Scalable Production of Enantiopure Indole Carboxylic Acids via Diastereoselective Alkylation
Scalable Production of Enantiopure Indole Carboxylic Acids via Diastereoselective Alkylation
The pharmaceutical industry constantly seeks robust methodologies for generating high-purity chiral intermediates, particularly for cardiovascular therapies. Patent CN1067394C introduces a groundbreaking approach for the preparation of enantiomerically pure cycloalkanoindole, azaindole, and pyrimido[1,2-a]indole carboxylic acids. These compounds serve as critical precursors for potent anti-atherosclerotic agents. Unlike traditional resolution techniques that discard half of the produced material, this invention leverages diastereoselective alkylation of chiral esters to achieve superior yields and purity. The core innovation lies in the strategic use of chiral auxiliaries, such as menthyl or bornyl groups, which direct the stereochemistry during the carbon-carbon bond formation at the alpha-position. 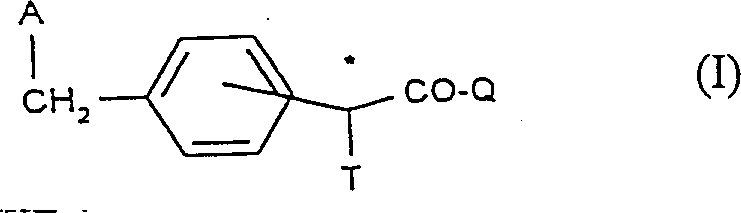 This structural versatility allows for the synthesis of a broad range of derivatives while maintaining strict control over the stereocenter, addressing a major bottleneck in the commercial manufacturing of complex API intermediates.
This structural versatility allows for the synthesis of a broad range of derivatives while maintaining strict control over the stereocenter, addressing a major bottleneck in the commercial manufacturing of complex API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of enantiomerically pure carboxylic acids has relied heavily on the separation of racemic mixtures through diastereomeric salt formation or chiral chromatography. These conventional pathways suffer from inherent economic and operational inefficiencies that severely impact the bottom line for large-scale production. Specifically, the maximum theoretical yield for resolving a racemate is capped at 50%, meaning half of the synthesized material is essentially waste unless complex recycling loops are implemented. Furthermore, chromatographic separation requires specialized, high-cost equipment and large volumes of solvents, driving up both capital expenditure and environmental disposal costs. The removal of the 'wrong' enantiomer often involves multiple crystallization steps that degrade overall throughput. For procurement managers, this translates to higher raw material costs and longer lead times, as the process is fundamentally limited by the physics of separation rather than the efficiency of synthesis.
The Novel Approach
The methodology described in CN1067394C circumvents these limitations by establishing chirality early in the synthesis through the use of optically pure starting materials. Instead of separating enantiomers post-synthesis, the process utilizes a chiral alcohol to esterify tolueneacetic acid, creating a chiral environment that dictates the outcome of subsequent reactions. The key breakthrough is the diastereoselective alkylation at the alpha-carbon, which proceeds with high specificity to favor one diastereomer significantly over the other. This approach not only bypasses the 50% yield ceiling but also simplifies purification, as the desired diastereomer can often be crystallized directly from the reaction mixture. By shifting the paradigm from separation to selective synthesis, this route offers a streamlined pathway that reduces solvent consumption, minimizes waste generation, and significantly enhances the overall mass balance of the manufacturing process.
