Advanced Manufacturing of 1,2-Indane Carboxylic Acid Derivatives for Pharmaceutical Applications
Introduction to Advanced Indane Synthesis Technology
The pharmaceutical industry continuously seeks robust and scalable methodologies for constructing complex chiral scaffolds, particularly those serving as critical intermediates for potent therapeutic agents. Patent CN1084742C discloses a groundbreaking improved process for the preparation of aromatic ring-fused cyclopentane derivatives, specifically focusing on 1,2-indane carboxylic acids and cyclopentano[b]pyridine derivatives. These compounds are of immense value as endothelin receptor antagonists, a class of drugs pivotal in treating cardiovascular and renal disorders. The core innovation lies in the strategic application of chiral auxiliaries to manage the stereochemical complexity of molecules possessing three distinct chiral centers, thereby overcoming the significant efficiency bottlenecks inherent in earlier synthetic routes. By leveraging this technology, manufacturers can achieve superior control over optical purity while streamlining the production workflow for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the advancements detailed in this patent, the synthesis of target indane carboxylic acids, such as (+)(1S,2R,3S)-3-[2-(2-hydroxyethyl-1-yloxy)-4-methoxyphenyl]-1-(3,4-methylenedioxyphenyl)-5-(prop-1-yloxy)-1,2-indane-2-carboxylic acid, was plagued by inefficiency. Historical methods, such as those reported in international publication WO94/25013, relied heavily on multi-step sequences that culminated in dismal overall yields, often ranging from merely 2% to 6% when excluding the racemic separation processes. The presence of three chiral centers in the target molecule necessitated cumbersome resolution steps, which not only discarded half of the synthesized material but also introduced significant costs related to solvent usage, purification, and waste disposal. Furthermore, the reliance on late-stage resolution often compromised the final optical purity, requiring additional recrystallization cycles that further eroded process economics and extended lead times for high-purity pharmaceutical intermediates.
The Novel Approach
In stark contrast, the novel approach outlined in CN1084742C introduces a highly efficient asymmetric synthesis strategy that fundamentally alters the production landscape. By employing a chiral auxiliary derived from readily available ephedrine, the process achieves high diastereoselectivity during the critical carbon-carbon bond-forming steps, effectively bypassing the need for inefficient racemic resolutions. This methodology utilizes a palladium-catalyzed coupling reaction to attach the chiral auxiliary, followed by a stereoselective Grignard addition mediated by a copper complex. The result is a streamlined pathway that significantly enhances the overall throughput and reduces the number of unit operations required. This shift from resolution-based to induction-based synthesis represents a paradigm shift in cost reduction in pharmaceutical intermediate manufacturing, offering a more sustainable and economically viable route to these complex bioactive molecules.
Mechanistic Insights into Chiral Auxiliary Directed Asymmetric Synthesis
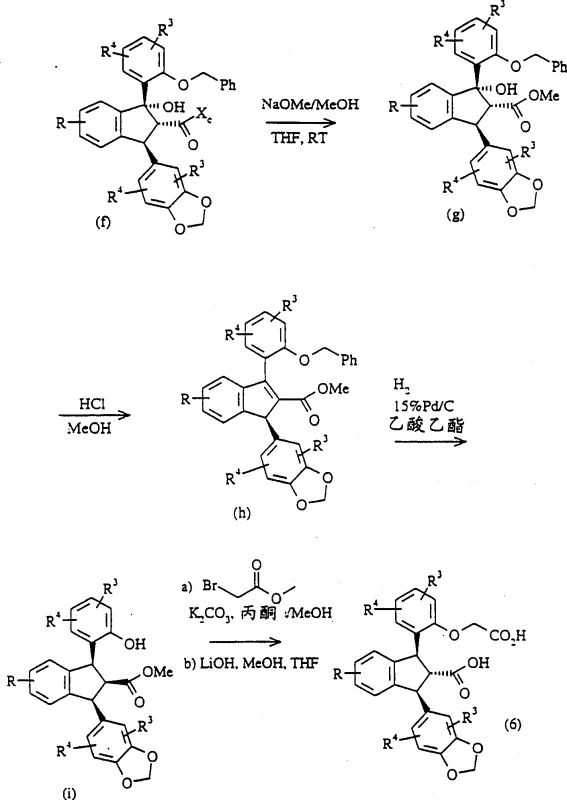
The heart of this technological breakthrough is the utilization of a specific chiral auxiliary, preferably a 4-substituted or 4,5-substituted 2-imidazolidinone derived from ephedrine, designated as compound (u) in the patent documentation. This auxiliary is not merely a passive spectator but an active director of stereochemistry. During the synthesis, the auxiliary is attached to the indane precursor via a palladium(II) acetate and tri-o-tolylphosphine catalyzed reaction. This step creates a rigid chiral environment around the reactive center. Subsequently, when the organometallic reagent, specifically a substituted 3,4-(methylenedioxy)phenylmagnesium bromide, is introduced in the presence of a copper(I) bromide-dimethyl sulfide complex, the bulky chiral auxiliary shields one face of the molecule. This steric hindrance forces the incoming nucleophile to attack from the less hindered face, ensuring the formation of the desired (1S, 2R, 3S) configuration with high fidelity. This precise control eliminates the formation of unwanted diastereomers, simplifying downstream purification.

Furthermore, the impurity profile of the final product is meticulously managed through this mechanistic design. Because the stereochemistry is established early in the sequence via the chiral auxiliary, subsequent steps such as the hydrogenation of benzyl protecting groups and the saponification of esters proceed without risking racemization at the sensitive chiral centers. The patent specifies conditions such as maintaining temperatures below 34°C during bromination and utilizing specific solvent systems like tetrahydrofuran for the Grignard coupling to minimize side reactions. The removal of the chiral auxiliary is achieved under mild hydrolytic conditions, often yielding the free acid directly or as a stable salt like the ethylenediamine salt. This robustness ensures that the final active pharmaceutical ingredient meets stringent purity specifications, a critical factor for regulatory approval and patient safety in the development of endothelin receptor antagonists.
