Advanced Catalytic Synthesis of Difluoromethyl Sulfoximines for High-Purity Pharma Intermediates
Advanced Catalytic Synthesis of Difluoromethyl Sulfoximines for High-Purity Pharma Intermediates
The landscape of fluorine chemistry in pharmaceutical development has been significantly advanced by the innovations detailed in patent CN101328143B, which introduces a robust and safe methodology for synthesizing fluorine-containing sulfoximine compounds. As the demand for fluorinated motifs in drug design continues to surge, particularly for the difluoromethyl (CF2H) group which acts as a lipophilic hydrogen bond donor and a bioisostere for hydroxymethyl groups, the need for reliable synthetic routes becomes paramount. This patent discloses a novel class of compounds characterized by a highly oxidized sulfur atom bonded to both oxygen and nitrogen, creating a chiral center that offers unique stereochemical opportunities. The breakthrough lies not merely in the structural novelty but in the establishment of a catalytic cycle that bypasses the perilous use of sodium azide, traditionally the standard yet hazardous reagent for introducing nitrogen functionality into sulfoxides. For R&D directors and process chemists, this represents a pivotal shift towards safer, more scalable chemistry that aligns with modern green chemistry principles while maintaining high purity standards essential for API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the preparation of sulfoximine-type compounds has relied heavily on methodologies that present substantial safety and operational challenges for industrial manufacturing. The most prevalent traditional route involves the reaction of sulfoxides with sodium azide (NaN3) in concentrated or fuming sulfuric acid, a process fraught with extreme danger due to the formation of explosive hydrazoic acid intermediates. Furthermore, when attempting to synthesize specific difluoromethyl-substituted variants, these classical azide-based protocols often fail to deliver the desired products or result in decomposition, leaving a critical gap in the availability of these valuable building blocks. The use of toxic heavy metal azides and corrosive acids necessitates specialized equipment, rigorous safety protocols, and complex waste treatment procedures, all of which drive up the cost of goods and extend lead times. Additionally, the harsh acidic conditions can be incompatible with sensitive functional groups often present in complex pharmaceutical scaffolds, limiting the substrate scope and requiring extensive protecting group strategies that further reduce overall efficiency.
The Novel Approach
In stark contrast to the hazardous legacy methods, the technology described in CN101328143B utilizes a copper-catalyzed nitrogen transfer reaction that operates under significantly milder and safer conditions. By employing iminoiodinane reagents, specifically those with the general formula Ph-I=N-SO2R2, in the presence of a copper(II) triflate catalyst, the synthesis achieves high yields without the need for explosive azides. This method allows for the direct conversion of fluorine-containing sulfoxides into the corresponding sulfoximines at temperatures ranging from room temperature to 80°C, utilizing common polar solvents like acetonitrile or DMF. The versatility of this approach is demonstrated by its compatibility with various Rf groups, including CH2F, CF2H, and CF3, thereby unlocking access to a diverse library of fluorinated intermediates previously difficult to obtain. This transition from stoichiometric hazardous reagents to a catalytic, controlled process fundamentally alters the risk profile of the manufacturing operation, making it an attractive option for cost reduction in pharmaceutical manufacturing where safety and throughput are critical metrics.
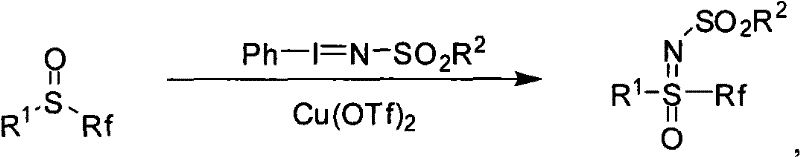
Mechanistic Insights into Copper-Catalyzed Nitrogen Transfer
The core of this technological advancement lies in the mechanistic pathway facilitated by the copper catalyst, which activates the iminoiodinane reagent for efficient nitrogen insertion into the sulfur-oxygen bond. The copper(II) species likely coordinates with the nitrogen atom of the iminoiodinane, enhancing its electrophilicity and promoting the transfer of the nitrene-like species to the lone pair of the sulfoxide sulfur. This concerted or stepwise insertion results in the formation of the S=N double bond while retaining the S=O bond, establishing the characteristic sulfoximine motif with high regioselectivity. The choice of the sulfonyl group on the nitrogen (R2) plays a crucial role in stabilizing the reagent and influencing the electronic properties of the transferring nitrogen, allowing for fine-tuning of the reaction kinetics. Understanding this mechanism is vital for process optimization, as it suggests that the reaction rate can be modulated by adjusting the electronic nature of the catalyst ligands or the solvent polarity, providing R&D teams with levers to maximize conversion and minimize side reactions.
