Advanced 5-CNAC Manufacturing Process for Reliable Pharmaceutical Intermediate Supplier
The pharmaceutical industry continuously seeks robust manufacturing routes for critical permeation enhancers like 5-CNAC (N-(5-chlorosalicyloyl)-8-aminocaprylic acid). A detailed analysis of patent CN101080384A reveals a significant technological breakthrough in the synthesis of this key pharmaceutical intermediate and its corresponding disodium monohydrate salt. This patent outlines a novel methodology that addresses longstanding challenges regarding purity, safety, and scalability associated with conventional production methods. By implementing a biphasic reaction system and optimizing reagent selection, the described process achieves superior impurity profiles and operational safety. For R&D directors and procurement specialists, understanding these technical nuances is vital for securing a reliable supply chain of high-purity salicylamides. The following insight report dissects the chemical innovations within this patent to demonstrate their value for commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
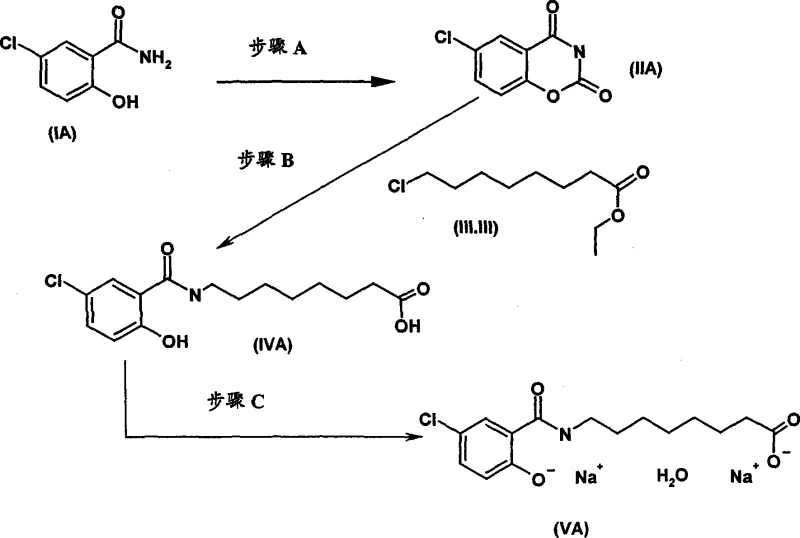
Historically, the synthesis of N-substituted salicylamides has relied on processes that present significant economic and environmental drawbacks. Prior art methods, such as those disclosed in earlier patents, typically utilize ethyl 8-bromooctanoate as the alkylating agent. This reagent is not only substantially more expensive due to the cost of bromination but also poses severe health and safety risks as a potential mutagenic alkylating agent. Furthermore, conventional cyclization steps often employ acetonitrile and pyridine, solvents that are difficult to recover and recycle efficiently. This leads to increased waste generation and higher operational costs for solvent disposal. The formation of emulsions during the final salt crystallization step in prior art further complicates downstream processing, resulting in inconsistent crystal morphology and potential filtration bottlenecks. These factors collectively hinder the ability to achieve cost reduction in API intermediate manufacturing while maintaining stringent quality standards.
The Novel Approach
The methodology described in patent CN101080384A introduces a paradigm shift by replacing hazardous bromo-reagents with safer chloro-equivalents. Specifically, the process utilizes ethyl 8-chlorooctanoate, which is more readily available and economically favorable. The innovation lies in the in situ generation of the reactive bromo-species using catalytic sodium bromide, thereby combining the safety of chloro-reagents with the reactivity of bromo-species. Additionally, the cyclization step employs a biphasic n-butyl acetate and water system with a recyclable alkyl-substituted pyridine base. This two-phase system effectively hydrolyzes unwanted O-acylated intermediates, ensuring that the final product achieves a purity level exceeding 98%. The crystallization process is also refined to occur from a homogeneous acetone and water mixture, preventing emulsion formation and enabling the production of large, easily filterable crystals. These improvements collectively enhance the commercial scale-up of complex salicylamides by streamlining operations and reducing waste.
Mechanistic Insights into Biphasic Cyclization and Catalytic Alkylation
The core chemical innovation resides in the meticulous control of reaction conditions to favor the desired N-acylation over O-acylation. In Step A, the use of a biphasic system comprising n-butyl acetate and water is critical. The water phase facilitates the rapid hydrolysis of any O-acylated intermediates that may form, while the organic phase retains the desired 6-chlorobenzoxazinedione product. The selection of 5-ethyl-2-methyl-pyridine as the base is particularly strategic; unlike simple pyridine, this alkyl-substituted derivative is water-insoluble, allowing it to remain in the organic phase and be recovered for reuse. This mechanistic design ensures that the reaction proceeds to completion with minimal starting material remaining, typically less than 2%. The ability to recycle the base significantly contributes to the overall process efficiency and reduces the consumption of auxiliary chemicals. For technical teams, this highlights the importance of phase behavior in optimizing selectivity and yield during the cyclization of salicylamide derivatives.
