Advanced Catalytic Route for Hexamethylindanol Production Enhancing Purity and Scalability
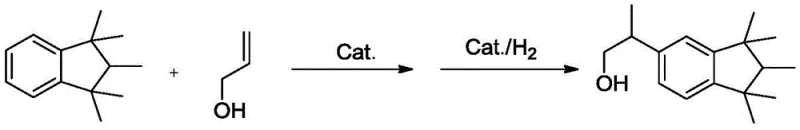
The chemical landscape for synthesizing high-value fragrance intermediates is undergoing a significant transformation, driven by the urgent need for safer, more sustainable, and economically viable production methods. A pivotal advancement in this domain is detailed in Chinese patent CN111170829B, which discloses a novel preparation method for hexamethylindanol, a critical precursor in the synthesis of Galaxolide musk. This patent introduces a sophisticated catalytic system that fundamentally alters the reaction pathway, moving away from hazardous traditional reagents toward a streamlined, hydrogen-mediated coupling and reduction process. By leveraging a specialized supported Ruthenium (Ru) catalyst, this technology addresses long-standing industry pain points related to safety, environmental compliance, and process efficiency. For global procurement and R&D teams, understanding the nuances of this patented route is essential for securing a competitive edge in the fragrance supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of hexamethylindanol has relied heavily on the Friedel-Crafts alkylation of 1,1,2,3,3-pentamethylindan with propylene oxide, typically catalyzed by aluminum trichloride (AlCl3). This legacy approach is fraught with substantial operational and safety liabilities that modern manufacturing facilities strive to eliminate. Firstly, the use of propylene oxide presents a severe safety hazard due to its extremely low flash point of -37°C, creating a high risk of fire and explosion during storage and handling. Furthermore, the reliance on AlCl3 generates significant quantities of corrosive waste acid during the post-reaction workup, necessitating complex and costly neutralization and disposal procedures that burden environmental compliance budgets. Additionally, conventional methods often require the use of excessive amounts of pentamethylindan acting as both reactant and solvent, or the use of toxic solvents like chlorobenzene, which complicates product isolation and increases raw material costs significantly.
The Novel Approach
In stark contrast, the methodology outlined in patent CN111170829B offers a paradigm shift by utilizing allyl alcohol as the alkylating agent in conjunction with a robust supported Ru catalyst. This innovative route effectively bypasses the need for hazardous propylene oxide and corrosive Lewis acids, thereby drastically reducing the safety footprint of the manufacturing process. The reaction proceeds through a tandem coupling and hydrogenation sequence under relatively mild conditions, typically between 40-70°C, which lowers energy consumption compared to harsher traditional protocols. Crucially, the use of a heterogeneous supported catalyst facilitates straightforward separation via filtration, eliminating the need for extensive aqueous workups to remove homogeneous catalyst residues. This not only simplifies the operational workflow but also enhances the purity profile of the final hexamethylindanol product, making it ideally suited for high-end fragrance applications where odor quality is paramount.
Mechanistic Insights into Supported Ru-Catalyzed Coupling and Reduction
The core of this technological breakthrough lies in the intricate design of the supported Ru catalyst, denoted as Ru-X-Y/Z. In this system, Ruthenium serves as the active metal center, while X represents a promoter such as Copper, Silver, or Zinc, and Y denotes a sophisticated phosphine ligand like BINAP or dppe. The carrier Z, which can range from carbon nanofiber membranes to molecular sieves, plays a critical role in stabilizing the active species and preventing leaching. The mechanistic advantage stems from the synergistic interaction between the metal center and the ligand environment; specifically, the lone pair electrons on the phosphorus atoms of the ligands form coordination bonds with the Ruthenium. This electronic modulation enhances the catalyst's ability to interact with the double bonds of the allyl alcohol substrate, effectively increasing the local concentration of reactants on the catalyst surface.
This enhanced surface interaction promotes the initial coupling reaction followed immediately by the in-situ reduction under a hydrogen atmosphere, typically maintained at 5-8 barg. The presence of the promoter X further fine-tunes the electronic properties of the active sites, effectively suppressing the formation of unwanted by-products such as pentamethylindanol isomers. This precise control over the reaction trajectory results in exceptional selectivity, with patent data indicating selectivity rates consistently exceeding 90%. For R&D directors, this level of control translates to a cleaner impurity profile, reducing the burden on downstream purification units and ensuring a consistent, high-quality output that meets stringent international fragrance standards.
