Scalable Production of Benzoxathiepine Derivatives for Cardiovascular API Manufacturing
Scalable Production of Benzoxathiepine Derivatives for Cardiovascular API Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for complex heterocyclic scaffolds, particularly those exhibiting potent cardiovascular activity. Patent CN1946708A introduces a groundbreaking synthetic methodology for preparing benzoxathiepine derivatives of general formula (1), which are critical intermediates for treating conditions such as stable and unstable angina, cardiac insufficiency, and arrhythmia. This innovation addresses significant historical challenges regarding reproducibility and safety that plagued earlier generation syntheses. By shifting from unstable aldehyde intermediates to stable amide precursors, the disclosed technology offers a transformative approach to manufacturing high-purity active pharmaceutical ingredients. The structural versatility of these compounds allows for extensive derivatization at the R1 through R6 positions, enabling the fine-tuning of pharmacological properties while maintaining a scalable production framework.
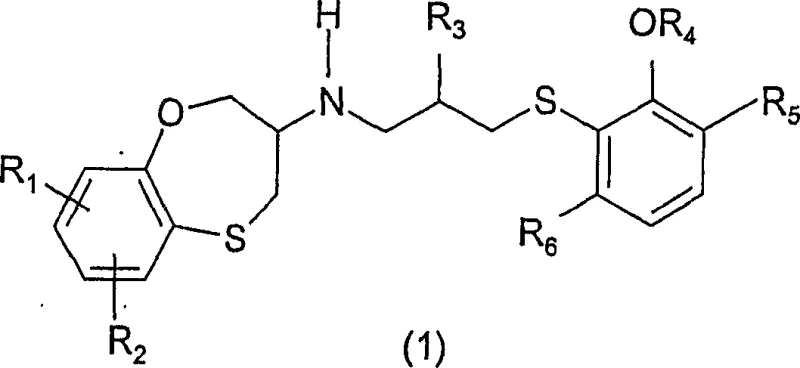
For procurement specialists and supply chain directors, the transition to this novel chemistry represents a strategic opportunity to secure a reliable pharmaceutical intermediate supplier capable of delivering consistent quality. The patent explicitly highlights the limitations of prior art methods, such as those described in WO 02/081464, which relied on hazardous reagents and non-reproducible low-temperature protocols. In contrast, the new process utilizes chemically and stereochemically stable compounds that can be purified via standard recrystallization techniques. This shift not only mitigates operational risks associated with explosive or malodorous reagents but also ensures a steady supply of key building blocks necessary for downstream API synthesis. Consequently, this technology serves as a cornerstone for cost reduction in API manufacturing by minimizing waste and maximizing throughput efficiency.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of benzoxathiepine derivatives relied heavily on reductive amination strategies involving highly unstable aldehyde intermediates. As detailed in the background of the invention, previous methods required the in situ generation of aldehydes at extremely low temperatures, often below -60°C, using reagents like oxalyl chloride. These conditions are technically demanding and pose significant safety hazards due to the potential explosiveness of certain reagents and the generation of toxic by-products like dimethyl sulfide. Furthermore, the instability of the aldehyde intermediates led to widely varying chemical yields and stereochemical purity, making the process virtually impossible to reproduce reliably on a semi-industrial or industrial scale. The reliance on Mitsunobu-type reactions in earlier routes also generated massive amounts of non-recyclable waste, such as triphenylphosphine oxide, creating severe environmental and disposal burdens that inflated production costs.
The Novel Approach
The innovative strategy presented in CN1946708A circumvents these pitfalls by employing a stable amide intermediate of formula (9) as the key precursor. Instead of struggling with fleeting aldehyde species, the new method couples a chiral amine with a chiral carboxylic acid or ester to form a robust amide bond. This amide intermediate is typically a crystalline solid that can be easily purified by recrystallization, ensuring high stereochemical integrity before the final reduction step. The subsequent reduction of the amide to the amine is performed under mild conditions using borane complexes, preserving the chiral centers established in the earlier steps. This fundamental change in synthetic logic transforms a fragile, laboratory-scale curiosity into a rugged, commercially viable process that guarantees consistent quality and safety profiles for large-scale manufacturing operations.
