Optimizing Methyl 4-Methylcinnamate Production for Commercial Scale-up and High Purity Standards
The pharmaceutical and fine chemical industries are constantly seeking robust synthetic routes for critical intermediates that balance high purity with economic feasibility. A significant advancement in this domain is detailed in patent CN102627559A, which outlines a novel preparation method for methyl 4-methylcinnamate, a vital precursor in the synthesis of antithrombotic agents such as ozagrel. This technology represents a paradigm shift from traditional base-catalyzed condensations, offering a streamlined, one-step process that utilizes lithium diisopropylamide (LDA) in glycol dimethyl ether (DME). For R&D directors and procurement specialists, understanding the mechanistic advantages of this route is crucial for evaluating long-term supply stability. The patent demonstrates that by strictly controlling reaction parameters, specifically maintaining temperatures below 5°C during addition and utilizing a non-protonic polar solvent, manufacturers can achieve yields that drastically outperform conventional sodium hydroxide or sodium alkoxide methods. This report analyzes the technical depth of this innovation to provide actionable insights for scaling production and optimizing procurement strategies for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of cinnamate derivatives via Claisen-Schmidt condensation has been plagued by significant inefficiencies that hinder commercial scalability and cost-effectiveness. Traditional protocols often rely on absolute ethanol as a solvent with 10% sodium hydroxide serving as the catalyst, a combination that frequently results in prolonged reaction times and suboptimal product yields. The presence of water in alkaline catalysts like NaOH limits their solubility in the organic phase, preventing effective contact with reactants and leading to incomplete conversions. Furthermore, the use of sodium alkoxide in methyl acetate solvents, while an improvement, still suffers from complicated post-treatment procedures and difficult purification challenges due to the formation of substantial by-products. A critical flaw in these legacy methods is the tendency for excessive methyl acetate to undergo self-condensation under strong alkaline conditions, generating impurities that are arduous to remove. These technical bottlenecks not only inflate manufacturing costs through material waste but also introduce variability in product quality, posing risks for downstream pharmaceutical applications where impurity profiles must be tightly controlled.
The Novel Approach
The innovative methodology presented in the patent data addresses these historical deficiencies by introducing a highly specific catalytic system centered on lithium diisopropylamide (LDA) and glycol dimethyl ether (DME). This approach fundamentally alters the reaction landscape by employing a strong, non-nucleophilic base that facilitates the rapid and selective formation of the lithium enolate from methyl acetate. By reversing the addition order—mixing the catalyst and ester before introducing p-tolualdehyde—the process effectively protects the carbonyl component from unwanted side reactions, ensuring the condensation proceeds along a single, desired direction. The use of DME as a solvent is particularly advantageous as it is a non-protonic polar solvent that solubilizes reactants effectively without forming hydrogen bonds that could inhibit the reaction mechanism. This strategic modification allows the reaction to proceed under mild conditions, typically between 0°C and 10°C, while achieving conversion rates that are substantially higher than those observed in ethanol-based systems. The result is a streamlined workflow that minimizes waste generation and simplifies the isolation of the final white solid product through straightforward distillation and recrystallization steps.
Mechanistic Insights into LDA-Catalyzed Claisen-Schmidt Condensation
At the core of this synthetic breakthrough is the precise control over enolate chemistry, which dictates the efficiency and selectivity of the condensation reaction. In conventional systems using weaker bases or protic solvents, the equilibrium often favors the starting materials or leads to thermodynamic mixtures that include self-condensation by-products. However, the utilization of LDA, a bulky and strong base, ensures the quantitative deprotonation of methyl acetate to form a kinetic lithium enolate. This species is highly reactive towards the electrophilic carbonyl carbon of p-tolualdehyde but is sterically hindered from attacking other ester molecules, thereby suppressing the formation of beta-keto esters that typically contaminate the product stream. The electron-donating methyl group on the aromatic ring of p-tolualdehyde generally slows down nucleophilic attack, but the high reactivity of the lithium enolate overcomes this electronic deactivation, accelerating the reaction rate significantly. This mechanistic precision is the key driver behind the observed yield improvements, transforming a sluggish process into a rapid and high-yielding transformation suitable for industrial throughput.
Furthermore, the impurity control mechanism inherent in this design is critical for meeting the stringent purity specifications required by regulatory bodies for pharmaceutical intermediates. The patent data highlights that by maintaining the reaction pH between 6.8 and 7.2 during the quenching phase with 10% sulfuric acid, the process ensures the complete neutralization of basic residues without degrading the sensitive ester functionality. The subsequent workup involves reduced pressure distillation to recover the DME solvent and excess p-tolualdehyde, which can be recycled, thereby enhancing the overall atom economy of the process. The final recrystallization from absolute ethanol serves as a polishing step that removes trace organic impurities, consistently delivering product with purity levels exceeding 99%. This level of control over the impurity profile reduces the burden on downstream processing and ensures that the material is fit for purpose in the synthesis of complex active pharmaceutical ingredients without requiring extensive additional purification.
