Revolutionizing Tetrahydroquinoxaline Production with Metal-Free Organic Catalysis for Commercial Scale
Revolutionizing Tetrahydroquinoxaline Production with Metal-Free Organic Catalysis for Commercial Scale
The landscape of heterocyclic synthesis is undergoing a significant transformation, driven by the urgent need for greener, more cost-effective, and scalable manufacturing processes. A pivotal advancement in this domain is detailed in patent CN112266364A, which discloses a novel preparation method for tetrahydroquinoxaline compounds. This technology represents a paradigm shift from traditional transition metal-catalyzed hydrogenation to a metal-free protocol utilizing organic small molecule catalysts. For R&D directors and procurement strategists in the pharmaceutical and fine chemical sectors, this innovation offers a compelling solution to long-standing challenges regarding catalyst cost, metal residue removal, and operational safety. By leveraging inexpensive quaternary ammonium salts and readily available hydride sources, this method achieves high yields under mild conditions, positioning it as a superior alternative for the commercial production of high-value intermediates.
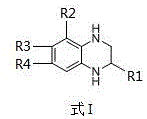
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of tetrahydroquinoxaline scaffolds has relied heavily on transition metal catalysis, typically employing precious metals such as palladium, platinum, or nickel under high-pressure hydrogen atmospheres. While effective, these conventional methods impose severe constraints on industrial scalability and cost efficiency. Transition metal catalysts are not only expensive but also exhibit high sensitivity to air and moisture, necessitating rigorous inert atmosphere handling and specialized equipment. Furthermore, the presence of residual heavy metals in the final product is a critical regulatory hurdle for pharmaceutical applications, often requiring complex and costly purification steps involving metal scavengers. These factors collectively inflate the manufacturing cost and extend the lead time, creating bottlenecks in the supply chain for key drug intermediates.
The Novel Approach
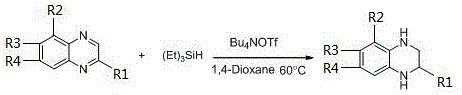
In stark contrast, the methodology outlined in patent CN112266364A introduces a robust, metal-free hydrogenation strategy that circumvents these traditional limitations. By utilizing organic small molecules such as tetrabutylammonium bromide (Bu4NBr) or tetrabutylammonium iodide (Bu4NI) as catalysts, the process eliminates the dependency on scarce transition metals entirely. The reaction employs benign hydride donors like pinacolborane (HBpin) or triethylsilane, operating effectively at mild temperatures ranging from 50°C to 70°C. This approach not only simplifies the operational setup by removing the need for high-pressure hydrogenation reactors but also ensures that the final product is free from toxic metal contaminants. The result is a streamlined, environmentally friendly process that delivers exceptional yields, often exceeding 90%, while drastically reducing the complexity of downstream purification.
Mechanistic Insights into Organic Small Molecule-Catalyzed Hydrogenation
The efficacy of this novel synthesis route lies in the unique activation mechanism facilitated by the quaternary ammonium salt catalysts. Unlike transition metals that activate hydrogen through oxidative addition, these organic catalysts likely function by enhancing the nucleophilicity of the hydride source or by stabilizing charged intermediates during the reduction of the electron-deficient quinoxaline ring. The catalyst interacts with the borane or silane reagent to generate a reactive hydride species capable of selectively reducing the C=N bonds of the quinoxaline core. This mechanistic pathway is highly chemoselective, preferentially targeting the heterocyclic double bonds while leaving other sensitive functional groups—such as halogens (fluoro, chloro, bromo) and nitro groups—intact. This level of precision is crucial for synthesizing complex pharmaceutical intermediates where orthogonal reactivity is required for subsequent coupling steps.
Furthermore, the reaction system demonstrates remarkable tolerance to various substituents at the R1, R2, R3, and R4 positions, as defined in the general structure. Whether the substrate bears electron-donating alkyl groups or electron-withdrawing halogens, the catalytic cycle maintains high efficiency, delivering consistent yields across a broad substrate scope. The absence of radical pathways, which are common in metal-catalyzed reductions, minimizes the formation of side products and oligomers, thereby simplifying the impurity profile. For quality control teams, this translates to a cleaner crude reaction mixture, reducing the burden on analytical resources and ensuring that the final active pharmaceutical ingredient (API) precursor meets stringent purity specifications with minimal effort.