Mechanistic Insights into Amide Reduction and Chemoselective Cyclization
The core of this technological advancement lies in the strategic assembly of the molecule through two distinct chiral fragments that are joined via an amide linkage. The first fragment, the benzoxathiepine amine of formula (5), is synthesized through a novel pathway involving the reaction of a diazonium salt with L-cystine dimethyl ester. This reaction forms a sulfur-carbon bond efficiently, followed by reduction and chemoselective cyclization to close the seven-membered ring. This route avoids the use of malodorous thiophenols and expensive protecting group manipulations required in older methods. The second fragment, the side chain of formula (7) or (8), is constructed by displacing a mesylate group with an appropriate arylthiophenol, a reaction that proceeds with inversion of configuration to establish the desired stereochemistry. These two fragments are then coupled using either aluminum hydride derivatives or carbonyldiimidazole activation to form the stable amide of formula (9).
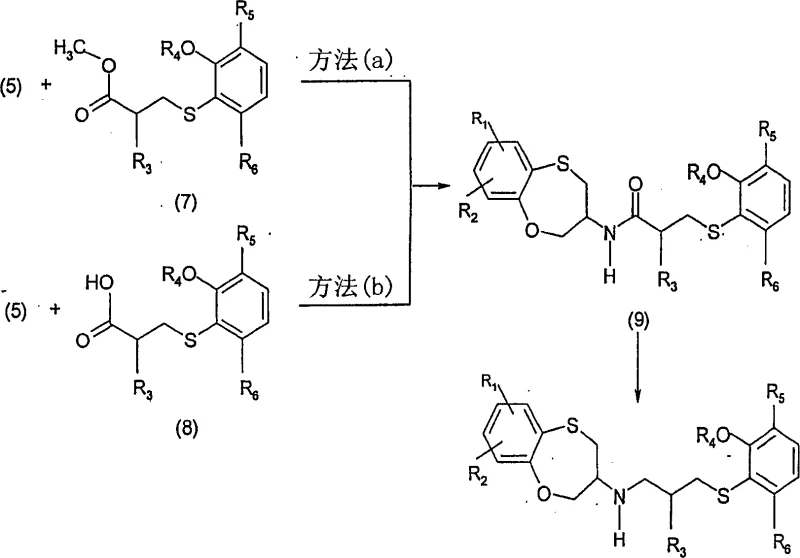
Following the formation of the amide intermediate, the final transformation involves the reduction of the carbonyl group to a methylene unit to yield the target amine of formula (1). This reduction is typically achieved using borane-THF complexes, which are highly selective for amides over other functional groups that might be present on the aromatic rings. The mechanistic elegance of this step ensures that the stereogenic centers at the C(3) position of the benzoxathiepine ring and the adjacent carbon in the side chain remain intact, preventing racemization. The ability to isolate and characterize the amide intermediate (9) provides a crucial quality control checkpoint, allowing manufacturers to verify purity before committing to the final reduction. This level of process control is essential for meeting the stringent regulatory requirements of the pharmaceutical industry and ensures that the final API intermediate possesses the necessary optical purity for therapeutic efficacy.
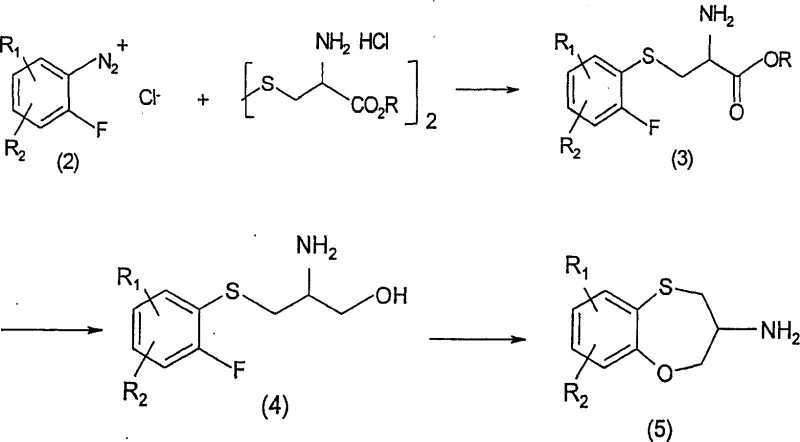
How to Synthesize Benzoxathiepine Derivatives Efficiently
The synthesis of these complex heterocyclic compounds requires precise control over reaction conditions and stereochemistry to ensure high yields and purity. The patented process outlines a logical sequence of transformations starting from readily available chiral pool materials. Detailed standardized synthetic steps for the preparation of the key amine and side-chain intermediates, followed by their coupling and final reduction, are provided in the experimental section of the patent documentation. Operators must adhere strictly to the specified stoichiometry and temperature profiles to maintain the integrity of the chiral centers throughout the synthesis. For a comprehensive guide on executing this synthesis with optimal results, please refer to the structured protocol below.
