Furthermore, the simplified work-up procedure, which avoids extensive washing and neutralization steps, reduces labor costs and utility consumption (water and steam), leading to substantial overall cost savings in the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on p-nitrotoluene, a widely available commodity chemical, ensures a stable and secure raw material supply, mitigating risks associated with specialty reagent shortages. The mild reaction conditions (40-60°C) reduce the risk of thermal runaways and safety incidents, ensuring continuous operation and minimizing unplanned downtime. The robustness of the catalyst and the simplicity of the process make it highly suitable for toll manufacturing or multi-purpose facilities, providing flexibility in sourcing and production planning for global supply chains.
- Scalability and Environmental Compliance: The process generates significantly less hazardous waste compared to traditional nitration or reduction methods, simplifying compliance with increasingly stringent environmental regulations. The absence of heavy metal waste streams and waste acid reduces the burden on wastewater treatment plants and lowers disposal costs. The scalability of the gas-liquid-solid reaction system is well-understood in the chemical industry, facilitating a smooth transition from laboratory development to commercial scale-up of complex pharmaceutical intermediates without major process redesigns.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this oxidative coupling technology. These answers are derived directly from the experimental data and technical disclosures within the patent documentation, providing a clear understanding of the process capabilities and limitations. Understanding these details is essential for technical teams evaluating the feasibility of adopting this method for their specific production needs.
Q: What are the primary advantages of using oxygen over traditional nitrating agents for this synthesis?
A: Using oxygen as the oxidant eliminates the generation of large quantities of waste acid associated with mixed acid nitration methods. It significantly reduces environmental hazards and post-treatment costs while maintaining high selectivity for the 4,4'-dinitrobibenzyl product.
Q: How does the M-FDU-12@ILanion catalyst improve process efficiency?
A: The catalyst combines the high surface area of the FDU-12 mesoporous support with the active sites of transition metals (Ir, Mn, Ti) and guanidinium ionic liquids. This heterogeneous structure allows for easy separation via hot filtration and enables the catalyst to be recycled multiple times without significant loss of activity.
Q: Is this method suitable for large-scale industrial manufacturing?
A: Yes, the method operates under mild conditions (40-60°C) and uses ethanol as a solvent, which is safer and more scalable than processes requiring strong acids or high-pressure hydrogenation. The simple work-up procedure facilitates continuous production and reduces operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,4'-Dinitrobibenzyl Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this green oxidative coupling technology for the production of high-value intermediates like 4,4'-dinitrobibenzyl. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of materials. Our state-of-the-art facilities are equipped to handle the specific requirements of heterogeneous catalysis and gas-liquid reactions, maintaining stringent purity specifications through our rigorous QC labs and advanced analytical capabilities. We are committed to delivering products that meet the highest quality standards required by the global pharmaceutical and agrochemical industries.
We invite you to collaborate with us to leverage this innovative synthesis route for your projects. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. Please contact our technical procurement team to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in green chemistry and process optimization can drive value and efficiency in your supply chain.