Mechanistic Insights into Diastereoselective Alkylation and Coupling
The chemical elegance of this process lies in the precise control of stereochemistry during the alkylation and subsequent functionalization steps. The reaction begins with the formation of a chiral ester, typically using (-)-menthol or similar chiral alcohols, which acts as a stereochemical director. When treated with a strong base such as potassium tert-butoxide in a polar aprotic solvent like DMF, the alpha-proton is abstracted to form an enolate. The bulky chiral auxiliary shields one face of the enolate, forcing the incoming cycloalkyl halide to attack from the less hindered side. This steric differentiation results in the formation of the desired diastereomer in high excess. Crucially, the subsequent halogenation of the aromatic methyl group is performed under radical conditions that do not disturb the acidic alpha-proton, thereby preventing racemization. 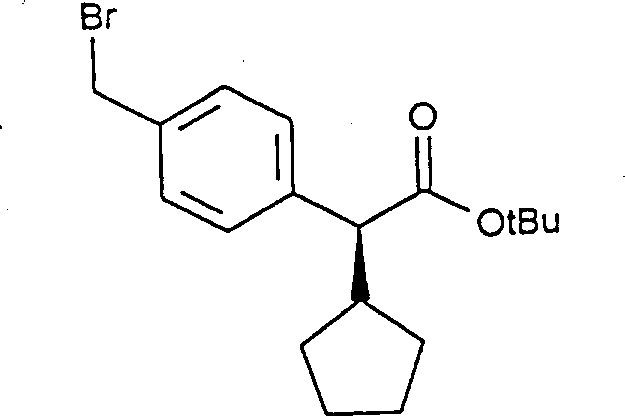 This stability is vital, as it ensures that the optical purity established in the first step is retained throughout the synthesis. The final coupling with the indole nucleus proceeds via nucleophilic substitution, again without compromising the chiral center, resulting in a highly enantiopure intermediate ready for final deprotection.
This stability is vital, as it ensures that the optical purity established in the first step is retained throughout the synthesis. The final coupling with the indole nucleus proceeds via nucleophilic substitution, again without compromising the chiral center, resulting in a highly enantiopure intermediate ready for final deprotection.
Impurity control is intrinsically built into this mechanism through the physical properties of the diastereomers. Because the desired product and the minor diastereomer have distinct physical properties, particularly solubility, they can be separated by simple crystallization. The patent highlights that the desired diastereomer crystallizes directly from the reaction mixture, allowing for the isolation of high-purity material even from crude products. Furthermore, the mother liquor containing the unwanted diastereomer is not discarded; it can be subjected to epimerization conditions using specific bases and solvents to convert the minor isomer back into the major one. This dynamic kinetic resolution capability effectively pushes the yield towards 100% theoretically, drastically reducing the impurity load and ensuring that the final API intermediate meets stringent regulatory specifications for chiral purity without the need for preparative HPLC.
How to Synthesize Enantiopure Indole Carboxylic Acids Efficiently
The synthesis of these high-value intermediates follows a logical sequence designed for industrial robustness. It begins with the esterification of the achiral acid with a chiral alcohol, followed by the critical diastereoselective alkylation which sets the stereocenter. The process continues with benzylic halogenation and nucleophilic coupling with the heterocyclic core, concluding with the removal of the chiral auxiliary to reveal the free acid. This sequence avoids the pitfalls of late-stage resolution and maximizes material throughput. For detailed operational parameters, including specific temperatures, stoichiometry, and workup procedures, please refer to the standardized guide below.
- Esterify tolueneacetic acid with a chiral alcohol such as menthol to form a chiral ester intermediate.
- Perform diastereoselective alkylation at the alpha-carbon using a base like potassium tert-butoxide and a cycloalkyl halide.
- Halogenate the toluene methyl group and couple with the indole nucleus, followed by hydrolysis to remove the chiral auxiliary.
Commercial Advantages for Procurement and Supply Chain Teams
For supply chain leaders and procurement specialists, the adoption of this synthetic route offers transformative advantages in terms of cost structure and supply reliability. Traditional chiral resolution methods are resource-intensive, requiring significant amounts of solvents and energy for separation processes that inherently waste half the input material. By contrast, this diastereoselective approach optimizes atom economy, meaning less raw material is required to produce the same amount of active ingredient. The ability to recycle mother liquors through epimerization further enhances this efficiency, turning what would be waste into valuable product. This reduction in material intensity directly correlates to lower variable costs per kilogram, providing a competitive edge in pricing negotiations. Additionally, the reliance on standard chemical reagents like N-bromosuccinimide and common bases ensures that the supply chain is not vulnerable to shortages of exotic catalysts or specialized chiral columns.
