Industrial Scale-Up of 3-Ethoxycarbonyl-5-Pyridine Pinacol Borate via Optimized Suzuki Coupling
Industrial Scale-Up of 3-Ethoxycarbonyl-5-Pyridine Pinacol Borate via Optimized Suzuki Coupling
The pharmaceutical industry constantly demands robust, scalable synthetic routes for key heterocyclic building blocks, and the preparation of 3-ethoxycarbonyl-5-pyridine pinacol borate stands as a critical example where process optimization directly impacts supply chain stability. Patent CN103012451A introduces a transformative industrialized preparation method that effectively resolves longstanding technical bottlenecks associated with complicated post-treatment processes and dangerously low re-crystallization yields found in legacy methodologies. By leveraging a refined Suzuki-Miyaura cross-coupling reaction coupled with a novel solvent exchange crystallization technique, this technology enables the production of high-purity intermediates essential for modern drug discovery pipelines. For R&D directors and procurement specialists seeking a reliable pharma intermediate supplier, understanding the nuances of this patented approach reveals significant opportunities for cost reduction in pharmaceutical manufacturing and enhanced operational safety. The transition from laboratory-scale curiosity to tonnage-level production requires not just chemical efficacy but also economic viability, which this patent addresses by eliminating high-risk solvents like diethyl ether and streamlining the isolation of the final boronate ester.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
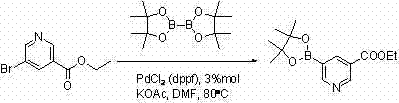
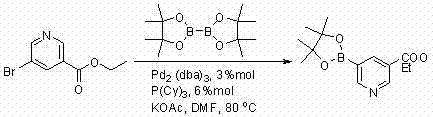
Prior to the innovations detailed in the provided intellectual property, the synthesis of this specific pyridine boronate was plagued by inefficiencies that rendered large-scale production economically unfeasible and operationally hazardous. Existing literature describes methods utilizing PdCl2(dppf) as a catalyst, which unfortunately suffer from abysmally low reaction conversion ratios hovering around 45%, resulting in a crude product mixture dominated by impurities rather than the desired target molecule. Furthermore, alternative approaches employing Pd2(dba)3 and P(Cy)3, while achieving better conversion rates near 83%, were severely hampered by archaic post-treatment protocols that relied on diethyl ether for extraction. The use of diethyl ether presents substantial safety risks in an industrial setting due to its high volatility and flammability, creating unacceptable hazards for plant operators and requiring expensive explosion-proof infrastructure. Additionally, the traditional purification involved double recrystallization using normal hexane and acetone, a tedious process that tragically capped the final isolated yield at a mere 30%, wasting over two-thirds of the valuable starting materials and driving up the cost of goods sold significantly.
The Novel Approach
The breakthrough methodology presented in the patent data fundamentally re-engineers the downstream processing to maximize material throughput while minimizing safety liabilities. Instead of relying on dangerous diethyl ether, the novel process utilizes methyl tert-butyl ether (MTBE) for the extraction phase, which offers a much higher flash point and improved operational safety profile for commercial scale-up of complex heterocyclic intermediates. The innovation extends beyond solvent selection to the crystallization strategy, where the organic phase is concentrated and subjected to a solvent swap from MTBE to n-heptane, inducing precipitation under controlled thermal conditions between 0°C and 10°C. This specific solvent system facilitates the formation of high-quality crystals that trap fewer impurities, allowing the process to achieve isolated yields exceeding 60%, effectively doubling the output compared to previous art. By simplifying the workup to a single filtration through diatomite followed by a streamlined washing and crystallization sequence, the new approach drastically reduces processing time and solvent consumption, making it an ideal candidate for high-purity pyridine boronate esters required in sensitive medicinal chemistry applications.
