Advanced Synthesis Of Quinoline Derivatives For Commercial Scale-Up And High Purity Pharmaceutical Intermediates
Advanced Synthesis Of Quinoline Derivatives For Commercial Scale-Up And High-Purity Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for complex heterocyclic compounds, particularly those serving as critical intermediates for respiratory and neurological therapeutics. Patent CN1125815C discloses a groundbreaking methodology for the preparation of pharmaceutically active quinoline compounds, specifically targeting the synthesis of (-)-(S)-N-(α-ethylbenzyl)-3-hydroxy-2-phenylquinoline-4-carboxamide and its analogs. These compounds function as potent NK-3 receptor antagonists, demonstrating significant therapeutic potential for treating chronic obstructive pulmonary disease (COPD), asthma, and various central nervous system disorders. The innovation lies not merely in the final molecule but in the strategic redesign of the synthetic pathway to overcome historical bottlenecks associated with low yields and cumbersome purification processes. By leveraging specific reaction conditions and novel intermediate conversions, this technology offers a viable path toward high-purity pharmaceutical intermediates suitable for large-scale commercial production.
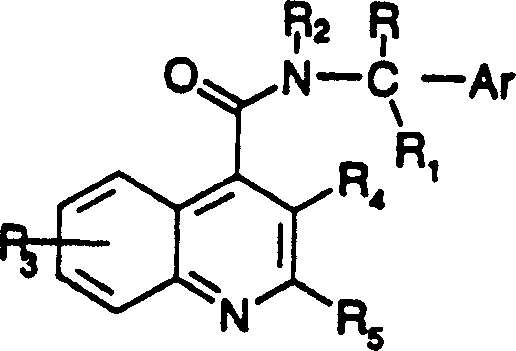
Historically, the synthesis of quinoline-based NK-3 receptor antagonists relied heavily on methodologies disclosed in earlier patents such as WO95/32948, which presented substantial limitations for industrial application. The conventional approach typically employed dicyclohexylcarbodiimide (DCC) mediated coupling reactions to link the quinoline carboxylic acid moiety with the requisite chiral amine. While chemically feasible on a small laboratory scale, this legacy method suffered from severe inefficiencies when translated to manufacturing environments. The DCC coupling often resulted in isolated yields ranging merely from 30% to 50%, primarily due to the formation of stubborn by-products that necessitated rigorous chromatographic purification. Furthermore, the reliance on expensive starting materials like α-methoxyacetophenone increased the raw material costs significantly, while the generation of dicyclohexylurea waste created environmental and disposal challenges. These factors combined to make the conventional route economically unviable for the cost-sensitive generic and bulk pharmaceutical markets, creating a clear demand for a more efficient alternative.
In stark contrast, the novel approach detailed in CN1125815C introduces a paradigm shift by optimizing both the reagent selection and the reaction mechanism to drastically enhance efficiency. The new method replaces the problematic DCC coupling with a sophisticated activation strategy using thionyl chloride in the presence of a tertiary amine base, specifically triethylamine, under strictly anhydrous conditions. This modification alone mitigates the formation of difficult-to-remove urea by-products. More critically, the process incorporates a unique thermal conversion step where reaction intermediates and specific by-products, previously considered waste, are converted into the desired final product upon heating to 65-70°C. This mechanistic advantage allows the overall yield of the desired quinoline carboxamide to increase from the historical 30-50% range to greater than 70%. By eliminating the need for chromatographic purification and utilizing more accessible starting materials like isatin and α-acetoxyacetophenone, this approach delivers substantial cost reduction in API manufacturing while simultaneously improving the environmental profile of the synthesis.
Mechanistic Insights Into Thionyl Chloride Activated Amidation And By-Product Conversion
To fully appreciate the technical superiority of this synthesis, one must examine the intricate mechanistic pathways that govern the transformation of the alpha-hydroxy acid intermediate into the final amide. The process begins with the condensation of isatin and an alpha-acetoxyketone in an aqueous lithium hydroxide solution, where the base concentration is meticulously controlled between 3 to 5 equivalents to ensure complete ring opening and coupling without degrading the sensitive quinoline nucleus. Following the isolation of the resulting 3-hydroxy-2-phenylquinoline-4-carboxylic acid, the critical activation phase commences. Unlike traditional methods that might struggle with the phenolic hydroxyl group at the 3-position, this protocol utilizes thionyl chloride to activate the carboxyl group while simultaneously managing the reactivity of the phenol. The reaction mixture is cooled to below 2°C before the addition of the activator to control exotherms and prevent side reactions, ensuring the formation of a reactive acyl chloride species that is primed for nucleophilic attack by the chiral amine.