How to Synthesize Tetrahydroquinoxaline Efficiently
The practical implementation of this synthesis is straightforward and amenable to standard laboratory and plant equipment. The process begins with the precise weighing of the quinoxaline starting material and the organic catalyst, followed by the addition of the hydride source and solvent under ambient or slightly controlled atmospheric conditions. The reaction mixture is then heated to a moderate temperature, typically around 60°C, and stirred until conversion is complete, as monitored by standard analytical techniques. Upon completion, the workup involves a simple solvent removal step followed by column chromatography, avoiding the need for aqueous quenches or complex extractions often associated with metal hydrides. This operational simplicity significantly reduces the technical barrier for adoption, allowing manufacturing teams to integrate this route into existing production lines with minimal retrofitting.
- Charge a reaction vessel with quinoxaline starting material, an organic small molecule catalyst (e.g., Bu4NBr), and a hydride source (e.g., pinacolborane) in an organic solvent.
- Stir the reaction mixture at a constant temperature between 50°C and 70°C until the hydrogenation is complete.
- Remove volatile solvents under reduced pressure and purify the crude residue via column chromatography to isolate the high-purity tetrahydroquinoxaline product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this metal-free technology offers transformative economic and logistical benefits. The primary advantage stems from the drastic reduction in raw material costs; replacing expensive transition metal catalysts and ligands with commodity quaternary ammonium salts results in immediate savings on the bill of materials. Additionally, the elimination of heavy metals removes the necessity for costly metal scavenging resins and the associated validation testing, further lowering the overall cost of goods sold (COGS). The mild reaction conditions also contribute to energy efficiency, as the process does not require high-temperature or high-pressure infrastructure, thereby reducing utility consumption and maintenance overheads in the manufacturing facility.
- Cost Reduction in Manufacturing: The substitution of precious metal catalysts with inexpensive organic salts fundamentally alters the cost structure of tetrahydroquinoxaline production. By removing the need for palladium or platinum complexes, manufacturers can achieve substantial cost savings on catalyst procurement. Moreover, the simplified downstream processing, which avoids complex metal removal steps, reduces solvent usage and waste disposal costs. This leaner manufacturing footprint allows for more competitive pricing strategies in the global market for pharmaceutical intermediates, enhancing margin potential without compromising on quality standards.
- Enhanced Supply Chain Reliability: Reliance on transition metals often exposes supply chains to geopolitical risks and price volatility associated with mining and refining operations. In contrast, the reagents used in this novel method, such as tetrabutylammonium bromide and pinacolborane, are widely available commodity chemicals with stable supply networks. This shift ensures greater continuity of supply and reduces the risk of production stoppages due to raw material shortages. Furthermore, the robustness of the reaction conditions means that the process is less susceptible to variations in environmental factors, leading to more predictable batch cycles and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this metal-free protocol aligns perfectly with modern green chemistry principles. The absence of toxic heavy metals simplifies waste stream management and reduces the environmental liability associated with hazardous waste disposal. The process generates fewer byproducts and utilizes safer reagents, making it easier to obtain regulatory approvals for new manufacturing sites. Scalability is inherently supported by the homogeneous nature of the reaction and the use of common organic solvents, facilitating a seamless transition from kilogram-scale development to multi-ton commercial production without the need for specialized high-pressure reactors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this metal-free hydrogenation technology. These insights are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their current manufacturing portfolios for high-purity heterocyclic compounds.
Q: What are the advantages of using organic small molecule catalysts over transition metals?
A: Organic catalysts like tetrabutylammonium bromide eliminate the risk of heavy metal contamination, removing the need for expensive scavenging steps and ensuring compliance with strict pharmaceutical impurity limits.
Q: Can this method tolerate sensitive functional groups on the quinoxaline ring?
A: Yes, the mild reaction conditions (50-70°C) and high chemoselectivity allow for the tolerance of halogens, nitro groups, and alkyl substituents without side reactions.
Q: Is this process suitable for large-scale manufacturing?
A: Absolutely. The use of cheap, commercially available reagents and simple workup procedures makes this method highly scalable and cost-effective for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tetrahydroquinoxaline Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting innovative synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of the metal-free hydrogenation method described in CN112266364A and is fully prepared to leverage this technology for your specific project needs. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale discovery to industrial manufacturing is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of tetrahydroquinoxaline intermediate delivered meets the highest international standards.
We invite you to collaborate with us to optimize your supply chain and reduce manufacturing costs through this advanced catalytic technology. Our technical sales team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a reliable tetrahydroquinoxaline supplier committed to delivering high-purity pharmaceutical intermediates with unmatched speed, quality, and cost efficiency.
