Advanced Synthesis of Tryptase Inhibitor Intermediates for Commercial Scale-Up
Introduction to Novel Tryptase Inhibitor Synthesis
The pharmaceutical landscape for treating mast cell-mediated inflammatory disorders, particularly asthma and chronic inflammation, relies heavily on the efficient production of high-purity tryptase inhibitors. Patent CN1950362A introduces a transformative methodology for preparing compounds of Formula I and their salts, addressing critical bottlenecks in the existing supply chain. This intellectual property outlines a robust synthetic pathway that circumvents the inherent dangers associated with handling unstable phenylacetylene derivatives, which are prone to violent decomposition at elevated temperatures. By shifting the paradigm towards in situ generation of reactive intermediates from stable silyl precursors, the technology ensures a safer operational environment for chemical manufacturing teams. Furthermore, the patent details innovative routes for synthesizing key piperidine intermediates that drastically reduce reliance on expensive and toxic reagents used in previous methodologies. For R&D Directors and Procurement Managers, this represents a significant opportunity to optimize the cost of goods sold (COGS) while enhancing process safety profiles. The strategic implementation of these methods allows for the reliable production of complex pharmaceutical intermediates required for next-generation anti-inflammatory therapies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methods, such as those disclosed in WO 2004/060884, suffer from severe technical and economic drawbacks that hinder commercial viability. The traditional synthesis of key intermediates often necessitates the use of 2-(trimethylsilyl)ethoxycarbonyl (Teoc) protecting groups, which require high-vacuum distillation at temperatures ranging from 156°C to 160°C. This energy-intensive step not only increases operational costs but also introduces significant thermal risks during scale-up. Additionally, conventional routes frequently employ expensive triflating agents to activate enolates for Suzuki coupling, alongside costly palladium catalysts like tetrakis(triphenylphosphine)palladium(0). The reliance on chromatographic purification for multiple intermediates further exacerbates waste generation and reduces overall throughput. From a supply chain perspective, the use of unstable phenylacetylenes as starting materials poses a critical safety hazard, as these compounds can decompose explosively, necessitating specialized containment and handling protocols that drive up insurance and facility costs. These cumulative inefficiencies create a fragile supply chain vulnerable to disruptions and cost volatility.
The Novel Approach
The methodology presented in CN1950362A offers a decisive break from these constraints by introducing a safer and more economical synthetic strategy. A cornerstone of this innovation is the in situ generation of phenylacetylene from corresponding silyl compounds using mild base catalysis, thereby eliminating the need to isolate hazardous acetylenic intermediates. This approach significantly mitigates safety risks associated with thermal decomposition. Furthermore, the patent describes an improved route for preparing the pivotal 4-[3-(tert-butoxycarbonylaminomethyl)phenyl]piperidine intermediate. By utilizing Boc protection instead of Teoc and employing direct hydrogenation strategies over platinum or palladium catalysts, the process avoids the use of toxic nickel chloride and expensive triflates. The new method favors crystallization and extraction over chromatography for purification, which is a critical advantage for industrial scale-up. This streamlined workflow reduces solvent consumption, minimizes waste disposal requirements, and enhances the overall robustness of the manufacturing process, making it highly attractive for commercial production.
Mechanistic Insights into Sonogashira Coupling and Intermediate Formation
The core chemical transformation in this patent involves a palladium and copper-catalyzed Sonogashira coupling reaction, which is meticulously optimized to ensure high fidelity and yield. The process begins with the treatment of a phenylethynylsilane compound with a base, such as potassium carbonate, in a solvent system comprising methanol or a methanol-tetrahydrofuran mixture. This step cleaves the silyl protecting group to generate the reactive phenylacetylene species in solution without isolation. Subsequently, this solution is merged with a bromo- or iodo-substituted furan-amide intermediate in the presence of a homogeneous palladium catalyst, specifically bis(triphenylphosphine)palladium(II) chloride, and a cuprous salt like copper(I) iodide. The reaction proceeds under mild thermal conditions, typically between 30°C and 70°C, facilitated by a hindered amine base such as triethylamine. This catalytic cycle efficiently forms the carbon-carbon triple bond linkage essential for the tryptase inhibitor's pharmacological activity. The careful selection of ligands and catalysts ensures that the reaction tolerates various functional groups, maintaining the integrity of the sensitive Boc-protected amine moieties throughout the synthesis.