How to Synthesize 1,2-Indane Carboxylic Acid Derivatives Efficiently
Executing this synthesis requires strict adherence to the optimized reaction parameters defined in the patent to ensure maximum yield and stereochemical integrity. The process begins with the activation of the benzoic acid starting material, typically involving bromination in dichloromethane followed by conversion to an acid chloride using thionyl chloride. This activated species is then coupled with the chiral auxiliary under inert atmosphere conditions to prevent catalyst deactivation. The subsequent Grignard addition must be performed at controlled low temperatures, often around -35°C to -10°C, to maintain diastereoselectivity. Following the coupling, the protecting groups are removed via catalytic hydrogenation, and the final ester hydrolysis is conducted using lithium hydroxide or sodium hydroxide depending on the desired salt form. For a comprehensive guide on the specific stoichiometry, temperature profiles, and workup procedures required to replicate this high-efficiency route, please refer to the standardized synthesis steps provided below.
- Preparation of the activated aryl halide intermediate via bromination and conversion to acid chloride.
- Coupling with a chiral imidazolidinone auxiliary using a palladium(II) acetate catalyst system to establish stereocontrol.
- Diastereoselective addition of a substituted phenylmagnesium bromide mediated by a copper(I) complex, followed by hydrolysis and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this improved synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The transition from a resolution-based process to an asymmetric induction process fundamentally alters the cost structure of the supply chain. By eliminating the discard of 50% of the material during racemic resolution, the effective capacity of existing manufacturing assets is nearly doubled without capital expenditure. This efficiency translates directly into substantial cost savings in pharmaceutical intermediate manufacturing, allowing for more competitive pricing models in a crowded therapeutic market. Additionally, the reliance on industrially available starting materials such as ephedrine, 3-hydroxybenzoic acid, and common solvents like acetonitrile and toluene ensures a resilient supply chain that is less susceptible to the volatility of exotic reagent markets.
- Cost Reduction in Manufacturing: The elimination of expensive and wasteful resolution steps significantly lowers the cost of goods sold (COGS). By achieving high diastereoselectivity upfront, the process reduces the burden on purification units, lowering solvent consumption and energy costs associated with repeated crystallizations. The use of catalytic amounts of palladium and copper, which can potentially be recovered, further optimizes the raw material spend. This lean manufacturing approach ensures that the production of complex indane derivatives remains economically viable even at large commercial scales, providing a distinct margin advantage over competitors relying on legacy technologies.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply continuity by reducing the risk of batch failures due to poor optical purity. The use of stable intermediates and well-defined reaction conditions minimizes process variability, ensuring consistent delivery schedules. Furthermore, the ability to produce different salt forms, such as the disodium salt or the ethylenediamine salt, from a common intermediate provides flexibility in meeting diverse customer formulation requirements. This adaptability reduces lead time for high-purity pharmaceutical intermediates, allowing partners to respond swiftly to market demands and clinical trial timelines without compromising on quality or compliance standards.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as filtration, distillation, and crystallization that are easily transferable from pilot plant to full-scale production. The avoidance of hazardous reagents and the optimization of solvent recovery systems align with modern green chemistry principles, reducing the environmental footprint of the manufacturing process. This commitment to environmental compliance not only mitigates regulatory risks but also appeals to increasingly eco-conscious stakeholders. The streamlined waste profile facilitates easier disposal and treatment, ensuring that the commercial scale-up of complex pharmaceutical intermediates proceeds smoothly without encountering environmental bottlenecks.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthesis is crucial for stakeholders evaluating its implementation. The following questions address common inquiries regarding the practical application and benefits of the technology described in CN1084742C. These answers are derived directly from the experimental data and theoretical framework provided in the patent, ensuring accuracy and relevance for technical decision-makers. Whether you are concerned about the stability of the chiral auxiliary or the specifics of the salt formation, the information below provides a solid foundation for further discussion with our technical team.
Q: What is the primary advantage of the chiral auxiliary method described in CN1084742C?
A: The use of an ephedrine-derived imidazolidinone auxiliary allows for high diastereoselectivity during the carbon-carbon bond forming steps, avoiding the low yields associated with racemic resolution.
Q: How does this process improve upon the prior art methods like WO94/25013?
A: Prior art methods suffered from overall yields as low as 2-6% due to multiple resolution steps. This improved process utilizes asymmetric induction to significantly increase throughput and reduce waste.
Q: Is this synthesis scalable for commercial production?
A: Yes, the patent details examples performed on multi-gram to kilogram scales using standard industrial solvents like toluene, THF, and acetonitrile, indicating strong potential for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Indane Carboxylic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of life-saving medications. Our expertise in process chemistry allows us to master complex synthetic pathways like the one described in CN1084742C, ensuring that we can deliver materials that meet the rigorous demands of the pharmaceutical industry. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our state-of-the-art facilities to maintain stringent purity specifications throughout the manufacturing lifecycle. Our rigorous QC labs employ advanced analytical techniques to verify the stereochemical integrity and chemical purity of every batch, guaranteeing that our clients receive a reliable pharmaceutical intermediate supplier product that accelerates their drug development programs.
We invite global partners to collaborate with us to unlock the full potential of this advanced synthesis technology. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and project timelines. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized processes can enhance your supply chain efficiency. Contact us today to discuss how our manufacturing capabilities can support your next breakthrough in endothelin receptor antagonist therapy, ensuring a secure and cost-effective supply of these vital chemical building blocks.