How to Synthesize Hexamethylindanol Efficiently
Implementing this advanced synthesis route requires careful attention to catalyst preparation and reaction parameters to maximize yield and longevity. The process begins with the meticulous preparation of the supported catalyst, involving the mixing of Ru precursors with ligands and promoters in an aqueous medium, followed by precipitation and high-temperature calcination to activate the surface. Once the catalyst is prepared, the synthesis involves reacting 1,1,2,3,3-pentamethylindan with allyl alcohol at a molar ratio ranging from 1:1.1 to 1:2. The reaction is initiated at moderate temperatures before introducing hydrogen to drive the reduction to completion. The detailed standardized synthesis steps, including specific calcination temperatures and aging times required to reproduce the high-performance catalyst described in the patent, are outlined below.
- Prepare the supported Ru catalyst by mixing Ru precursors, ligands (like BINAP), and promoters in water, followed by precipitation and calcination.
- React 1,1,2,3,3-pentamethylindan with allyl alcohol in the presence of the catalyst at 40-70°C for 4-6 hours.
- Introduce hydrogen gas to 5-8 barg pressure for 3-8 hours to complete the reduction, followed by continuous rectification to isolate the product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this Ru-catalyzed process represents a strategic opportunity to optimize total cost of ownership and mitigate supply risks. The elimination of hazardous raw materials like propylene oxide not only reduces insurance and safety compliance costs but also simplifies logistics, as allyl alcohol is a widely available commodity chemical with a more manageable safety profile. Furthermore, the heterogeneous nature of the catalyst means it can be recovered and potentially reused, significantly lowering the recurring cost of catalytic materials compared to single-use homogeneous systems. The simplified post-processing, which avoids the generation of large volumes of waste acid, leads to substantial reductions in wastewater treatment expenses and environmental fees, directly impacting the bottom line.
- Cost Reduction in Manufacturing: The transition to this novel catalytic system eliminates the need for expensive solvent recovery processes associated with using excess pentamethylindan as a solvent. By operating without corrosive aluminum trichloride, the process significantly reduces equipment maintenance costs and extends the lifespan of reactor vessels, as there is no acidic corrosion to manage. Additionally, the high selectivity of the reaction minimizes the formation of by-products, which means less raw material is wasted on non-target compounds, thereby improving the overall atom economy and reducing the effective cost per kilogram of the final hexamethylindanol product.
- Enhanced Supply Chain Reliability: Relying on a robust supported catalyst that does not leach active metals ensures consistent batch-to-batch performance, reducing the risk of production delays caused by catalyst deactivation or failure. The use of commercially available and stable reagents like allyl alcohol ensures that the supply chain is not vulnerable to the volatility often seen with specialized or hazardous epoxides. This stability allows for more accurate production planning and inventory management, ensuring that downstream customers receive their orders on time without unexpected interruptions due to raw material shortages or safety shutdowns.
- Scalability and Environmental Compliance: The process is inherently designed for scalability, utilizing continuous rectification for product separation which is easily adaptable from pilot scale to multi-ton commercial production. The absence of heavy metal waste streams and corrosive acids aligns perfectly with increasingly strict global environmental regulations, future-proofing the manufacturing site against regulatory changes. This green chemistry approach not only enhances the corporate sustainability profile but also streamlines the permitting process for capacity expansion, as the environmental impact assessment for this cleaner technology is significantly more favorable than for traditional methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented hexamethylindanol synthesis method. These answers are derived directly from the experimental data and technical specifications provided in the source patent documentation, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing production lines or for sourcing high-quality intermediates from suppliers who utilize this advanced methodology.
Q: How does the new Ru-catalyzed process improve safety compared to traditional methods?
A: The new process eliminates the use of propylene oxide, which has a dangerously low flash point of -37°C, and replaces corrosive aluminum trichloride with an environmentally friendly supported catalyst.
Q: What represents the key advantage of the supported Ru catalyst regarding product separation?
A: The heterogeneous nature of the supported catalyst allows for easy filtration and separation from the reaction mixture, preventing metal contamination and simplifying downstream purification.
Q: What conversion and selectivity rates can be achieved with this method?
A: According to patent data, the process achieves raw material conversion rates exceeding 90% and product selectivity above 90%, ensuring high efficiency and reduced waste.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Hexamethylindanol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting cutting-edge synthetic routes to maintain competitiveness in the global fragrance market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering high-purity hexamethylindanol that adheres to stringent purity specifications, supported by our rigorous QC labs which utilize state-of-the-art analytical instrumentation to verify every batch. Our capability to implement complex catalytic systems, such as the supported Ru technology described herein, positions us as a strategic partner capable of meeting the most demanding quality and volume requirements.
We invite you to engage with our technical procurement team to discuss how this advanced manufacturing route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the potential economic efficiencies achievable through this technology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project, ensuring that your fragrance formulations are built on a foundation of quality, safety, and reliability.