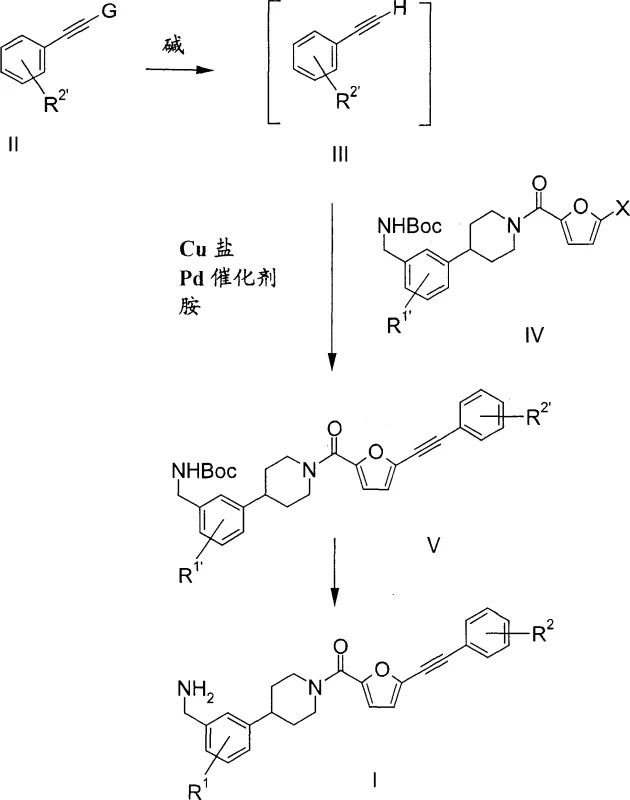
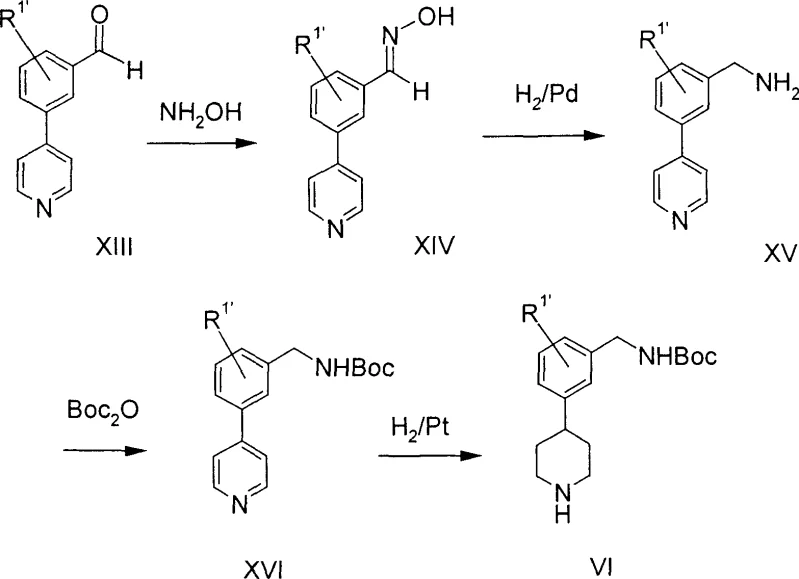
Parallel to the final coupling, the synthesis of the piperidine intermediate is achieved through a novel sequence that avoids the pitfalls of prior art. One preferred embodiment involves the conversion of a pyridylbenzaldehyde derivative into an oxime, followed by catalytic hydrogenation to yield the corresponding benzylamine. This amine is then protected with di-tert-butyl dicarbonate to form a stable Boc-protected intermediate. The final step involves the hydrogenation of the pyridine ring to a piperidine using a platinum catalyst under acidic conditions, a transformation that surprisingly leaves the Boc group intact. This chemoselectivity is crucial for maintaining process efficiency, as it eliminates the need for re-protection steps. The mechanistic understanding of these hydrogenation conditions allows for precise control over reaction parameters, ensuring that the final intermediate is produced with high purity and minimal byproduct formation. This level of control is essential for meeting the stringent quality standards required for pharmaceutical active ingredients.
How to Synthesize Tryptase Inhibitor Intermediates Efficiently
Implementing this synthesis route requires a systematic approach to reaction engineering and process control to maximize yield and safety. The detailed standardized synthesis steps involve precise control of stoichiometry, temperature, and pressure during the hydrogenation and coupling phases. Operators must ensure that the in situ generation of phenylacetylene is complete before introducing the coupling partners to prevent the accumulation of unreacted silyl species. The purification strategy relies heavily on pH-controlled extractions and crystallization from solvent systems like isopropanol and diisopropyl ether, which are scalable and cost-effective. Adhering to these protocols ensures that the final product meets the required specifications for downstream drug formulation. For a comprehensive guide on the specific operational parameters, please refer to the standardized procedure outlined below.