- Synthesize the chiral amine intermediate (Formula 5) via diazonium salt reaction with L-cystine ester followed by reduction and cyclization.
- Prepare the chiral side-chain acid or ester (Formula 7/8) by displacing a mesylate with an arylthiophenol.
- Couple the amine and side-chain fragments to form a stable amide intermediate (Formula 9), then reduce with borane to yield the final amine.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this synthetic route offers profound advantages for supply chain stability and cost management. The elimination of cryogenic conditions and hazardous reagents drastically simplifies the engineering requirements for production facilities, allowing for the use of standard glass-lined or stainless steel reactors without the need for specialized low-temperature equipment. This simplification directly translates to lower capital expenditure and reduced operational complexity. Furthermore, the ability to purify intermediates via crystallization rather than difficult chromatographic separations enhances the overall throughput of the manufacturing line. These factors combine to create a more resilient supply chain capable of meeting fluctuating market demands without compromising on quality or delivery timelines.
- Cost Reduction in Manufacturing: The new process eliminates the need for expensive and potentially explosive reagents such as alkyl diazodicarboxylates and reduces the consumption of solvents associated with complex workups. By avoiding the generation of massive quantities of phosphine oxide waste, the cost of waste treatment and disposal is significantly lowered. Additionally, the higher overall yield resulting from fewer synthetic steps and improved reproducibility means that less raw material is required to produce the same amount of final product. These efficiencies collectively drive down the cost of goods sold, providing a competitive edge in the pricing of high-purity pharmaceutical intermediates.
- Enhanced Supply Chain Reliability: The reliance on stable, isolable intermediates mitigates the risk of batch failures that were common with the unstable aldehyde route. Since the key amide intermediate can be stockpiled and tested before final conversion, manufacturers can maintain a buffer inventory that ensures continuous supply even if upstream disruptions occur. The use of commercially available starting materials like L-cystine esters and substituted thiophenols further reduces dependency on custom-synthesized reagents that may have long lead times. This robustness ensures that procurement managers can secure a reliable pharmaceutical intermediate supplier who can guarantee on-time delivery for critical API projects.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, significantly reducing the E-factor by minimizing waste generation. The avoidance of heavy metal catalysts and the reduction of solvent usage align with increasingly strict environmental regulations globally. The scalability of the reaction is proven by the successful execution of the steps on a multi-gram to kilogram scale in the patent examples, demonstrating that the chemistry translates effectively from the laboratory to the pilot plant. This ease of scale-up allows for rapid commercialization of new drug candidates utilizing this scaffold, accelerating time-to-market for pharmaceutical partners.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this synthetic pathway is crucial for stakeholders evaluating its implementation. The following questions address common concerns regarding the practical application of this technology in a commercial setting. The answers are derived directly from the technical specifications and experimental data provided in the patent literature, ensuring accuracy and relevance for decision-makers.
Q: Why is the new amide reduction method superior to the previous reductive amination route?
A: The new method avoids unstable aldehyde intermediates and cryogenic conditions (<-60°C), utilizing stable crystalline amides instead, which significantly improves reproducibility and safety.
Q: How does this process improve stereochemical purity?
A: By using chiral starting materials like L-cystine and maintaining mild reaction conditions during coupling and reduction, the process achieves diastereomeric excess values >97% without racemization.
Q: Is this synthetic route suitable for industrial scale-up?
A: Yes, the elimination of explosive reagents and extreme temperatures, combined with the ability to purify intermediates via recrystallization, makes the process fully adaptable to semi-industrial and industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzoxathiepine Derivative Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, leveraging advanced synthetic technologies like those described in CN1946708A to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. We are committed to maintaining stringent purity specifications through our rigorous QC labs, guaranteeing that every batch of benzoxathiepine intermediate meets the highest industry standards. Our dedication to process optimization allows us to offer cost-effective solutions without compromising on the quality essential for pharmaceutical applications.
We invite you to collaborate with us to explore how this innovative synthetic route can enhance your supply chain and reduce your manufacturing costs. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to support your R&D and commercialization efforts. Let us be your partner in bringing life-saving cardiovascular medications to market faster and more efficiently.
