Scalable Asymmetric Oxidation for High-Purity Neurokinin Receptor Antagonist Intermediates
Scalable Asymmetric Oxidation for High-Purity Neurokinin Receptor Antagonist Intermediates
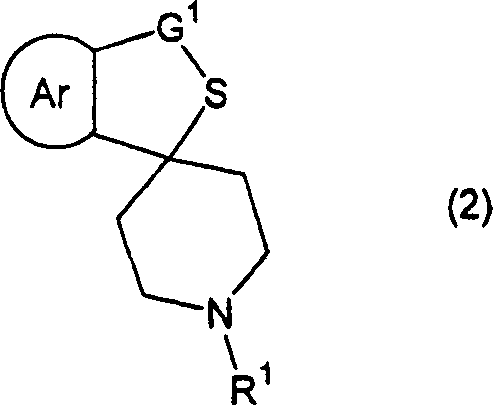
The pharmaceutical industry constantly seeks robust pathways for synthesizing chiral intermediates, particularly for complex targets like neurokinin receptor antagonists. Patent CN1729196A introduces a transformative methodology for the preparation of optically active cyclic sulfoxides, which serve as critical building blocks in this therapeutic class. Unlike traditional stoichiometric approaches that rely on expensive chiral oxidants or inefficient resolution techniques, this invention leverages a sophisticated titanium-tartrate catalytic system. By utilizing cumene hydroperoxide as the terminal oxidant in the presence of a chiral titanium(IV) alkoxide complex, the process achieves exceptional stereocontrol. This breakthrough not only addresses the economic limitations of prior art but also establishes a scalable framework for commercial scale-up of complex pharmaceutical intermediates. The technology represents a significant leap forward in asymmetric synthesis, enabling manufacturers to produce high-value chiral sulfoxides with improved atom economy and reduced environmental impact.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of enantiomerically enriched cyclic sulfoxides was plagued by significant economic and operational inefficiencies. One prevalent method involved the use of stoichiometric amounts of chiral oxidants, such as Davis oxaziridines. While these reagents could deliver high enantiomeric excess, their prohibitive cost and the difficulty in recovering the stoichiometric byproducts rendered them unsuitable for large-scale industrial production. Alternatively, manufacturers relied on the optical resolution of racemic sulfoxides. This classical approach inherently caps the maximum theoretical yield at 50%, as the undesired enantiomer must be discarded or subjected to costly recycling processes. Furthermore, alternative catalytic methods using tert-butyl hydroperoxide often resulted in disappointingly low yields (20-46%) and poor enantioselectivity (17-54% ee), failing to meet the stringent purity requirements of modern drug development. These bottlenecks created a pressing need for a more efficient, cost-effective solution.
The Novel Approach
The methodology disclosed in CN1729196A fundamentally reshapes the production landscape by introducing a highly efficient catalytic asymmetric oxidation. By employing a complex formed from optically active tartrate diesters and titanium(IV) alkoxides, the process activates cumene hydroperoxide for stereoselective oxygen transfer. This novel approach delivers cyclic sulfoxides in outstanding yields ranging from 90% to 95%, coupled with high enantiomeric excess values exceeding 87%. Crucially, the reaction conditions are optimized to minimize over-oxidation to the corresponding sulfone, a common side reaction that compromises purity. The use of readily available reagents like titanium tetraisopropoxide and diethyl or diisopropyl tartrate drastically lowers the raw material costs compared to stoichiometric chiral oxidants. This shift enables cost reduction in pharmaceutical intermediates manufacturing by maximizing material throughput and eliminating the 50% yield loss associated with racemic resolution, thereby establishing a new standard for industrial viability.
