Advanced Synthesis of 4-Haloalkylnicotinonitriles for High-Efficiency Agrochemical Production
Advanced Synthesis of 4-Haloalkylnicotinonitriles for High-Efficiency Agrochemical Production
The global demand for high-performance agrochemicals continues to drive innovation in intermediate synthesis, particularly for heterocyclic compounds that serve as the backbone of modern insecticides. Patent CN1244560C introduces a groundbreaking methodology for the preparation of 4-haloalkylnicotinonitriles, a class of compounds essential for producing next-generation crop protection agents. This technology represents a significant departure from traditional synthetic routes, offering a more direct and economically viable pathway to these valuable building blocks. By utilizing a condensation reaction between 3-amino-1-haloalkyl-2-propen-1-one and specific nitrile-containing precursors, the process achieves high selectivity and yield without the need for hazardous activated acid derivatives. For R&D directors and procurement specialists in the agrochemical sector, understanding this proprietary chemistry is crucial for securing a reliable supply chain of high-purity intermediates.
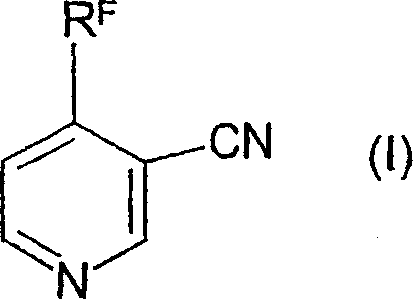
The core innovation lies in the ability to construct the pyridine ring system efficiently while maintaining the integrity of sensitive haloalkyl groups, such as the trifluoromethyl moiety. Conventional methods often struggle with the harsh conditions required to introduce these groups or involve multi-step sequences that degrade overall process efficiency. In contrast, the disclosed method operates under relatively mild conditions, utilizing reduced pressure to drive the condensation equilibrium forward. This approach not only minimizes the formation of unwanted by-products but also facilitates the removal of low-boiling constituents like alcohols, thereby pushing the reaction to completion. The resulting intermediates can be directly cyclized to form the target nicotinonitriles, which can subsequently be hydrolyzed to nicotinamides or further derivatized into active pesticide ingredients.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4-haloalkylnicotinic acid derivatives has relied on pathways starting from 4-haloalkylnicotinic acids, which themselves require complex preparation involving activated intermediates. Traditional routes frequently necessitate the conversion of carboxylic acids into acid chlorides using thionyl chloride or oxalyl chloride, reagents that are corrosive, moisture-sensitive, and generate significant amounts of acidic waste. Furthermore, subsequent reactions with ammonia to form amides, followed by dehydration to nitriles, add unnecessary unit operations that increase both capital expenditure and operational complexity. These multi-step processes often suffer from cumulative yield losses and require rigorous purification steps to remove residual halogens and heavy metals, posing challenges for environmental compliance and cost management in large-scale manufacturing facilities.
The Novel Approach
The novel approach detailed in the patent circumvents these bottlenecks by employing a convergent synthesis strategy that builds the pyridine ring from acyclic precursors. As illustrated in the reaction scheme below, the process begins with the condensation of an enaminone derivative with various nitrile sources, such as acetals or vinyl nitriles. This single-step construction of the carbon-nitrogen framework eliminates the need for pre-formed pyridine rings and avoids the use of aggressive chlorinating agents entirely. The reaction is designed to proceed through stable intermediates that can be isolated or carried forward in a one-pot sequence, drastically simplifying the workflow. By removing the dependency on acid chlorides and ammonia, the new method enhances operator safety and reduces the environmental footprint associated with waste disposal, making it an attractive option for sustainable chemical manufacturing.
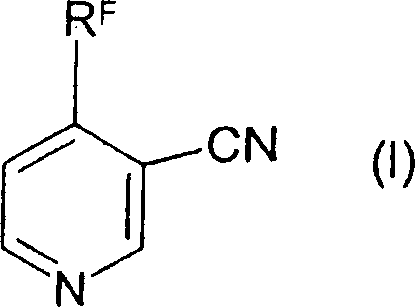
Mechanistic Insights into Condensation and Cyclization
The mechanistic pathway involves a sophisticated interplay of nucleophilic attack and elimination reactions governed by base catalysis. Initially, the amino group of the 3-amino-1-haloalkyl-2-propen-1-one acts as a nucleophile, attacking the electrophilic center of the nitrile precursor. In the case of acetals like 3,3-dimethoxypropionitrile, this leads to the displacement of an alkoxide group, forming a linear intermediate containing both the enaminone and nitrile functionalities. The reaction conditions, specifically the application of reduced pressure (5 to 150 mbar), are critical for shifting the equilibrium by continuously removing the volatile alcohol by-product. This thermodynamic driving force ensures high conversion rates even at moderate temperatures ranging from 0°C to 30°C, preserving the stability of the sensitive trifluoromethyl group against decomposition or defluorination.
Following the condensation, the linear intermediate undergoes an intramolecular cyclization to close the pyridine ring. This step is typically catalyzed by weak bases such as lithium carbonate or potassium carbonate in an alcoholic solvent. The mechanism likely proceeds through a tautomeric equilibrium where the enamine nitrogen attacks the nitrile carbon or an adjacent electrophilic site, followed by aromatization. The patent highlights the existence of various tautomers and isomers during this phase, which rapidly interconvert in solution before locking into the stable aromatic pyridine structure upon acidification. Understanding this dynamic equilibrium is vital for optimizing reaction times and selecting the appropriate base to maximize the selectivity towards the desired 4-substituted isomer, minimizing the formation of regioisomeric impurities that could complicate downstream processing.
