Advanced Synthesis of 2-Hydroxynaphthalene-1-Boric Acid for Commercial Scale Production
The landscape of fine chemical manufacturing for advanced intermediates is constantly evolving, driven by the need for higher purity and regioselectivity in complex molecular architectures. A pivotal advancement in this domain is detailed in patent CN109305981B, which discloses a robust method for synthesizing 2-hydroxynaphthalene-1-boric acid. This compound serves as a critical building block for high-performance OLED materials and sophisticated pharmaceutical agents, where structural precision is non-negotiable. Historically, the preparation of this specific isomer has been plagued by instability and poor regiocontrol, often yielding the thermodynamically favored but undesired 3-isomer. The disclosed technology overcomes these hurdles through a strategic four-step sequence involving bromine positioning, hydroxyl protection, boration, and careful demethylation. For R&D directors and procurement specialists seeking a reliable pharmaceutical intermediate supplier, understanding this mechanistic breakthrough is essential for securing supply chains for next-generation electronic and medicinal compounds.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies for synthesizing naphthalene-based boronic acids often relied on direct boration strategies that proved fundamentally flawed for the 1-substituted isomer. As illustrated in the reaction pathway below, attempting to boronate 2-naphthol directly typically results in substitution at the 3-position due to electronic activation differences across the naphthalene ring system. This lack of regioselectivity means that even with optimized catalysts, the detected product is predominantly 2-hydroxynaphthalene-3-boric acid with concentrations exceeding 90 percent, failing to meet the stringent specifications required for OLED or API synthesis. Furthermore, conventional hydrolysis deprotection steps often occur under acidic conditions where the target 1-boric acid moiety is highly unstable, leading to rapid deboronation and reversion to the starting 2-naphthol material. This inherent instability in traditional routes results in dismal yields, often less than 5 percent, making commercial scale-up economically unviable and technically frustrating for process chemists.
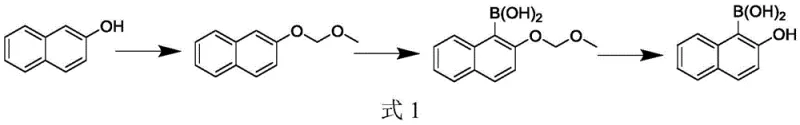
The Novel Approach
To circumvent these thermodynamic and kinetic barriers, the patented process introduces a bromine-blocking strategy that fundamentally alters the reaction trajectory. By initially installing a bromine atom at the 1-position of 2-naphthol, the synthesis effectively locks the substitution site, ensuring that subsequent boration occurs exclusively at the desired location. This approach, depicted in the following scheme, replaces the problematic direct etherification and hydrolysis sequence with a more stable alkylation protection and low-temperature demethylation protocol. The use of a bromine directing group not only guarantees regioselectivity but also facilitates the lithium-halogen exchange necessary for introducing the boron functionality with high precision. This strategic pivot from direct functionalization to a protected, blocked intermediate route transforms an unreliable laboratory curiosity into a commercially viable manufacturing process capable of delivering high-purity materials consistently.
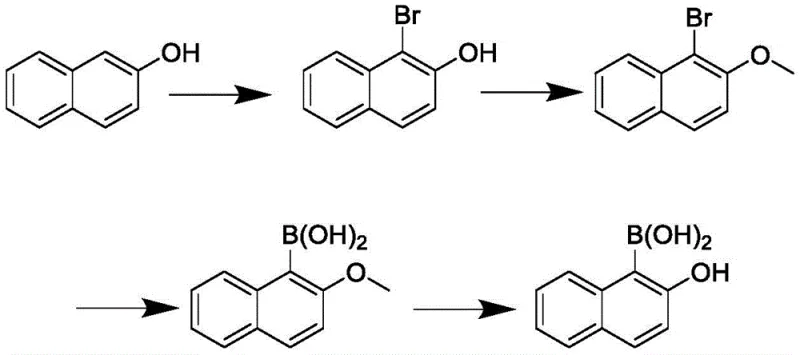
Mechanistic Insights into Regioselective Bromination and Cryogenic Boration
The success of this synthesis hinges on the precise control of reaction conditions, particularly during the initial bromination and the subsequent lithiation steps. In the first stage, 2-naphthol is treated with a brominating reagent such as N-bromosuccinimide (NBS) or dibromohydantoin at a strictly controlled temperature range of 0-5°C. This low-temperature environment is critical for kinetic control, preventing poly-bromination and ensuring that the electrophilic substitution occurs selectively at the 1-position ortho to the hydroxyl group. Following this, the hydroxyl group is protected via alkylation using sodium hydride and dimethyl sulfate, converting the acidic phenol into a stable methyl ether. This protection is vital because free phenolic hydroxyls can interfere with the strong bases used in the next step and contribute to the instability observed in acidic deprotection phases of older methods. The resulting 1-bromo-2-methoxynaphthalene serves as a robust platform for the introduction of the boron moiety.
The core transformation involves a halogen-lithium exchange followed by quenching with a borate ester, a process that demands rigorous exclusion of moisture and precise thermal management. The intermediate 1-bromo-2-methoxynaphthalene is subjected to n-butyllithium at cryogenic temperatures between -65°C and -60°C to generate the corresponding aryl lithium species. This extreme cold is necessary to suppress side reactions such as nucleophilic attack on the methoxy group or decomposition of the organolithium intermediate. Upon formation, the aryl lithium is immediately trapped with triisopropyl borate to form the boronic acid ester, which is subsequently hydrolyzed to the free acid. The final demethylation step utilizes boron tribromide at even lower temperatures, specifically maintained at or below -70°C. This ultra-low temperature protocol is the key innovation that prevents the acid-catalyzed deboronation seen in prior art, allowing the cleavage of the methyl ether to reveal the final 2-hydroxynaphthalene-1-boric acid with a purity greater than or equal to 99.5 percent.