Mechanistic Insights into Titanium-Tartrate Catalyzed Asymmetric Oxidation
The core of this technology lies in the precise assembly of the chiral catalyst species. The reaction initiates with the formation of a coordination complex between the titanium(IV) alkoxide and the chiral tartrate diester. Critical to the success of this system is the controlled addition of water and alcohol, which modifies the coordination sphere of the titanium center, enhancing both its Lewis acidity and its ability to bind the oxidant. The resulting chiral environment dictates the facial selectivity of the oxygen transfer from the hydroperoxide to the sulfur atom of the cyclic sulfide substrate. This mechanism ensures that the oxygen atom is delivered to a specific face of the planar sulfide, generating the desired sulfoxide enantiomer with high fidelity. The choice of cumene hydroperoxide over other peroxides is mechanistically significant, as its specific steric and electronic properties facilitate a smoother transition state within the chiral pocket, leading to superior reaction rates and selectivity compared to tert-butyl hydroperoxide systems.
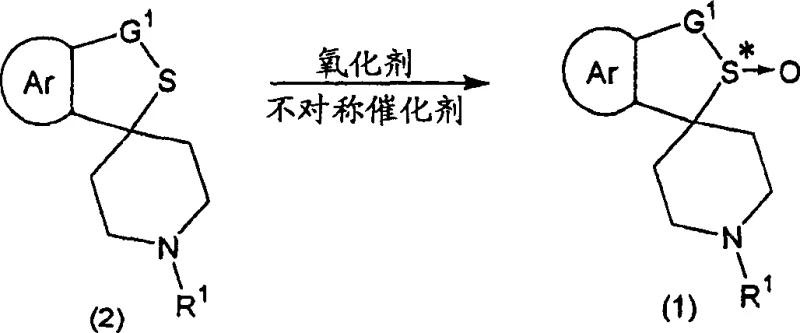
Impurity control is another vital aspect of this mechanistic design. A major challenge in sulfide oxidation is preventing the formation of the sulfone byproduct, which occurs if the sulfoxide undergoes a second oxidation. The patent data indicates that by carefully controlling the stoichiometry of the oxidant (preferably 1.0 to 1.5 equivalents) and maintaining low reaction temperatures (typically -20°C to -5°C), the formation of sulfone impurities is suppressed to minimal levels (often below 6%). Furthermore, the process allows for subsequent purification steps, such as recrystallization or salt formation with chiral acids like mandelic acid, which can upgrade the enantiomeric excess to >99%. This robust impurity profile is essential for meeting the rigorous quality standards required for high-purity pharmaceutical intermediates, ensuring that downstream synthesis of the final neurokinin antagonist proceeds without contamination from difficult-to-remove oxidative byproducts.
How to Synthesize Optically Active Cyclic Sulfoxides Efficiently
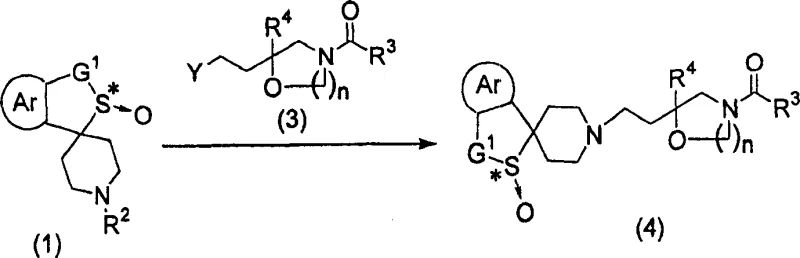
Implementing this catalytic oxidation requires strict adherence to moisture control and temperature management to ensure reproducibility and high stereochemical outcomes. The process begins with the preparation of the catalyst solution under an inert atmosphere, followed by the sequential addition of substrates and oxidants at cryogenic temperatures. The specific molar ratios of titanium, tartrate, and water are critical parameters that influence the catalyst's active species distribution. For a comprehensive understanding of the operational parameters, including solvent choices like chlorobenzene or o-dichlorobenzene and workup procedures involving sodium metabisulfite quenching, operators should refer to the standardized protocols derived from the patent examples. The following guide outlines the critical phases of this synthesis, ensuring that technical teams can replicate the high yields and selectivity demonstrated in the intellectual property.
- Prepare the chiral catalyst by mixing optically active tartrate diester (e.g., diisopropyl tartrate) with titanium(IV) alkoxide in an inert solvent like chlorobenzene.
- Add alcohol and controlled amounts of water to the catalyst mixture to activate the complex, then introduce the cyclic sulfide substrate at low temperature.