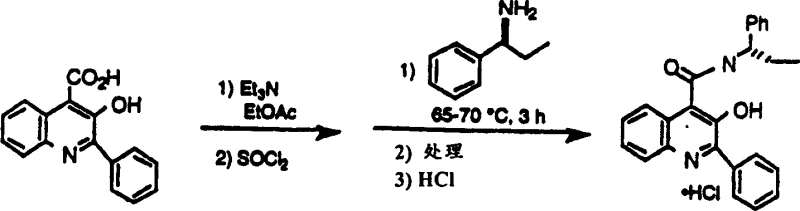
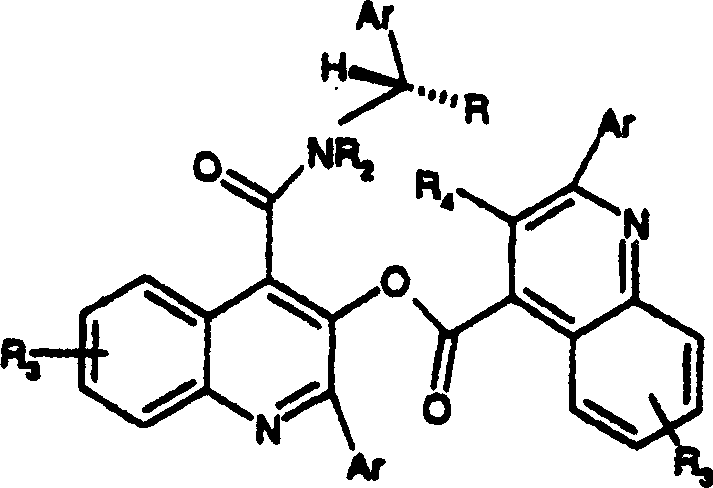
A distinguishing feature of this technology is its ability to manage and utilize reaction by-products that would typically reduce overall yield in other processes. During the coupling of the activated acid with the amine, such as (S)-1-phenylpropylamine, a specific ester by-product known as Formula (VI) is initially formed alongside the desired amide. In conventional syntheses, this ester would represent a loss of material requiring separation. However, mechanistic studies within the patent reveal that upon heating the reaction mixture to 65-70°C, this Formula (VI) intermediate undergoes a rearrangement or further reaction with excess amine to convert directly into the target quinoline carboxamide. This phenomenon effectively recycles what would be waste into product, thereby boosting the mass balance and purity of the crude output. This intrinsic self-correction mechanism reduces the burden on downstream purification units and ensures that the final crude product meets stringent purity specifications with minimal additional processing, a critical factor for regulatory compliance in pharmaceutical manufacturing.
How To Synthesize Quinoline Carboxamide Efficiently
Implementing this synthesis requires precise adherence to the specified reaction parameters to maximize the benefits of the novel mechanistic pathway. The process is designed to be scalable, moving seamlessly from kilogram-level development to multi-ton commercial production without losing yield or purity. Operators must focus on maintaining strict temperature controls during the activation phase and ensuring anhydrous conditions to prevent hydrolysis of the reactive intermediates. The detailed standardized synthesis steps involve specific molar ratios of lithium hydroxide, thionyl chloride, and triethylamine, along with defined heating and cooling cycles that are critical for the conversion of the Formula (VI) by-product. For technical teams looking to replicate or license this technology, the following guide outlines the critical operational phases derived directly from the patent examples, ensuring that the theoretical yield advantages are realized in practical application.
- Condense isatin with alpha-acetoxyketone in aqueous lithium hydroxide at 50-85°C to form the alpha-hydroxy acid intermediate.
- Activate the isolated acid using thionyl chloride and triethylamine in ethyl acetate under anhydrous conditions below 2°C.
- Couple with chiral amine and heat to 65-70°C to convert intermediates and by-products into the final quinoline carboxamide.