Mechanistic Insights into Pd2(dba)3-Catalyzed Suzuki Coupling
The core of this synthetic transformation relies on the palladium-catalyzed cross-coupling between ethyl 5-bromonicotinate and bis(pinacolato)diboron, a reaction that proceeds through a well-defined catalytic cycle involving oxidative addition, transmetallation, and reductive elimination steps. The selection of Pd2(dba)3 combined with tricyclohexylphosphine (P(Cy)3) as the ligand system is critical, as this electron-rich phosphine environment facilitates the oxidative addition of the aryl bromide bond to the palladium center more effectively than the dppf ligand used in inferior methods. The steric bulk of the tricyclohexylphosphine ligand helps stabilize the active palladium species against aggregation into inactive palladium black, ensuring that the catalyst remains active throughout the extended reaction period of 6 to 10 hours at elevated temperatures of 80°C to 85°C. Potassium acetate serves as the base in this system, activating the diboron reagent to form a nucleophilic boronate species capable of transmetallating with the palladium-aryl intermediate, ultimately releasing the desired 3-ethoxycarbonyl-5-pyridine pinacol borate product upon reductive elimination. Understanding this mechanistic pathway allows process chemists to fine-tune reaction parameters such as temperature and stoichiometry to minimize side reactions like protodeboronation or homocoupling, which are common pitfalls in pyridine functionalization.
Impurity control is another paramount aspect of this mechanism, particularly given the sensitivity of boronate esters to hydrolysis and the potential for residual palladium contamination in the final API intermediate. The patent specifies a rigorous nitrogen sparging protocol before and during the addition of reagents to maintain an inert atmosphere, preventing oxidation of the phosphine ligand and degradation of the sensitive boron species. Following the reaction, the immediate filtration through diatomite acts as a primary scavenging step, physically removing bulk palladium particulates and insoluble salts before the liquid-liquid extraction begins. The subsequent washing steps with saturated brine and water are designed to strip away polar impurities, including residual DMF solvent and inorganic salts, without exposing the product to acidic or basic conditions that might cleave the pinacol boronate group. Finally, the controlled crystallization from n-heptane ensures that any remaining organic impurities remain in the mother liquor, yielding a solid product with HPLC purity greater than 99%, which meets the stringent specifications required for downstream pharmaceutical synthesis.
How to Synthesize 3-Ethoxycarbonyl-5-Pyridine Pinacol Borate Efficiently
Executing this synthesis on a commercial scale requires strict adherence to the optimized parameters defined in the patent embodiments to ensure consistent quality and yield. The process begins with the preparation of the catalyst mixture in dimethylformamide (DMF), where precise molar ratios of palladium source, ligand, and base are maintained to drive the reaction to completion. Operators must carefully control the addition rate of the bromo-nicotinate solution to manage the exotherm and maintain the reaction temperature within the narrow window of 75°C to 85°C, as deviations can lead to incomplete conversion or catalyst decomposition. Once the reaction is deemed complete via sampling, the workup procedure shifts focus to efficient phase separation and solvent management, utilizing MTBE for its favorable partition coefficients. The final crystallization step is temperature-sensitive, requiring slow cooling to 0°C to 10°C to maximize crystal growth and recovery, a detail that is often overlooked in rushed laboratory preparations but is vital for industrial success. For a comprehensive breakdown of the exact masses, volumes, and timing for each unit operation, please refer to the standardized synthesis guide below.
- Charge Pd2(dba)3, P(Cy)3, KOAc, and bis(pinacolato)diboron in DMF, degas with nitrogen, and heat to 75-80°C.
- Add a solution of ethyl 5-bromonicotinate in DMF dropwise while maintaining temperature, then react at 80-85°C for 6-10 hours.