How to Synthesize 2-Hydroxynaphthalene-1-Boronic Acid Efficiently
Implementing this synthesis route requires adherence to strict operational parameters to maximize yield and safety, particularly regarding the handling of pyrophoric reagents and cryogenic conditions. The process begins with the dissolution of 2-naphthol in a polar aprotic solvent like DMF, followed by the controlled addition of the brominating agent. Once the 1-bromo intermediate is secured and protected as the methyl ether, the reaction shifts to an inert atmosphere setup using THF and toluene for the boration step. The critical phase involves the dropwise addition of n-butyllithium while maintaining the internal reactor temperature below -60°C, followed by the addition of the borate ester. Finally, the demethylation is performed in dichloromethane with boron tribromide, requiring efficient heat exchange systems to maintain the -70°C setpoint. For a detailed breakdown of the specific molar ratios, solvent volumes, and workup procedures, please refer to the standardized protocol below.
- Bromination of 2-naphthol at the 1-position using NBS at 0-5°C to ensure regioselectivity.
- Protection of the hydroxyl group via alkylation with dimethyl sulfate and sodium hydride.
- Lithium-halogen exchange followed by quenching with triisopropyl borate at cryogenic temperatures.
- Demethylation using boron tribromide at -70°C to reveal the final hydroxyl group without deboronation.
Commercial Advantages for Procurement and Supply Chain Teams
From a supply chain perspective, this patented methodology offers significant advantages over legacy production methods, primarily driven by the drastic improvement in process reliability and raw material efficiency. Traditional routes suffered from unpredictable yields and difficult purification profiles due to the formation of isomeric impurities, which often necessitated costly chromatographic separations or multiple recrystallizations. By contrast, this new route leverages cheap and readily available starting materials like 2-naphthol and NBS, avoiding the need for exotic or prohibitively expensive catalysts. The high atom economy of the bromination step, which achieves conversion rates nearing 99 percent, ensures that raw material costs are minimized while waste generation is kept to a manageable level. For procurement managers focused on cost reduction in electronic chemical manufacturing, the ability to source this intermediate from a process that eliminates complex isomer separation translates directly into lower unit costs and more stable pricing structures.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of stoichiometric reagents that are commodity chemicals significantly lowers the bill of materials. Furthermore, the high selectivity of the bromine-blocking strategy removes the need for expensive preparative HPLC or extensive recycling of off-spec isomers, streamlining the production workflow. The process utilizes standard industrial solvents such as toluene, DMF, and DCM, which are easily recovered and recycled, further enhancing the economic viability of large-scale production runs.
- Enhanced Supply Chain Reliability: The robustness of the four-step sequence ensures consistent batch-to-batch quality, reducing the risk of supply disruptions caused by failed batches or out-of-specification products. The final product purity of greater than 99.5 percent meets the rigorous standards required for downstream applications in OLED fabrication and pharmaceutical synthesis without requiring additional tolling or purification services. This reliability allows supply chain heads to reduce safety stock levels and optimize inventory turnover, knowing that the commercial scale-up of complex pharmaceutical intermediates is supported by a chemically sound and reproducible process.
- Scalability and Environmental Compliance: The reaction conditions, while requiring low temperatures, are well within the capabilities of standard jacketed reactors found in modern multipurpose chemical plants. The workup procedures involve straightforward aqueous extractions and filtrations, avoiding the generation of heavy metal waste streams associated with palladium-catalyzed boration methods. This aligns with increasingly stringent environmental regulations and simplifies the permitting process for manufacturing sites, ensuring long-term reducing lead time for high-purity intermediates by avoiding regulatory bottlenecks associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-hydroxynaphthalene-1-boric acid. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering transparency into the process capabilities. Understanding these details helps stakeholders assess the feasibility of integrating this intermediate into their own value chains.
Q: Why is direct boration of 2-naphthol ineffective for producing the 1-isomer?
A: Direct boration fails because the energy levels at different sites of 2-naphthol differ, causing the boronic acid group to preferentially substitute at the 3-position rather than the desired 1-position, resulting in the wrong isomer.
Q: How does this patent ensure the stability of the final product during deprotection?
A: The process utilizes a specific demethylation protocol with boron tribromide at strictly controlled low temperatures (-70°C) to prevent the target product from undergoing deboronation and reverting to raw material under acidic conditions.
Q: What is the achieved purity and yield of this novel synthesis method?
A: The invention achieves a single target product with high purity greater than or equal to 99.5 percent and a final yield greater than or equal to 30 percent, significantly outperforming prior art methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Hydroxynaphthalene-1-Boronic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires more than just chemical knowledge; it demands engineering excellence and unwavering commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the intricate low-temperature steps of this synthesis are managed with precision and safety. We understand that for applications in OLEDs and advanced therapeutics, stringency is paramount; therefore, our facilities operate with stringent purity specifications and are equipped with rigorous QC labs capable of detecting trace impurities that could compromise device performance or drug efficacy. We are prepared to deploy this optimized bromine-positioning technology to secure your supply of this critical building block.
We invite you to engage with our technical team to discuss how this advanced synthesis route can be tailored to your specific volume requirements and cost targets. By partnering with us, you gain access to a Customized Cost-Saving Analysis that evaluates the total landed cost benefits of switching to this high-yield process. We encourage potential partners to contact our technical procurement team to request specific COA data from our pilot batches and to receive comprehensive route feasibility assessments that demonstrate our capability to be your long-term strategic partner in fine chemical manufacturing.