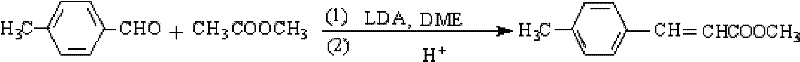
How to Synthesize Methyl 4-Methylcinnamate Efficiently
Implementing this synthesis route requires strict adherence to the specified molar ratios and temperature controls to replicate the high yields reported in the patent embodiments. The process begins with the preparation of the catalytic mixture in a dry environment to prevent the degradation of the LDA reagent, followed by the controlled addition of the aldehyde to manage the exotherm. Detailed operational parameters, including the specific molar ratios of DME to LDA to p-tolualdehyde ranging from 3.0:1.2:1.2, are critical for maximizing the conversion efficiency. Operators must ensure that the temperature does not exceed 5°C during the dropwise addition to maintain the selectivity of the enolate formation. The following guide outlines the standardized steps derived from the patent data to ensure consistent batch-to-batch quality and operational safety during scale-up.
- Mix glycol dimethyl ether solvent with lithium diisopropylamide catalyst and methyl acetate, then slowly add p-tolualdehyde below 5°C.
- Maintain reaction temperature between 0°C and 10°C for 3 to 6 hours to ensure complete condensation.
- Quench with 10% sulfuric acid to pH 6.8-7.2, separate organic layer, and recrystallize for high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this LDA-catalyzed route offers profound advantages for procurement managers and supply chain heads focused on cost reduction in pharmaceutical intermediates manufacturing. The primary economic driver is the substantial increase in reaction yield, which directly translates to a lower cost of goods sold per kilogram of finished product. By eliminating the need for excessive raw material ratios that characterize older methods, this process optimizes material utilization and reduces the volume of waste streams that require costly disposal. The ability to recover and recycle both the solvent and the excess aldehyde further enhances the economic viability of the process, creating a closed-loop system that minimizes raw material procurement costs. Additionally, the simplification of the workflow into a one-step reaction reduces the operational overhead associated with multi-stage synthesis, including labor costs, energy consumption, and equipment occupancy time. These factors combine to create a more resilient supply chain capable of responding to market demand fluctuations without compromising on margin or quality standards.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of readily available organic bases like LDA significantly lowers the input cost profile compared to noble metal-catalyzed alternatives. The high selectivity of the reaction means that less raw material is wasted on by-product formation, leading to substantial cost savings in material procurement. Furthermore, the mild reaction conditions reduce energy consumption for heating and cooling, contributing to a lower overall carbon footprint and operational expenditure. The simplified purification process also reduces the consumption of solvents and adsorbents typically required for chromatography or extensive washing, driving down variable costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as p-tolualdehyde and methyl acetate ensures that the supply chain is not vulnerable to the bottlenecks often associated with specialized or scarce reagents. The robustness of the reaction conditions allows for flexible manufacturing schedules, as the process is less sensitive to minor variations in ambient conditions compared to highly sensitive catalytic systems. This stability ensures consistent lead times for high-purity pharmaceutical intermediates, allowing downstream customers to plan their production schedules with greater confidence. The ability to scale from laboratory to commercial production without significant re-engineering of the process further secures the supply continuity for long-term contracts.
- Scalability and Environmental Compliance: The green chemistry principles embedded in this method, such as solvent recovery and reduced waste generation, align with increasingly strict environmental regulations governing chemical manufacturing. The absence of heavy metal residues simplifies the regulatory approval process for the final API, removing a significant barrier to market entry. The process is inherently scalable, as the exotherm is manageable and the workup procedures utilize standard unit operations like distillation and crystallization that are common in existing manufacturing facilities. This ease of scale-up reduces the capital expenditure required for new production lines, enabling faster time-to-market for new drug candidates relying on this intermediate.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis method, derived directly from the patent specifications and comparative data. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios. The answers provided reflect the specific advantages observed in the experimental embodiments, focusing on yield, purity, and operational parameters.
Q: Why is LDA preferred over NaOH for this condensation reaction?
A: LDA is a strong, non-nucleophilic base that effectively generates the lithium enolate of methyl acetate without promoting side reactions like self-condensation, which are common with aqueous NaOH systems.
Q: What is the expected purity of the final methyl 4-methylcinnamate product?
A: Using the described recrystallization process with absolute ethanol, the patent data indicates purity levels reaching up to 99.7%, suitable for sensitive pharmaceutical applications.
Q: How does this method improve supply chain reliability?
A: The one-step reaction process eliminates complex multi-stage synthesis and difficult purification steps associated with older methods, significantly reducing production lead time and operational complexity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Methyl 4-Methylcinnamate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the global pharmaceutical supply chain, and we are committed to delivering solutions that meet the rigorous demands of modern drug development. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We leverage advanced process chemistry expertise to optimize routes like the LDA-catalyzed condensation described herein, guaranteeing stringent purity specifications and rigorous QC labs testing for every batch. Our commitment to quality assurance means that every shipment of methyl 4-methylcinnamate is accompanied by comprehensive documentation, providing our partners with the confidence they need to advance their clinical and commercial programs without delay.
We invite procurement leaders and R&D directors to engage with us for a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality standards. By partnering with our technical procurement team, you can access specific COA data and route feasibility assessments that demonstrate the tangible benefits of switching to our optimized manufacturing processes. Whether you require pilot-scale quantities for clinical trials or multi-ton volumes for commercial launch, our flexible production capabilities and dedication to customer success make us the ideal partner for your supply chain needs. Contact us today to discuss how we can support your project with reliable, high-purity chemical solutions.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