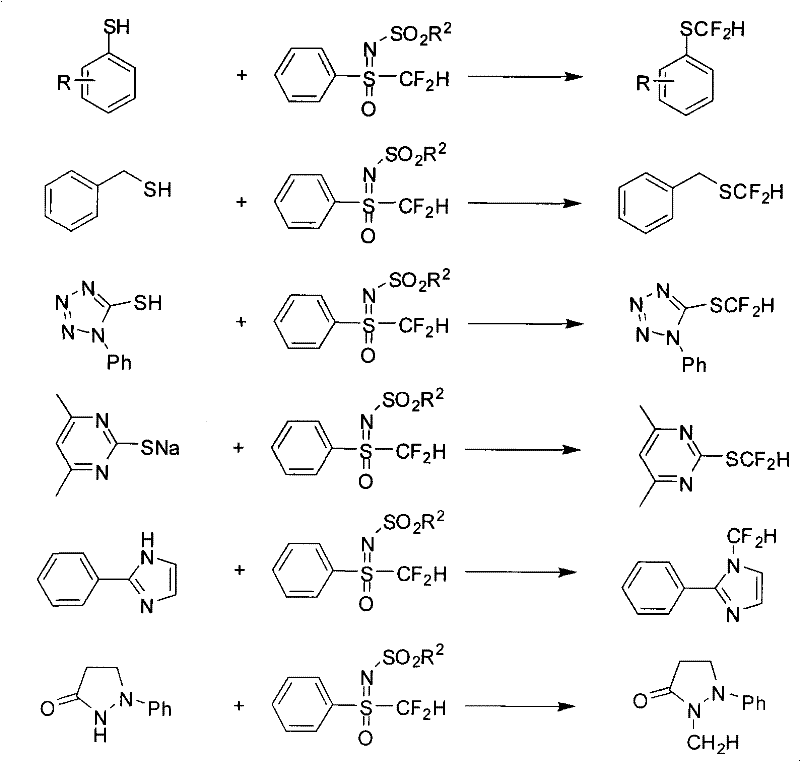
Beyond the primary synthesis, the utility of these fluorine-containing sulfoximines is exemplified by their ability to act as potent difluoromethylating agents for various nucleophiles, a feature that significantly broadens their application scope. As illustrated in the reaction schemes, the acidic proton alpha to the sulfur in the monofluoromethyl variant can be deprotonated to form a nucleophilic carbanion, which then attacks electrophiles like alkyl halides. More importantly for drug discovery, the difluoromethyl sulfoximine can react with sulfur and nitrogen nucleophiles, such as thiols and heterocycles, to install the CF2H group directly onto the target molecule. This transformation proceeds via a nucleophilic substitution mechanism where the sulfoximine moiety acts as a leaving group or a directing group, facilitating the formation of C-S or C-N bonds with the difluoromethyl unit intact. This dual functionality—being both a stable intermediate and a reactive reagent—makes these compounds invaluable tools for constructing complex fluorinated architectures found in modern agrochemicals and pharmaceuticals.
