Scalable Production of 6-Fluoro-2-Methylindole: A Breakthrough in Catalytic Hydrogenation Technology
The pharmaceutical and agrochemical industries constantly seek robust synthetic routes for heterocyclic scaffolds, particularly indole derivatives which serve as critical building blocks for bioactive molecules. Patent CN1286832C introduces a transformative methodology for producing indole compounds, specifically addressing the long-standing challenges associated with the synthesis of 6-fluoro-2-methylindole. This patent discloses a highly efficient one-pot reductive cyclization process that converts 2-nitrobenzyl carbonyl compounds directly into the corresponding indole structure with exceptional yield and purity. Unlike legacy methods that suffer from harsh conditions and poor atom economy, this invention leverages a sophisticated interplay between catalytic hydrogenation and in-situ acylation. For procurement specialists and R&D leaders, this technology represents a significant opportunity to optimize supply chains for high-purity agrochemical intermediates. By shifting from stoichiometric metal reductions to catalytic processes, manufacturers can achieve substantial improvements in process safety and environmental compliance while maintaining rigorous quality standards required for downstream fungicide production.
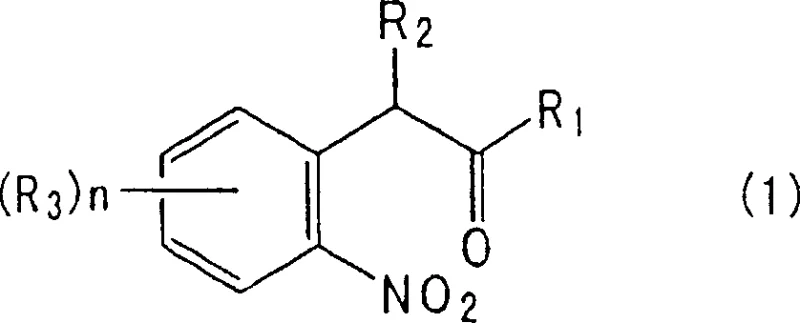
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-substituted indoles has been plagued by inefficient methodologies that impose severe operational burdens on manufacturing facilities. Early literature, such as DE262327, describes thermal cyclization requiring temperatures as high as 360°C with barium oxide, leading to significant energy consumption and equipment degradation. Alternative routes involving phenylhydrazones necessitate reaction temperatures around 240°C with sodium hydroxide, often resulting in complex impurity profiles and low isolated yields. Furthermore, the most common industrial approach has traditionally involved the reduction of nitro precursors using stoichiometric amounts of iron or zinc in acidic media. While chemically feasible, these methods generate massive quantities of iron oxide or zinc oxide sludge as waste by-products. The disposal of this heavy metal waste not only incurs high environmental compliance costs but also complicates the workup procedure, requiring extensive filtration and washing steps that reduce overall throughput. Additionally, direct catalytic hydrogenation without modification often leads to over-reduction, producing unwanted indoline by-products that are difficult to separate, thereby compromising the purity of the final pharmaceutical intermediates.
The Novel Approach
The methodology outlined in CN1286832C fundamentally reengineers the reduction landscape by introducing a dual-function system where an acylating agent and a base coexist with the hydrogen donor and reduction catalyst. This innovative strategy allows for the conversion of 2-nitrobenzyl carbonyl compounds, such as 4-fluoro-2-nitrophenylacetone, into indoles in a single reaction vessel. Experimental data from the patent demonstrates that using a palladium on carbon catalyst with acetic anhydride and sodium acetate at a mild 50°C can achieve yields as high as 95%. This represents a drastic improvement over the roughly 70% yields observed in unmodified catalytic reductions. The process effectively suppresses the formation of indoline by-products, which are typically formed via the reduction of intermediate N-oxides. By streamlining the synthesis into a single stage with minimal waste generation, this approach offers a clear pathway for cost reduction in agrochemical intermediates manufacturing. The ability to operate under atmospheric pressure and moderate temperatures further enhances the safety profile, making it an attractive option for commercial scale-up of complex heterocyclic compounds.