- Slowly add the oxidant (cumene hydroperoxide) while maintaining temperatures between -20°C and -5°C to ensure high enantioselectivity and minimize over-oxidation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement and supply chain leaders, the adoption of this catalytic technology offers substantial strategic benefits beyond mere technical performance. The shift from stoichiometric chiral reagents to a catalytic system fundamentally alters the cost structure of the synthesis. By eliminating the need for expensive, single-use chiral oxidants, the direct material costs are significantly reduced. Additionally, the high yield of the reaction means that less starting material is required to produce the same amount of product, further driving down the cost of goods sold. The use of commodity chemicals like titanium alkoxides and tartrates ensures a stable and reliable supply chain, mitigating the risks associated with sourcing specialized reagents from limited vendors. This stability is crucial for maintaining continuous production schedules and avoiding delays caused by raw material shortages.
- Cost Reduction in Manufacturing: The transition to a catalytic process eliminates the massive waste inherent in resolution strategies, where half the material is discarded. This improvement in atom economy translates directly to lower waste disposal costs and higher overall process efficiency. Furthermore, the avoidance of precious metal catalysts or exotic chiral auxiliaries simplifies the purification workflow, reducing the consumption of solvents and chromatography media. These cumulative efficiencies result in a leaner manufacturing process that is economically superior to legacy methods, providing a competitive edge in pricing for the final API.
- Enhanced Supply Chain Reliability: The reagents utilized in this process, such as cumene hydroperoxide and titanium tetraisopropoxide, are widely produced industrial chemicals with established global supply networks. This abundance ensures that manufacturers are not vulnerable to the supply disruptions that often plague niche fine chemical suppliers. The robustness of the reaction conditions also allows for flexibility in sourcing, as slight variations in reagent grades can often be accommodated without compromising the final product quality. This reliability is essential for long-term supply agreements and ensures that pharmaceutical partners can depend on consistent delivery timelines.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing solvents and conditions that are amenable to large-scale reactor operations. The ability to run the reaction at relatively low catalyst loadings reduces the heavy metal burden in the waste stream, simplifying effluent treatment and ensuring compliance with increasingly stringent environmental regulations. The high selectivity of the reaction minimizes the generation of hazardous byproducts, aligning with green chemistry principles. This environmental compatibility not only reduces regulatory risk but also enhances the sustainability profile of the manufacturing site, a key factor for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric oxidation technology. These insights are derived directly from the experimental data and claims presented in the patent documentation, providing clarity on the practical aspects of the synthesis. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing capabilities. The answers highlight the versatility of the method across different substrates and the specific conditions required to maximize performance.
Q: What are the advantages of this catalytic method over traditional resolution?
A: Traditional resolution methods often discard 50% of the material (the unwanted enantiomer), leading to low theoretical yields. This catalytic asymmetric oxidation achieves yields of 90-95% with high enantiomeric excess, significantly improving material efficiency.
Q: Which oxidants are compatible with this titanium-tartrate system?
A: The patent specifically identifies cumene hydroperoxide and p-isopropyl cumene peroxide as the preferred oxidants. These provide superior reactivity and selectivity compared to tert-butyl hydroperoxide in this specific catalytic environment.
Q: How is the optical purity further enhanced after the reaction?
A: While the reaction itself provides high ee (87-89%+), the optical purity can be upgraded to >99% through recrystallization or by forming diastereomeric salts with chiral acids like mandelic acid or camphor sulfonic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Optically Active Sulfoxide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality chiral intermediates in the development of next-generation therapeutics. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless. We are equipped with rigorous QC labs and adhere to stringent purity specifications to guarantee that every batch of optically active sulfoxide meets the exacting standards required for neurokinin antagonist synthesis. Our commitment to excellence extends beyond mere compliance; we actively optimize processes to enhance yield and reduce environmental impact, mirroring the efficiencies described in cutting-edge patents like CN1729196A.
We invite you to collaborate with us to leverage this advanced technology for your supply chain needs. Our experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our catalytic capabilities can lower your overall production costs. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us partner with you to secure a reliable, cost-effective, and high-quality supply of these vital pharmaceutical building blocks.
