Advanced Manufacturing of (2-Nitrophenyl)acetonitrile Derivatives for High-Volume Agrochemical Production
Introduction to Advanced Nitro-Aromatic Synthesis
The global demand for high-performance agrochemicals continues to drive innovation in the synthesis of critical intermediates, particularly within the nitro-aromatic sector. Patent CN1578760A introduces a transformative methodology for the manufacturing of (2-nitrophenyl)acetonitrile derivatives, which serve as pivotal building blocks for next-generation herbicides and fungicides. This intellectual property outlines a robust three-step sequence that circumvents the traditional limitations associated with direct benzylation, offering a pathway that is both chemically elegant and commercially viable for large-scale operations. By shifting the synthetic strategy from hazardous halide displacement to a controlled formylation and dehydration protocol, this technology addresses long-standing challenges in impurity profiles and process safety. For procurement leaders and technical directors alike, understanding the nuances of this patent is essential for securing a reliable agrochemical intermediate supplier capable of delivering consistent quality in a volatile market.
The significance of this invention lies in its ability to produce complex nitrile structures with exceptional purity, a parameter that directly influences the efficacy of the final pesticide active ingredient. Traditional methods often struggle with side reactions that generate stubborn impurities, necessitating costly and time-consuming purification steps that erode profit margins. In contrast, the approach detailed in CN1578760A leverages readily available starting materials, such as substituted nitrotoluenes, and converts them through a series of high-yielding transformations. This strategic pivot not only enhances the chemical efficiency of the production line but also aligns with modern environmental standards by reducing the reliance on toxic alkylating agents. As we delve deeper into the technical specifics, it becomes clear that this process represents a substantial leap forward in cost reduction in agrochemical manufacturing, providing a competitive edge for companies that adopt these advanced synthetic routes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of (2-nitrophenyl)acetonitrile derivatives has relied heavily on the nucleophilic substitution of 2-nitrobenzyl halides with cyanide sources. While conceptually straightforward, this conventional pathway is plagued by significant practical drawbacks that hinder its efficiency in an industrial setting. The primary issue arises from the high reactivity of the benzyl halide intermediate, which tends to undergo secondary benzylation reactions with the newly formed nitrile product. This side reaction generates bis-benzylated by-products that are structurally similar to the target molecule, making their removal via standard crystallization or distillation extremely difficult and often resulting in substantial yield losses. Furthermore, the handling of cyanide salts in the presence of reactive alkyl halides poses severe safety risks, requiring specialized containment infrastructure and rigorous waste treatment protocols that drive up operational expenditures. These factors collectively contribute to a process that is not only economically inefficient but also environmentally burdensome, creating a pressing need for alternative synthetic strategies that can deliver high-purity agrochemical intermediates without the associated liabilities.
The Novel Approach
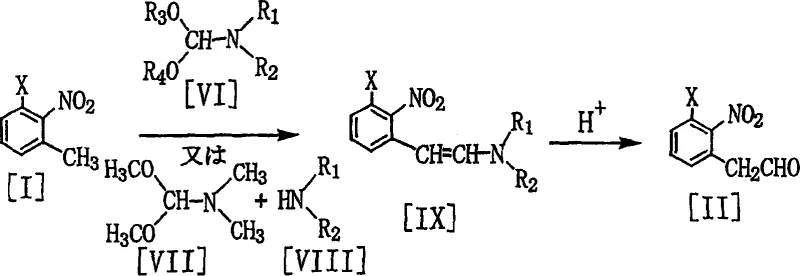
The methodology disclosed in the patent offers a sophisticated alternative that effectively bypasses the pitfalls of direct benzylation by employing a formylation-oximation-dehydration sequence. Instead of introducing the nitrile group via displacement, the process first installs an aldehyde functionality onto the nitro-toluene scaffold using N,N-dimethylformamide dimethyl acetal (DMF-DMA). This initial step generates an enamine intermediate which is subsequently hydrolyzed to the corresponding phenylacetaldehyde, setting the stage for a clean conversion to the nitrile. By avoiding the use of benzyl halides entirely, the new route eliminates the mechanism responsible for the troublesome bis-benzylated impurities, thereby streamlining the purification process and significantly boosting overall recovery rates. Moreover, the reaction conditions are milder and more controllable, allowing for better management of exotherms and improved safety profiles during scale-up. This innovative approach demonstrates how strategic reagent selection can fundamentally alter the economic landscape of chemical production, offering a clear path toward commercial scale-up of complex nitro compounds with enhanced reliability.