Commercial Advantages For Procurement And Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this synthetic route offers transformative benefits that extend beyond simple chemistry metrics. The elimination of chromatographic purification represents a significant reduction in manufacturing overhead, as it removes the need for large volumes of organic solvents, silica gel, and the associated labor and time costs of column operations. This simplification of the downstream processing workflow leads to substantially reduced production cycles, allowing for faster turnaround times on customer orders and improved responsiveness to market demand fluctuations. Furthermore, the reliance on commercially available starting materials like isatin and alpha-acetoxyacetophenone, which are produced by multiple global suppliers, mitigates the risk of raw material shortages. This diversification of the supply base ensures continuity of supply, a critical metric for supply chain heads managing the inventory of high-value pharmaceutical intermediates.
- Cost Reduction In Manufacturing: The economic impact of this process is driven by the drastic simplification of the purification train. By avoiding the use of expensive coupling reagents like DCC and eliminating chromatography, the direct material costs are significantly lowered. Additionally, the increase in yield from approximately 50% to over 70% means that less raw material is required to produce the same amount of final product, effectively reducing the cost of goods sold. The ability to convert by-products into the main product further enhances atom economy, minimizing waste disposal costs and maximizing the value extracted from every kilogram of input material, resulting in substantial cost savings for the final API manufacturer.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of robust, commodity-grade chemicals rather than specialized, single-source reagents. The process tolerances are designed to accommodate standard industrial equipment, reducing the need for custom-built reactors or specialized containment systems required for highly toxic reagents like phosgene, which was used in some older methods. This compatibility with standard stainless steel reactors facilitates easier technology transfer between manufacturing sites. Consequently, procurement managers can secure long-term contracts with greater confidence, knowing that the production process is less susceptible to disruptions caused by reagent scarcity or complex equipment maintenance issues.
- Scalability And Environmental Compliance: The environmental footprint of this synthesis is markedly smaller than conventional alternatives, aligning with modern green chemistry principles and increasingly strict regulatory standards. The reduction in solvent usage and the elimination of solid waste from chromatography columns simplify waste stream management and lower the costs associated with environmental compliance. The process is inherently scalable, as demonstrated by the successful execution of the reaction in multi-liter reactors with consistent exotherm control. This scalability ensures that the method can support commercial scale-up of complex pharmaceutical intermediates from pilot batches to hundred-ton annual production capacities without requiring fundamental changes to the chemistry, providing a stable foundation for long-term business growth.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this quinoline synthesis technology. These answers are derived from the specific experimental data and claims found within the patent documentation, providing clarity on the operational requirements and expected outcomes. Understanding these details is essential for R&D directors evaluating the feasibility of integrating this route into existing production lines and for quality assurance teams establishing control strategies for the new process.
Q: How does this method improve yield compared to conventional DCC-mediated coupling?
A: Conventional methods using DCC often result in yields between 30-50% and require chromatographic purification to remove urea by-products. This novel process achieves yields greater than 70% by utilizing a specific activation sequence that allows side-products to convert into the desired product upon heating, eliminating the need for chromatography.
Q: What are the key starting materials for this quinoline synthesis?
A: The process utilizes commercially available and cost-effective starting materials, specifically isatin (or substituted isatins) and alpha-acetoxyketones. These are reacted in the presence of a base such as lithium hydroxide, which is preferred over sodium hydroxide for improved reaction kinetics and impurity profiles.
Q: Is chromatographic purification required for the final product?
A: No, one of the primary advantages of this invention is the elimination of chromatographic purification steps. The reaction conditions are optimized to convert potential by-products, such as formula (VI) intermediates, directly into the final active compound during the heating phase, significantly reducing manufacturing costs and waste.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable Quinoline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient, high-yield synthetic routes in the competitive landscape of pharmaceutical intermediate manufacturing. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patented technologies like CN1125815C are fully realized in our facilities. We are committed to delivering high-purity quinoline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with advanced analytical instrumentation. Our capability to handle complex chemistries, including moisture-sensitive activations and precise thermal conversions, positions us as a strategic partner for companies seeking to optimize their supply chain for respiratory and CNS therapeutics.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis method can be tailored to your specific project requirements. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this more efficient route. We encourage you to request specific COA data and route feasibility assessments to verify our capability to deliver consistent quality at scale. Let us help you secure a reliable supply of high-quality intermediates while driving down your overall manufacturing costs through superior process chemistry.