- Cost Reduction in Manufacturing: The elimination of chromatographic purification steps represents a massive reduction in operational expenditure. Chromatography is not only slow but also expensive due to the cost of stationary phases and the energy required for solvent recovery. By achieving high purity through crystallization, this process removes the need for such costly unit operations. Furthermore, the high yield obtained from the diastereoselective alkylation means that the effective cost of the chiral auxiliary is amortized over a much larger quantity of product. The process avoids the 50% yield penalty associated with racemic resolution, effectively doubling the output capacity of existing reactor infrastructure without additional capital investment. This efficiency gain allows for substantial cost savings that can be passed down the supply chain or reinvested in R&D.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by complex processes that have low tolerance for variation. This method utilizes robust reaction conditions that are less sensitive to minor fluctuations in temperature or mixing, making it ideal for multi-ton scale production. The starting materials, such as tolueneacetic acid and chiral terpene alcohols, are commodity chemicals with stable global supply networks, reducing the risk of raw material bottlenecks. Moreover, the crystallization-driven purification provides a consistent quality output that is less prone to the batch-to-batch variability often seen in chromatographic separations. This predictability allows for more accurate production planning and shorter lead times for delivering high-purity pharmaceutical intermediates to downstream customers.
- Scalability and Environmental Compliance: Scaling chiral processes is notoriously difficult, but this route is designed with scalability in mind. The exothermic nature of the alkylation and halogenation steps can be managed effectively in large reactors using standard cooling systems. From an environmental perspective, the reduction in solvent usage and the elimination of silica gel waste from chromatography significantly lower the E-factor (environmental factor) of the process. The ability to recycle mother liquors minimizes liquid waste discharge, aligning with increasingly strict environmental regulations in chemical manufacturing hubs. This green chemistry profile not only reduces disposal costs but also enhances the sustainability credentials of the final pharmaceutical product, a growing priority for global healthcare companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. Understanding these details is crucial for evaluating the feasibility of integrating this route into your existing manufacturing portfolio. The answers are derived directly from the experimental data and claims within the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: How does this method improve upon conventional chiral separation?
A: Conventional methods rely on chromatographic separation or fractional crystallization of racemates, which inherently limits yield to 50% and requires expensive equipment. This patented process utilizes diastereoselective alkylation of a chiral ester, allowing for yields significantly higher than 50% and enabling the recycling of mother liquors through epimerization.
Q: What ensures the optical purity during the halogenation step?
A: The process is designed such that the halogenation of the toluene methyl group occurs under conditions that do not induce racemization at the chiral alpha-carbon of the carboxylic acid function. This preserves the enantiomeric excess established during the initial alkylation step.
Q: Can the unwanted diastereomers be utilized?
A: Yes, a key advantage of this technology is the ability to epimerize the undesired diastereomer back into the desired configuration using appropriate solvents and bases. This allows the mother liquor to be recycled directly into the alkylation step, optimizing atom economy and reducing waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Enantiopure Indole Carboxylic Acids Supplier
The technical potential of CN1067394C is immense, offering a clear path to high-quality, cost-effective cardiovascular drug intermediates. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production required to bring this chemistry to life. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, including enantiomeric excess and diastereomeric ratios, ensuring that every batch meets the exacting standards of the global pharmaceutical industry. We understand that transitioning a novel synthetic route from the lab to the plant requires expertise in process safety, thermal hazard analysis, and crystallization engineering, all of which are core competencies of our technical team.
We invite you to discuss how this advanced synthesis can optimize your supply chain and reduce your overall cost of goods. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data from pilot batches and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals for anti-atherosclerotic drug development.