Mechanistic Insights into Formylation and Dehydration Chemistry
The core of this synthetic breakthrough lies in the mechanistic elegance of the formylation step, which utilizes the Vilsmeier-Haack-type reactivity of DMF acetals to functionalize the benzylic position of the nitrotoluene. In this transformation, the electron-deficient aromatic ring, activated by the ortho-nitro group, undergoes condensation with the formamide acetal to form a stable 2-(2-dialkylaminovinyl)nitrobenzene derivative. This enamine species serves as a protected equivalent of the aldehyde, preventing premature oxidation or polymerization while allowing for precise control over the reaction stoichiometry. Subsequent acidic hydrolysis cleaves the enamine bond, releasing the free phenylacetaldehyde derivative in high purity. The ability to perform this sequence without isolating the sensitive enamine intermediate is a key feature of the process, as it minimizes exposure to air and moisture, thereby reducing degradation and maximizing throughput. This level of mechanistic control is critical for R&D teams focused on optimizing impurity profiles, as it ensures that the aldehyde precursor entering the subsequent steps is of consistent quality, which is a prerequisite for achieving the stringent specifications required in pharmaceutical and agrochemical applications.
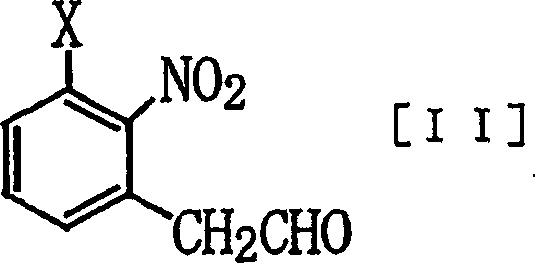
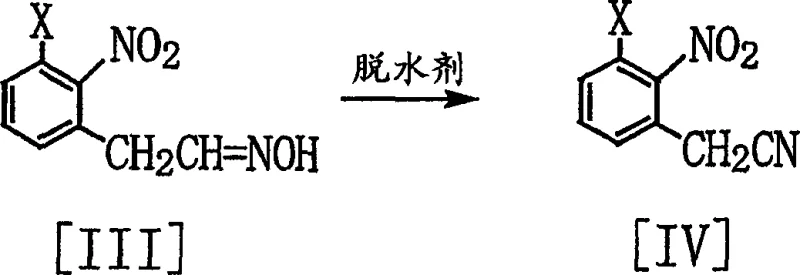
Following the formation of the aldehyde, the process proceeds through an oximation step where the carbonyl group is condensed with hydroxylamine to yield the corresponding oxime. This intermediate is then subjected to dehydration using agents such as acetic anhydride or thionyl chloride to furnish the final nitrile product. The dehydration mechanism involves the activation of the oxime hydroxyl group, facilitating the elimination of water and the formation of the carbon-nitrogen triple bond. The choice of dehydrating agent is flexible, with the patent highlighting options ranging from acid halides to carbodiimides, allowing manufacturers to select reagents based on cost, availability, and waste disposal considerations. Crucially, this dehydration step proceeds cleanly without affecting the sensitive nitro group or other substituents on the aromatic ring, demonstrating excellent chemoselectivity. For supply chain managers, this robustness translates to reducing lead time for high-purity agrochemical intermediates, as the process is less susceptible to batch-to-batch variability and requires fewer corrective interventions during production.
How to Synthesize (2-Nitrophenyl)acetonitrile Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control and reagent addition rates, to ensure optimal conversion and safety. The process begins with the heating of the nitrotoluene substrate with DMF-DMA, typically in a solvent such as toluene or xylene, to drive the formation of the enamine. Once the enamine is generated, acidic hydrolysis is performed, often in a biphasic system to facilitate the separation of the organic aldehyde product from aqueous by-products. The subsequent oximation is conducted under mild basic conditions to promote nucleophilic attack by the hydroxylamine, followed by the final dehydration step which may require elevated temperatures depending on the chosen activating agent. Detailed standard operating procedures for each of these stages, including specific molar ratios and workup protocols, are essential for replicating the high yields reported in the patent examples. For technical teams looking to adopt this technology, the following guide outlines the critical operational milestones necessary for successful execution.
