Advanced Manufacturing of 2-Substituted Benzaldehydes for Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical intermediates, particularly those serving as the backbone for potent therapeutic agents. Patent CN1078229A discloses a groundbreaking methodology for the preparation of 2-substituted benzaldehydes, a class of compounds indispensable in the synthesis of leukotriene antagonists used for treating asthma and other inflammatory conditions. This technology represents a significant departure from traditional multi-step syntheses, offering a streamlined pathway that enhances both chemical efficiency and process safety. By leveraging directed ortho-lithiation strategies on protected imine intermediates, the process achieves high regioselectivity and yield without relying on costly noble metal catalysts. 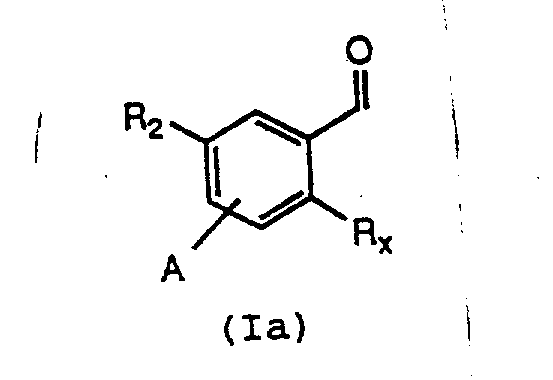 The structural versatility of these intermediates allows for the incorporation of diverse side chains, making them valuable building blocks for a wide array of bioactive molecules. For R&D directors and procurement specialists, understanding the nuances of this patented approach is crucial for securing a reliable supply of high-purity pharmaceutical intermediates.
The structural versatility of these intermediates allows for the incorporation of diverse side chains, making them valuable building blocks for a wide array of bioactive molecules. For R&D directors and procurement specialists, understanding the nuances of this patented approach is crucial for securing a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted benzaldehydes has been plagued by significant operational and economic challenges that hinder efficient large-scale manufacturing. Traditional routes often rely on palladium-catalyzed coupling reactions, such as the addition of substituted 1-alkynyl compounds to 2-halobenzaldehydes. While effective on a small scale, these methods introduce expensive transition metals that require rigorous and costly removal processes to meet stringent pharmaceutical purity standards. Furthermore, alternative pathways involving the conversion of 2-methoxybenzoic acid to oxazoline derivatives followed by Grignard reactions are inherently inefficient. These multi-step sequences involve harsh reagents, cryogenic conditions, and complex workup procedures that drastically increase the cost of goods sold (COGS). The reliance on stoichiometric amounts of specialized reagents and the generation of substantial chemical waste further exacerbate the environmental footprint, making these conventional methods unattractive for modern green chemistry initiatives and cost-sensitive supply chains.
The Novel Approach
In stark contrast, the novel process described in the patent utilizes a clever strategy of functional group protection and directed metallation to overcome these historical bottlenecks. By converting the starting 2-methylbenzaldehyde into a stable imine or hydrazone derivative, the reactive carbonyl group is masked, preventing unwanted nucleophilic attacks by strong bases. This protection allows for the selective deprotonation of the ortho-methyl group using organolithium reagents, creating a highly reactive nucleophile ready for alkylation. 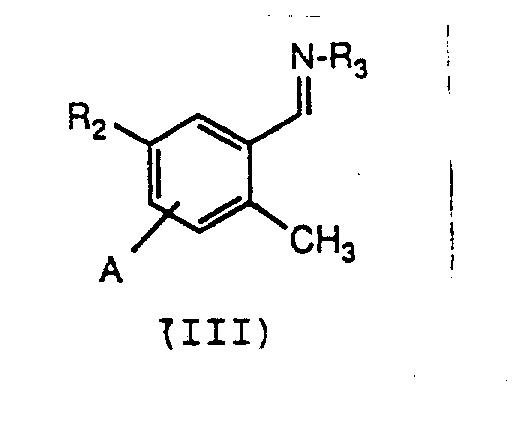 This approach eliminates the need for palladium catalysts entirely, replacing them with more economical and manageable lithium bases. The subsequent alkylation with various electrophiles introduces the desired side chain in a single pot, followed by a straightforward acid hydrolysis to regenerate the aldehyde functionality. This telescoped sequence not only reduces the number of isolation steps but also significantly improves the overall atom economy, presenting a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
This approach eliminates the need for palladium catalysts entirely, replacing them with more economical and manageable lithium bases. The subsequent alkylation with various electrophiles introduces the desired side chain in a single pot, followed by a straightforward acid hydrolysis to regenerate the aldehyde functionality. This telescoped sequence not only reduces the number of isolation steps but also significantly improves the overall atom economy, presenting a compelling value proposition for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Ortho-Lithiation and Alkylation