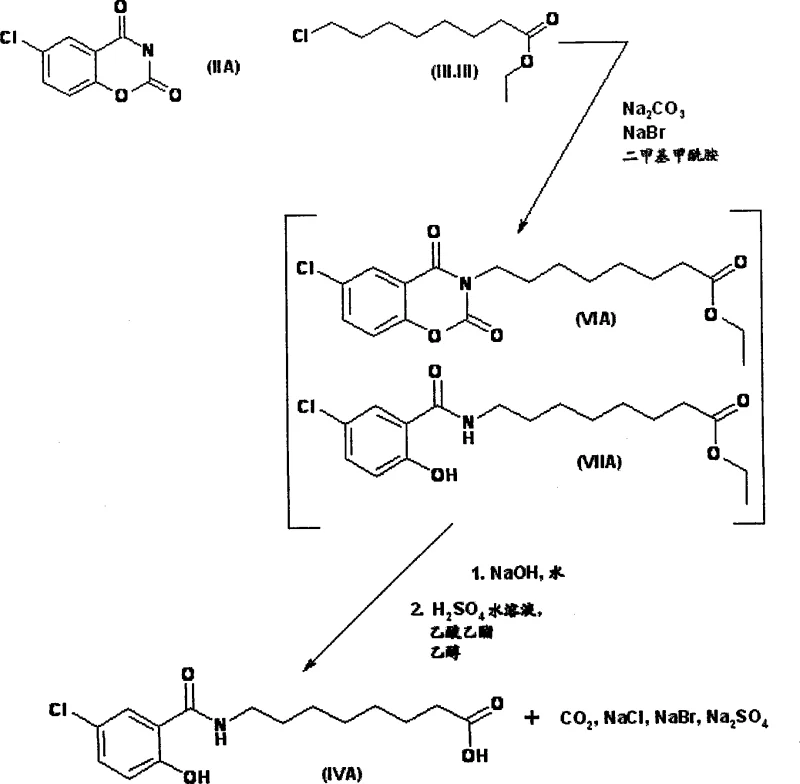
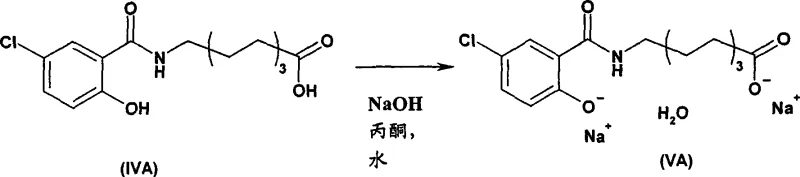
In Step B, the alkylation mechanism leverages the halide exchange capability of sodium bromide. Although ethyl 8-chlorooctanoate is less reactive than its bromo-counterpart, the presence of catalytic sodium bromide in dimethylformamide facilitates the in situ formation of the more reactive ethyl 8-bromooctanoate. This allows the alkylation to proceed with high selectivity, typically around 95%, while minimizing the formation of dialkylated by-products. The process avoids the isolation of intermediate esters, proceeding directly to saponification in the same vessel. This one-pot strategy not only reduces processing time but also minimizes material loss during transfer and purification steps. The subsequent crystallization in Step C utilizes a controlled addition of sodium hydroxide in an acetone and water mixture. This ensures the formation of a homogeneous solution prior to crystallization, avoiding the emulsions common in prior art. The result is a well-defined crystal lattice that can be efficiently dried to yield the disodium monohydrate with high purity.
How to Synthesize 5-CNAC Efficiently
Implementing this synthesis route requires precise control over temperature, reagent addition rates, and phase separation to maximize yield and purity. The process is designed to be scalable, moving seamlessly from laboratory optimization to industrial production without compromising on quality parameters. Operators must adhere to specific protocols regarding the slow addition of base during the alkylation step to prevent exothermic runaways and ensure complete conversion. The following guide outlines the standardized operational framework derived from the patent data, ensuring reproducibility and safety across different manufacturing scales. Detailed standard operating procedures for each unit operation are essential for maintaining compliance with Good Manufacturing Practices.
- Cyclization of 5-chlorosalicylamide in a biphasic n-butyl acetate/water system using 5-ethyl-2-methyl-pyridine to form 6-chlorobenzoxazinedione.
- Alkylation with ethyl 8-chlorooctanoate in DMF using sodium carbonate and catalytic sodium bromide to generate the intermediate ester.