- React 2-methylnitrobenzene derivative with N,N-dimethylformamide dimethyl acetal to form an enamine intermediate, followed by acidic hydrolysis to generate the phenylacetaldehyde derivative.
- Condense the resulting phenylacetaldehyde with hydroxylamine or its inorganic salts in the presence of a base to form the phenylacetaldoxime derivative.
- Dehydrate the phenylacetaldoxime using a dehydrating agent such as acetic anhydride or thionyl chloride at elevated temperatures to yield the final (2-nitrophenyl)acetonitrile.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers compelling advantages that extend far beyond simple chemical yield improvements. By eliminating the need for hazardous 2-nitrobenzyl bromides, manufacturers can significantly reduce the costs associated with raw material procurement, storage, and regulatory compliance. The removal of these dangerous intermediates simplifies the facility's safety infrastructure requirements, potentially lowering insurance premiums and reducing the capital expenditure needed for specialized containment systems. Furthermore, the avoidance of difficult-to-remove by-products means that downstream purification processes, such as chromatography or multiple recrystallizations, can be minimized or entirely eliminated. This streamlining of the production workflow results in substantial cost savings in terms of solvent consumption, energy usage, and labor hours, directly impacting the bottom line for any organization involved in cost reduction in agrochemical manufacturing. The cumulative effect of these efficiencies creates a more resilient and profitable supply chain capable of withstanding market fluctuations.
In terms of supply chain reliability, the new method utilizes starting materials that are commodity chemicals with stable global availability, reducing the risk of bottlenecks caused by the scarcity of specialized reagents. The ability to run the reaction steps continuously in the same vessel, as suggested by the patent, further enhances operational efficiency by reducing transfer losses and equipment cleaning times. This "one-pot" potential allows for faster batch turnover and increased plant capacity without the need for additional hardware investment. Additionally, the improved purity profile of the final product reduces the likelihood of customer rejections or返工 (rework), ensuring a smoother flow of goods to the end user. For supply chain heads, these factors translate into a more predictable delivery schedule and a stronger ability to meet just-in-time manufacturing demands, solidifying the position of the manufacturer as a reliable agrochemical intermediate supplier in a competitive global marketplace.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this process into their existing production portfolios. The clarity provided here aims to bridge the gap between theoretical chemistry and practical industrial application, ensuring that all parties have a shared understanding of the technology's capabilities and limitations.
Q: What are the primary advantages of this synthesis route over conventional benzylation methods?
A: This method avoids the use of hazardous 2-nitrobenzyl bromides, which often lead to difficult-to-remove benzylation by-products and lower yields. The new route utilizes safer formylation and dehydration steps, resulting in higher purity and easier refinement.
Q: Can this process be scaled for industrial production of agrochemical intermediates?
A: Yes, the patent explicitly describes the process as suitable for industrial production. The reactions can be performed continuously in the same vessel without isolating intermediates, which significantly simplifies the operational workflow and enhances scalability.
Q: What specific dehydrating agents are effective for the final nitrile formation step?
A: The patent lists several effective dehydrating agents including acid halides like acetyl chloride and thionyl chloride, acid anhydrides like acetic anhydride, and condensing agents like carbonyldiimidazole, offering flexibility based on cost and availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2-Nitrophenyl)acetonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your final products. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to maintaining stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to verify that every batch meets the exacting standards required by the global agrochemical industry. Our expertise in handling nitro-aromatic chemistry allows us to navigate the complexities of this synthesis with precision, delivering materials that empower your R&D and manufacturing teams to achieve their goals without compromise.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific volume and quality requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic benefits of switching to this superior manufacturing method. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that will drive value and efficiency across your entire supply chain. Partner with us to secure a sustainable and competitive advantage in the production of essential agrochemical building blocks.