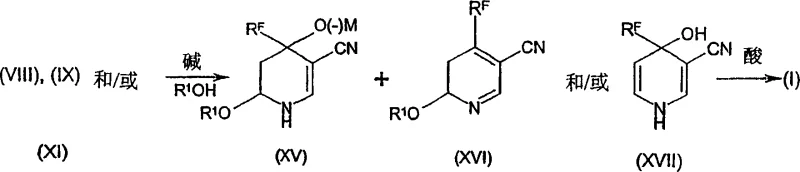
How to Synthesize 4-Haloalkylnicotinonitriles Efficiently
The synthesis protocol outlined in the patent provides a robust framework for producing these intermediates with high purity and consistency. The process is adaptable to both batch and continuous flow reactors, offering flexibility for different production scales. Key to the success of this method is the precise control of reaction parameters, including temperature, pressure, and stoichiometry, to ensure complete conversion while suppressing side reactions. The following guide summarizes the critical operational steps derived from the experimental examples, serving as a foundational reference for process chemists aiming to implement this technology.
- Condense 3-amino-1-haloalkyl-2-propen-1-one with compounds such as (R1Z)CH=CH-CN or (R1Z)2CH-CH2-CN under reduced pressure to form intermediates.
- Facilitate the elimination of alcohol or hydrogen halide molecules to generate the linear enaminone-nitrile precursor.
- Perform a ring-closure reaction in the presence of a weak base and alcohol solvent, followed by acidification to yield the final pyridine derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this synthesis route offers tangible benefits in terms of cost structure and supply reliability. By eliminating the need for expensive and hazardous activated acid derivatives, the process significantly reduces the raw material costs associated with reagents and waste treatment. The simplified workflow, which potentially allows for a one-pot operation, decreases the number of isolation and purification steps, leading to lower energy consumption and reduced solvent usage. This streamlining of the manufacturing process translates directly into improved margins and a more competitive pricing structure for the final agrochemical intermediates, enabling buyers to secure high-quality materials at optimized costs without compromising on specification standards.
- Cost Reduction in Manufacturing: The avoidance of acid chlorides and ammonia removes the necessity for specialized corrosion-resistant equipment and extensive scrubbing systems, resulting in substantial capital and operational expenditure savings. Furthermore, the high atom economy of the condensation reaction minimizes waste generation, lowering the costs associated with effluent treatment and regulatory compliance. The ability to recover and reuse solvents and catalysts, such as the weak bases employed in the cyclization step, further enhances the economic viability of the process, ensuring long-term cost stability for bulk purchasers.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, including 3-amino-1-haloalkyl-2-propen-1-one and various nitrile precursors, are commercially available or can be produced via established methods, reducing the risk of supply disruptions. The robustness of the reaction conditions, which tolerate a range of temperatures and pressures, ensures consistent production output even in varying operational environments. This reliability is crucial for maintaining uninterrupted supply chains for downstream pesticide manufacturers, who depend on timely deliveries of critical intermediates to meet seasonal agricultural demands and global market requirements.
- Scalability and Environmental Compliance: The process has been demonstrated to scale effectively from laboratory glassware to tubular reactors, indicating strong potential for commercial scale-up of complex agrochemical intermediates. The reduced use of hazardous reagents and the generation of less toxic by-products align with increasingly stringent environmental regulations, facilitating easier permitting and operation in diverse geographic regions. This environmental compatibility not only mitigates regulatory risks but also enhances the corporate sustainability profile of companies adopting this greener synthetic methodology, appealing to eco-conscious stakeholders and customers.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and experimental data, providing clarity on the process capabilities and limitations. Understanding these details helps stakeholders make informed decisions about integrating this method into their existing production portfolios.
Q: What are the primary advantages of this synthesis route over conventional acid chloride methods?
A: This novel process eliminates the need for activated acid derivatives like acid chlorides and avoids hazardous ammonia reaction steps, significantly simplifying the workflow and reducing safety risks associated with corrosive reagents.
Q: Can this process be scaled for commercial production of insecticide intermediates?
A: Yes, the patent demonstrates scalability through examples using tubular reactors and standard batch conditions, achieving high purity (up to 99%) and robust yields suitable for industrial manufacturing.
Q: What specific haloalkyl groups are compatible with this synthesis method?
A: The process is highly versatile, accommodating various halo(C1-C4) alkyl radicals, with a particular preference for trifluoromethyl (CF3) groups, which are critical for enhancing the biological activity of modern pesticides.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Haloalkylnicotinonitrile Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the agrochemical value chain. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising laboratory results of patent CN1244560C can be successfully translated into industrial reality. We are committed to delivering high-purity 4-haloalkylnicotinonitriles that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our dedication to quality assurance guarantees that every batch supplied adheres to the highest standards, providing our partners with the confidence needed to advance their own pesticide development programs.
We invite global agrochemical companies to collaborate with us to leverage this advanced synthesis technology for their specific needs. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and explore the feasibility of implementing this route in your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive efficiency and value in your manufacturing operations.