- Generate phenylacetylene in situ from silyl compounds using base catalysis to avoid handling unstable acetylenes.
- Perform Pd/Cu-catalyzed Sonogashira coupling with furan-amide intermediates under mild conditions.
- Execute Boc-deprotection using methanesulfonic acid to yield the final active pharmaceutical salt.
Commercial Advantages for Procurement and Supply Chain Teams
The adoption of the synthetic methods described in CN1950362A offers profound commercial benefits that extend beyond mere technical feasibility, directly impacting the bottom line for pharmaceutical manufacturers. By eliminating the need for hazardous phenylacetylene storage and handling, facilities can significantly reduce their safety compliance costs and insurance premiums. The shift from chromatographic purification to crystallization-based isolation drastically lowers solvent consumption and waste disposal fees, contributing to a more sustainable and cost-efficient operation. Moreover, the replacement of expensive reagents like Teoc-Cl and triflating agents with commodity chemicals such as Boc anhydride and simple mineral acids results in substantial raw material cost savings. These efficiencies translate into a more resilient supply chain capable of withstanding market fluctuations in reagent pricing. For procurement managers, this means securing a reliable source of high-quality intermediates at a reduced total cost of ownership, enabling more competitive pricing for the final therapeutic products.
- Cost Reduction in Manufacturing: The elimination of high-vacuum distillation steps and expensive triflating agents directly lowers energy and material expenditures. By utilizing stable silyl precursors instead of unstable acetylenes, the process reduces the need for specialized safety infrastructure, further driving down capital and operational expenses. The streamlined purification workflow minimizes solvent usage and waste generation, leading to significant savings in environmental compliance and disposal costs. Overall, the process design prioritizes atom economy and operational simplicity, ensuring that the manufacturing of tryptase inhibitor intermediates remains economically viable even at large scales.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials mitigates the risk of supply disruptions caused by the scarcity of specialized reagents. The robustness of the synthetic route, characterized by its tolerance to varying reaction conditions and its ability to produce high-purity products without complex purification, ensures consistent output quality. This reliability is critical for maintaining continuous production schedules and meeting the demanding delivery timelines of global pharmaceutical clients. By reducing the number of unit operations and intermediate isolations, the process also shortens the overall manufacturing cycle time, allowing for faster response to market demand.
- Scalability and Environmental Compliance: The process is inherently designed for scale-up, avoiding unit operations that are difficult to translate from laboratory to plant scale, such as flash chromatography. The use of common solvents like methanol, toluene, and isopropanol simplifies solvent recovery and recycling systems, aligning with green chemistry principles. The reduction in hazardous waste generation and the avoidance of toxic heavy metal residues in the final product facilitate easier regulatory approval and environmental permitting. This alignment with sustainability goals not only reduces regulatory risk but also enhances the corporate social responsibility profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and comparative examples provided within the patent documentation, offering clarity on safety, cost, and scalability. Understanding these aspects is crucial for stakeholders evaluating the integration of this synthesis route into their existing manufacturing portfolios. The answers reflect the specific advantages of the novel method over prior art, highlighting its potential to transform the production landscape for tryptase inhibitors.
Q: How does this patent improve safety over prior art methods?
A: The patent avoids isolating unstable phenylacetylenes which decompose violently at high temperatures, instead generating them in situ from stable silyl precursors.
Q: What are the cost advantages of the new intermediate synthesis?
A: The new route eliminates expensive reagents like Teoc-Cl and triflating agents, replacing them with cost-effective Boc protection and direct hydrogenation strategies.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the process minimizes chromatographic purifications in favor of crystallization and extraction, significantly enhancing scalability and throughput.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tryptase Inhibitor Intermediate Supplier
The technical advancements detailed in CN1950362A underscore the complexity and precision required to manufacture high-value pharmaceutical intermediates efficiently. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to quality is evidenced by our stringent purity specifications and rigorous QC labs, which ensure that every batch of tryptase inhibitor intermediate meets the highest global standards. We understand the critical nature of these compounds in the development of life-saving anti-inflammatory therapies and are dedicated to providing a supply chain partnership that prioritizes safety, consistency, and technical excellence. Our team of expert chemists is well-versed in the nuances of Sonogashira coupling and catalytic hydrogenation, ensuring seamless technology transfer and process optimization.
We invite you to collaborate with us to leverage these innovative synthetic routes for your specific project needs. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements and quality targets. We encourage potential partners to reach out for specific COA data and route feasibility assessments to verify how our capabilities align with your supply chain goals. Let us help you navigate the complexities of fine chemical manufacturing with a partner who values precision and reliability above all else.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