The core of this technological advancement lies in the precise control of the lithiation event on the aromatic ring. The process initiates with the formation of a Schiff base (Formula III), typically derived from the condensation of 2-methylbenzaldehyde with amines like tert-butylamine or hydrazines.  Upon treatment with a strong base such as n-butyllithium or lithium diisopropylamide (LDA), the ortho-methyl proton is abstracted. This deprotonation is facilitated by the coordination of the lithium cation to the nitrogen atom of the imine group, which acts as a directing group to position the base adjacent to the methyl group. This directed ortho-metalation (DoM) ensures that the lithiation occurs exclusively at the desired position, avoiding random substitution patterns that would complicate purification. The resulting benzylic carbanion is stabilized by the adjacent aromatic system and the coordinating imine nitrogen, creating a robust nucleophile capable of displacing leaving groups on a wide range of electrophiles.
Upon treatment with a strong base such as n-butyllithium or lithium diisopropylamide (LDA), the ortho-methyl proton is abstracted. This deprotonation is facilitated by the coordination of the lithium cation to the nitrogen atom of the imine group, which acts as a directing group to position the base adjacent to the methyl group. This directed ortho-metalation (DoM) ensures that the lithiation occurs exclusively at the desired position, avoiding random substitution patterns that would complicate purification. The resulting benzylic carbanion is stabilized by the adjacent aromatic system and the coordinating imine nitrogen, creating a robust nucleophile capable of displacing leaving groups on a wide range of electrophiles.
A critical discovery detailed in the patent is the unexpected benefit of operating at elevated temperatures when specific catalytic amines are employed. Conventionally, lithiation reactions are performed at cryogenic temperatures (e.g., -78°C) to prevent decomposition of the organolithium species. However, the data reveals that adding catalytic amounts of secondary amines like diisopropylamine or 2,2,6,6-tetramethylpiperidine allows the reaction to proceed efficiently between 15°C and 35°C. This thermal flexibility is attributed to the formation of mixed aggregates that modulate the basicity and nucleophilicity of the lithium species, thereby suppressing side reactions such as nucleophilic attack on the imine carbon. This mechanistic insight is pivotal for impurity control, as it minimizes the formation of by-products and simplifies the thermal management requirements for commercial reactors, directly translating to enhanced process robustness and reproducibility.
How to Synthesize 2-Substituted Benzaldehydes Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction parameters to maximize yield and purity. The process begins with the preparation of the imine intermediate, which serves as the crucial template for the subsequent lithiation. Following this, the choice of base and the addition of catalytic amines must be optimized based on the specific electrophile being used. The detailed standardized synthesis steps below outline the critical operational windows identified in the patent examples, ensuring that technical teams can replicate the high yields reported in the literature. Adhering to these protocols is essential for achieving the commercial viability and quality consistency required for pharmaceutical applications.
- Condense 2-methylbenzaldehyde with a primary amine or hydrazine to form a stable imine or hydrazone intermediate (Formula III).
- Treat the imine with a strong base like n-butyllithium or LDA, optionally with a catalytic amine, to deprotonate the ortho-methyl group, followed by addition of an electrophilic alkyl halide.