- Saponification and crystallization from acetone/water mixture with sodium hydroxide to yield the disodium monohydrate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the technical improvements in this patent translate directly into tangible business value and risk mitigation. The shift from bromo-reagents to chloro-reagents significantly lowers raw material costs while eliminating the regulatory and safety burdens associated with handling mutagenic substances. The ability to recycle the organic base reduces the consumption of auxiliary chemicals, further driving down the cost of goods sold. Moreover, the robustness of the one-pot alkylation and saponification sequence enhances supply chain reliability by reducing the number of unit operations and potential failure points. The improved crystallization behavior ensures consistent product quality and faster turnaround times, which is critical for meeting tight delivery schedules in the pharmaceutical sector. These factors collectively position this manufacturing route as a superior choice for reducing lead time for high-purity permeation enhancers.
- Cost Reduction in Manufacturing: The substitution of expensive ethyl 8-bromooctanoate with the more economical ethyl 8-chlorooctanoate results in substantial raw material savings. Furthermore, the catalytic nature of the sodium bromide additive means that only small quantities are required, minimizing reagent costs. The recyclability of the 5-ethyl-2-methyl-pyridine base eliminates the need for continuous purchase of fresh base, leading to significant long-term operational expenditure reductions. By avoiding the isolation of intermediates, the process also saves on solvent usage and energy consumption associated with additional drying and purification steps. These cumulative efficiencies drive a lower overall manufacturing cost without compromising the stringent purity specifications required for pharmaceutical applications.
- Enhanced Supply Chain Reliability: Utilizing ethyl 8-chlorooctanoate mitigates supply risks associated with specialized bromo-reagents, which may have limited availability or long lead times. The robustness of the biphasic system ensures consistent batch-to-batch quality, reducing the likelihood of production delays due to out-of-specification results. The simplified workflow, characterized by fewer isolation steps, decreases the potential for human error and equipment downtime. This operational stability allows for more accurate production planning and inventory management, ensuring a steady flow of high-purity 5-CNAC to downstream customers. Consequently, partners can rely on a more predictable and resilient supply chain for their critical API intermediate needs.
- Scalability and Environmental Compliance: The process is inherently designed for commercial scale-up, with reaction conditions that are easily manageable in large-scale reactors. The use of less hazardous reagents aligns with increasingly strict environmental and safety regulations, reducing the compliance burden on manufacturing facilities. The reduction in solvent waste and the ability to recycle key reagents contribute to a smaller environmental footprint, supporting sustainability goals. The improved crystallization process yields larger crystals that are easier to filter and dry, enhancing throughput capacity in existing facilities. This scalability ensures that production can be ramped up to meet growing market demand for 5-CNAC without requiring significant capital investment in new infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the experimental data and process descriptions within the patent documentation. They provide clarity on the specific advantages of this method over traditional approaches, helping stakeholders make informed decisions about adoption. Understanding these details is crucial for evaluating the feasibility of integrating this technology into existing manufacturing portfolios.
Q: Why is ethyl 8-chlorooctanoate preferred over ethyl 8-bromooctanoate?
A: Ethyl 8-chlorooctanoate is significantly cheaper and less hazardous. The process uses catalytic sodium bromide to generate the reactive bromo-species in situ, avoiding the handling of mutagenic bromo-reagents.
Q: How does the biphasic system improve purity?
A: The n-butyl acetate/water system allows for the hydrolysis of unwanted O-acylated intermediates while keeping the desired product in the organic phase, resulting in purity greater than 98%.
Q: What are the advantages of the new crystallization method?
A: Crystallization from a homogeneous acetone/water solution avoids emulsion formation, allowing for better control over crystal size and polymorph, which facilitates filtration and drying.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-CNAC Supplier
The technical advancements detailed in patent CN101080384A underscore the potential for producing high-quality 5-CNAC with enhanced efficiency and safety. NINGBO INNO PHARMCHEM leverages extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to life. Our facility is equipped with rigorous QC labs and adheres to stringent purity specifications to ensure that every batch meets the exacting standards of the global pharmaceutical industry. We understand the critical nature of permeation enhancers in drug delivery systems and are committed to providing a stable and high-quality supply. Our team of experts is ready to assist in translating these patent insights into reliable commercial reality for your specific project needs.
We invite you to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your supply chain. Request a Customized Cost-Saving Analysis to understand the specific economic advantages for your organization. Our team is prepared to provide specific COA data and route feasibility assessments to support your decision-making process. By partnering with us, you gain access to advanced manufacturing capabilities and a dedicated support structure focused on your success. Contact us today to initiate a conversation about optimizing your 5-CNAC supply strategy.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