Mechanistic Insights into Acylation-Assisted Reductive Cyclization
To fully appreciate the technical superiority of this process, one must understand the intricate mechanistic pathway that governs the selectivity. During the reduction of the nitro group, the reaction proceeds through a 1-hydroxyindole intermediate. In conventional systems, this intermediate exists in a tautomeric equilibrium with a 2-methylindolenine N-oxide. This N-oxide species is highly susceptible to further reduction by the hydrogenation catalyst, leading to the formation of the saturated indoline ring, which is an undesirable impurity. The breakthrough in this patent lies in the immediate trapping of the 1-hydroxyindole intermediate. The presence of an acylating agent, such as acetic anhydride, reacts rapidly with the hydroxyl group to form a stable 1-acyloxyindole ester. This acylation step effectively pulls the equilibrium away from the N-oxide tautomer, rendering the intermediate inert to further reduction of the double bond. Consequently, the subsequent hydrolysis or workup yields the aromatic indole system with high fidelity.
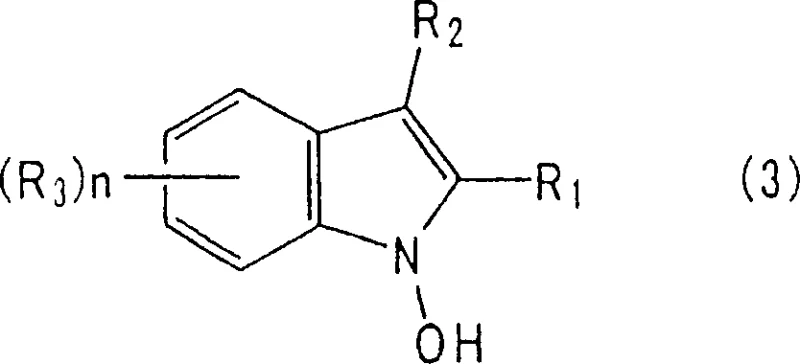
This mechanistic control is critical for ensuring the integrity of the impurity profile, a key concern for R&D Directors overseeing process validation. The base, typically an alkali metal salt like sodium acetate or potassium carbonate, plays a dual role by neutralizing the acid generated during acylation and potentially activating the catalyst surface. The patent specifies that the amount of acylating agent should ideally be between 1 to 5 moles per mole of substrate to ensure complete trapping of the intermediate. Furthermore, the choice of catalyst is pivotal; palladium supported on activated carbon is highlighted as particularly effective due to its balance of activity and selectivity. This deep understanding of the reaction kinetics allows for precise tuning of parameters to minimize trace impurities, ensuring that the resulting high-purity indole compounds meet the stringent specifications required for active pharmaceutical ingredient (API) synthesis or advanced agrochemical formulations.
How to Synthesize 6-Fluoro-2-Methylindole Efficiently
Implementing this synthesis route requires careful attention to reagent quality and reaction monitoring to maximize the benefits of the patented technology. The process begins with the preparation or sourcing of high-quality 2-nitrobenzyl carbonyl precursors, which can themselves be synthesized via novel condensation reactions described in the patent to ensure optimal purity. The core transformation involves suspending the nitro ketone in a solvent such as toluene, adding the palladium catalyst, and introducing the acylating agent and base before initiating hydrogen flow. Maintaining the temperature around 50°C is crucial to balance reaction rate with selectivity. Detailed standard operating procedures for this specific transformation, including precise stoichiometric ratios and workup protocols, are essential for reproducibility. For technical teams looking to adopt this methodology, the following guide outlines the critical operational steps derived from the patent examples to ensure successful implementation.
- Prepare the 2-nitrobenzyl carbonyl starting material, such as 4-fluoro-2-nitrophenylacetone, ensuring high purity to minimize side reactions during reduction.
- Conduct reductive cyclization in a solvent like toluene using a palladium on carbon catalyst, hydrogen gas, acetic anhydride, and a base like sodium acetate at 50°C.
- Filter the catalyst and perform aqueous workup to isolate the indole compound, achieving yields exceeding 95% without heavy metal waste.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this catalytic hydrogenation technology offers profound advantages that extend beyond simple yield improvements. For Procurement Managers, the elimination of stoichiometric metal reagents like iron powder or zinc dust translates directly into reduced raw material volatility and simplified logistics. Traditional methods require the handling and storage of large volumes of metal powders, which pose safety risks and require dedicated containment infrastructure. By switching to a catalytic system with recoverable palladium on carbon, the facility can significantly lower its inventory overhead and reduce the frequency of raw material deliveries. Moreover, the removal of heavy metal waste streams simplifies the supply chain for waste disposal services, which are often subject to fluctuating regulatory costs and availability. This process stability ensures a more predictable production schedule, reducing lead time for high-purity agrochemical intermediates and allowing for more responsive fulfillment of customer orders.