- Hydrolyze the resulting alkylated imine intermediate using aqueous acid to release the final 2-substituted benzaldehyde product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this lithiation-based methodology offers transformative benefits that extend far beyond simple chemical yield improvements. The elimination of palladium catalysts removes a major cost driver and a significant supply chain risk associated with volatile precious metal markets. Moreover, the ability to perform the reaction at near-ambient temperatures reduces energy consumption and alleviates the need for specialized cryogenic equipment, thereby lowering capital expenditure (CAPEX) for manufacturing facilities. The use of commercially available starting materials, such as o-tolualdehyde and common alkyl halides, ensures a stable and diversified supply base, mitigating the risk of raw material shortages. This process stability translates directly into reliable delivery schedules and consistent product quality, which are paramount for maintaining uninterrupted production of downstream active pharmaceutical ingredients.
- Cost Reduction in Manufacturing: The economic advantages of this process are driven primarily by the simplification of the synthetic route and the removal of expensive reagents. By avoiding palladium-catalyzed couplings, manufacturers eliminate the costs associated with purchasing precious metals and the complex downstream processing required to remove trace metal residues to ppm levels. Additionally, the telescoped nature of the alkylation and hydrolysis steps reduces solvent usage and labor hours, leading to substantial cost savings in operational expenditures. The improved yields observed with catalytic amine additives further enhance the economic profile by maximizing the output per batch, effectively lowering the unit cost of the final intermediate.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on commodity chemicals rather than specialized, custom-synthesized reagents. The starting materials and reagents, including organolithium bases and alkyl halides, are produced by multiple global suppliers, reducing dependency on single-source vendors. The robustness of the reaction conditions, particularly the tolerance for higher temperatures, minimizes the risk of batch failures due to minor temperature fluctuations or equipment limitations. This reliability ensures a continuous flow of high-purity pharmaceutical intermediates, allowing downstream partners to plan their production schedules with greater confidence and reduced safety stock requirements.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this process aligns well with modern green chemistry principles. The reduction in step count inherently decreases the volume of waste generated, while the absence of heavy metals simplifies wastewater treatment and disposal protocols. The exothermic nature of the lithiation can be safely managed at larger scales due to the wider operating temperature window, facilitating a smoother transition from pilot plant to commercial production. This scalability ensures that the supply of complex pharmaceutical intermediates can be rapidly ramped up to meet market demand without compromising on environmental compliance or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational feasibility and strategic advantages of the method. Understanding these details helps stakeholders make informed decisions about integrating this process into their existing manufacturing portfolios.
Q: Why are imine intermediates preferred over direct lithiation of benzaldehydes?
A: Direct lithiation of benzaldehydes is problematic due to the reactivity of the carbonyl group towards strong bases. Converting the aldehyde to an imine or hydrazone (Schiff base) protects the carbonyl functionality and directs lithiation specifically to the ortho-methyl position, preventing unwanted side reactions and improving regioselectivity.
Q: What is the advantage of using catalytic amines in the lithiation step?
A: The patent data indicates that adding catalytic amounts of organic amines, such as diisopropylamine or tetramethylpiperidine, prior to the base significantly improves yields. This modification allows the reaction to proceed at higher temperatures (15°C to 35°C) compared to traditional cryogenic conditions, minimizing side reactions and simplifying thermal management during scale-up.
Q: Can this process be scaled for commercial production of leukotriene antagonists?
A: Yes, the process is designed for industrial scalability. It utilizes readily available starting materials like o-tolualdehyde and avoids expensive transition metal catalysts like palladium. The one-pot nature of the alkylation and subsequent hydrolysis reduces unit operations, making it highly suitable for large-scale manufacturing of pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Substituted Benzaldehydes Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of life-saving medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the sophisticated lithiation chemistry described in CN1078229A can be executed with precision and consistency. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 2-substituted benzaldehydes meets the exacting standards required by global regulatory bodies. Our commitment to process excellence means that we can deliver complex intermediates with the reliability and quality necessary to support your drug development timelines.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our advanced manufacturing capabilities can add value to your portfolio. Let us be your partner in navigating the complexities of fine chemical synthesis and securing a sustainable future for your pharmaceutical projects.