- Filter through diatomite, extract with methyl tert-butyl ether (MTBE), wash with brine, concentrate, swap solvent to n-heptane, and crystallize at 0-10°C.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates into tangible strategic benefits that extend far beyond simple chemical yield improvements. The elimination of diethyl ether from the workflow removes a significant safety hazard, thereby reducing insurance premiums and the need for specialized ATEX-rated equipment, which represents a substantial capital expenditure saving for manufacturing facilities. Furthermore, the dramatic increase in isolated yield means that less raw material is required to produce the same amount of finished goods, directly lowering the variable cost per kilogram and improving the overall margin structure for the final drug product. The robustness of the process, demonstrated by successful scaling from gram quantities to multi-kilogram batches in the patent examples, assures supply chain continuity by minimizing the risk of batch failures or off-spec material that could disrupt production schedules. Additionally, the use of common, commercially available solvents like DMF, MTBE, and n-heptane ensures that sourcing remains stable and unaffected by niche chemical market fluctuations, providing a reliable foundation for long-term supply agreements.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substantial improvement in material efficiency, as the yield has been effectively doubled compared to legacy methods without increasing the complexity of the reaction setup. By avoiding the loss of product during multiple recrystallization steps and utilizing a more efficient extraction solvent, the process minimizes waste disposal costs and maximizes the utility of expensive palladium catalysts and boron reagents. This efficiency gain allows manufacturers to offer more competitive pricing structures while maintaining healthy margins, a critical factor in the highly price-sensitive generic pharmaceutical market. Moreover, the simplified workup reduces the total man-hours required per batch, lowering labor costs and increasing the throughput capacity of existing reactor trains without the need for new hardware investments.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals and standard unit operations that can be easily replicated across different manufacturing sites globally. The process does not rely on exotic reagents or cryogenic conditions that might be difficult to source or maintain in certain geographic regions, ensuring that production can continue uninterrupted even during regional supply disruptions. The high purity of the final product reduces the burden on downstream customers who would otherwise need to perform additional purification steps, thereby shortening their own lead times and accelerating their time-to-market for new therapies. This reliability makes the manufacturer a preferred partner for multinational corporations seeking to de-risk their supply chains against regulatory audits and quality failures.
- Scalability and Environmental Compliance: From an environmental perspective, the switch to MTBE and n-heptane aligns better with modern green chemistry principles compared to the use of diethyl ether and large volumes of acetone, facilitating easier compliance with increasingly strict environmental regulations. The process generates less hazardous waste per kilogram of product, simplifying the permitting process for new manufacturing lines and reducing the environmental footprint of the facility. Scalability is inherently built into the design, as evidenced by the patent examples which successfully demonstrate the reaction on scales ranging from 100 grams to over 25 kilograms with consistent results. This proven scalability gives confidence to investors and stakeholders that the technology can meet the demands of clinical trial material production as well as full commercial launch volumes without requiring fundamental process re-engineering.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 3-ethoxycarbonyl-5-pyridine pinacol borate, derived directly from the technical specifications and experimental data provided in the patent documentation. These insights are intended to clarify the operational advantages and quality standards associated with this specific manufacturing route for potential partners and technical evaluators. Understanding these details is crucial for assessing the fit of this intermediate within your specific synthetic pathways and supply chain requirements.
Q: What is the primary advantage of the new workup procedure described in CN103012451A?
A: The new procedure replaces hazardous diethyl ether extraction and complex double recrystallization with a safer MTBE extraction and n-heptane solvent swap, significantly improving isolation yield from approximately 30% to over 60%.
Q: Why is the Pd2(dba)3/P(Cy)3 catalyst system preferred over PdCl2(dppf) for this substrate?
A: Literature data indicates that the PdCl2(dppf) system results in low conversion rates around 45%, whereas the Pd2(dba)3/P(Cy)3 system achieves high conversion rates exceeding 83%, making it essential for efficient production.
Q: How is high purity (>99%) achieved in the final product?
A: High purity is secured through a rigorous purification sequence involving diatomite filtration to remove palladium residues, followed by controlled crystallization using a methyl tert-butyl ether and n-heptane mixture at low temperatures between 0°C and 10°C.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Ethoxycarbonyl-5-Pyridine Pinacol Borate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires a partner with deep technical expertise and a commitment to quality excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the optimized Suzuki coupling process described above can be implemented seamlessly within our state-of-the-art facilities. We adhere to stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to guarantee that every batch of 3-ethoxycarbonyl-5-pyridine pinacol borate meets or exceeds the >99% HPLC purity benchmark established in the patent. Our dedication to process safety and environmental stewardship means that we have already integrated the safer solvent systems and waste reduction strategies outlined in this technology, ready to deliver sustainable value to our global clientele.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can support your specific project needs and timeline. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to our optimized supply source for your next campaign. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements, ensuring that your supply chain is built on a foundation of scientific rigor and commercial reliability.