- Cost Reduction in Manufacturing: The economic impact of this process is driven primarily by the simplification of the downstream processing train. In traditional iron/zinc reductions, the filtration of metal sludge is a bottleneck that consumes significant labor and time, often requiring multiple washing cycles to remove residual metals. This new method allows for simple catalyst filtration through celite, followed by a straightforward aqueous wash. The absence of heavy metal contamination means that expensive purification steps, such as chelation or specialized adsorption, are rendered unnecessary. Additionally, the high selectivity minimizes the loss of valuable starting material to by-products, improving the overall mass balance. While specific percentage savings depend on local utility costs, the qualitative reduction in waste treatment fees and labor hours results in a substantially lower cost of goods sold (COGS).
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the environmental regulations surrounding heavy metal usage. Facilities utilizing iron or zinc reductions face increasing scrutiny and potential shutdowns due to effluent limits. By adopting this cleaner catalytic technology, manufacturers future-proof their operations against tightening environmental legislation. The starting materials, such as substituted nitrobenzenes and dicarbonyl compounds, are commodity chemicals with robust global supply networks, reducing the risk of raw material shortages. The mild reaction conditions also reduce wear and tear on reactor vessels and ancillary equipment, leading to higher asset availability and fewer unplanned maintenance downtimes. This reliability is crucial for maintaining long-term contracts with major agrochemical companies.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat transfer and mixing, but this exothermic hydrogenation is well-suited for large-scale reactors. The use of common solvents like toluene and the ability to run at near-atmospheric pressure removes the need for specialized high-pressure autoclaves, lowering capital expenditure for capacity expansion. From an environmental standpoint, the process aligns with green chemistry principles by maximizing atom economy and minimizing hazardous waste. The catalyst can be recovered and reused multiple times without significant loss of activity, further reducing the consumption of precious metals. This sustainability profile enhances the corporate image of the manufacturer and facilitates easier permitting for plant expansions, ensuring long-term supply chain continuity for critical intermediates.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this patented process is essential for stakeholders evaluating its integration into existing production lines. The following questions address common concerns regarding reaction safety, impurity control, and raw material flexibility. These answers are derived directly from the experimental data and claims within CN1286832C, providing a factual basis for decision-making. Whether you are assessing the feasibility of switching from a legacy process or validating the quality of incoming materials, these insights clarify the operational realities of implementing this advanced synthetic route.
Q: How does this process improve upon traditional iron or zinc reduction methods?
A: Traditional methods using iron or zinc generate massive amounts of heavy metal oxide waste, creating significant environmental disposal costs. This patented catalytic hydrogenation method eliminates solid metal waste, uses recoverable palladium catalysts, and operates under milder conditions, drastically reducing the environmental footprint and downstream purification complexity.
Q: What is the key mechanism preventing over-reduction to indolines?
A: The process utilizes an acylating agent (like acetic anhydride) and a base co-existing during hydrogenation. This traps the reactive 1-hydroxyindole intermediate as a stable 1-acyloxyindole, preventing its tautomerization to the N-oxide form which is prone to over-reduction. This ensures high selectivity for the desired indole product.
Q: Is this method suitable for large-scale commercial production?
A: Yes, the reaction operates at moderate temperatures (around 50°C) and atmospheric to low pressure, which are ideal parameters for industrial scale-up. The use of common solvents like toluene and the ability to recover the heterogeneous catalyst further enhance its viability for manufacturing hundreds of kilograms to metric tons annually.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Fluoro-2-Methylindole Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to advanced synthetic methodologies requires a partner with deep technical expertise and proven manufacturing capabilities. As a leading CDMO, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of patent CN1286832C can be realized at an industrial level. Our facilities are equipped with state-of-the-art hydrogenation reactors and rigorous QC labs capable of detecting trace impurities down to ppm levels. We understand that consistency is key in the supply of pharmaceutical intermediates, and our stringent purity specifications guarantee that every batch meets the demanding requirements of global regulatory bodies. By leveraging our expertise in catalytic processes, we can help you secure a stable supply of high-quality indole derivatives while optimizing your overall production costs.
We invite you to collaborate with us to explore how this technology can enhance your product portfolio. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact our technical procurement team to request specific COA data and route feasibility assessments. Together, we can drive innovation in the agrochemical and pharmaceutical sectors, ensuring that your supply chain is both resilient and economically competitive in a rapidly evolving market.