How to Synthesize N-(p-Tolylsulfonyl)-S-difluoromethyl-S-phenylsulfoximine Efficiently
The practical implementation of this synthesis requires precise control over reaction parameters to ensure reproducibility and high purity, critical factors for any reliable fluorine-containing intermediate supplier. The process begins with the careful selection of high-purity starting materials, specifically the fluorinated sulfoxide and the iminoiodinane reagent, which are combined in a dry, polar aprotic solvent under an inert atmosphere to prevent moisture-induced degradation. The addition of the copper catalyst must be managed to ensure homogeneous distribution, followed by a controlled heating phase that balances reaction rate with thermal safety. While the general procedure is robust, specific optimization of stoichiometry and workup procedures is often required to isolate the product in its highest crystalline form, free from copper residues or unreacted starting materials. For detailed operational parameters and specific stoichiometric ratios validated through extensive experimentation, please refer to the standardized synthesis steps provided below.
- Prepare the reaction mixture by combining the fluorine-containing sulfoxide substrate and the iminoiodinane reagent (Ph-I=N-SO2R2) in a polar solvent such as acetonitrile or DMF.
- Add the copper(II) triflate catalyst (Cu(OTf)2) to the mixture under an inert atmosphere to initiate the nitrogen transfer reaction.
- Heat the reaction mixture to a temperature between room temperature and 80°C and stir for 10 to 24 hours to ensure complete conversion to the sulfoximine product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this copper-catalyzed methodology offers profound advantages that extend well beyond the laboratory bench, directly impacting the bottom line and operational resilience. The elimination of sodium azide removes a major bottleneck associated with the storage, handling, and disposal of highly regulated explosive materials, thereby simplifying compliance requirements and reducing insurance and safety infrastructure costs. Furthermore, the use of common solvents and commercially available copper salts ensures that the supply chain for raw materials is robust and less susceptible to geopolitical disruptions or vendor shortages that often plague specialty reagents. The mild reaction conditions also translate to lower energy consumption, as the process does not require extreme cryogenic cooling or high-pressure vessels, contributing to a smaller carbon footprint and reduced utility expenses. These factors collectively enhance the economic viability of producing fluorinated intermediates at scale, making it a strategically sound choice for long-term sourcing agreements.
- Cost Reduction in Manufacturing: The shift away from hazardous azide chemistry eliminates the need for expensive blast-proof facilities and specialized waste treatment protocols required for neutralizing toxic azide byproducts. By utilizing a catalytic amount of copper triflate rather than stoichiometric quantities of dangerous reagents, the material cost per kilogram of product is significantly optimized. Additionally, the higher selectivity of the new method reduces the formation of difficult-to-remove impurities, lowering the cost associated with downstream purification processes like chromatography or recrystallization. This streamlined workflow allows for a more efficient allocation of resources, driving down the overall cost of goods sold without compromising on the quality of the final API intermediate.
- Enhanced Supply Chain Reliability: The reliance on stable, shelf-stable reagents like iminoiodinanes ensures a consistent supply of critical inputs, mitigating the risk of production stoppages due to reagent degradation or availability issues. Unlike azide-based processes that may face strict transportation regulations limiting shipment sizes, the reagents in this novel pathway can be sourced and transported with greater flexibility, ensuring continuity of supply even during logistical challenges. This reliability is crucial for maintaining just-in-time inventory levels and meeting the stringent delivery schedules demanded by global pharmaceutical clients, positioning the manufacturer as a dependable partner in the value chain.
- Scalability and Environmental Compliance: The inherent safety of the copper-catalyzed reaction facilitates easier scale-up from gram to multi-ton quantities, as the exothermic profile is manageable and does not pose the runaway reaction risks associated with azide chemistry. This scalability is complemented by improved environmental compliance, as the process generates less hazardous waste and avoids the release of toxic gases, aligning with increasingly stringent global environmental regulations. The ability to scale safely and sustainably ensures that the manufacturing capacity can grow in tandem with market demand, providing a secure foundation for future business expansion and partnership opportunities.
Frequently Asked Questions (FAQ)
To address common inquiries regarding the technical specifications and commercial viability of this technology, we have compiled a set of answers based on the detailed data within the patent literature. These responses cover critical aspects ranging from safety profiles to substrate compatibility, providing clarity for technical decision-makers evaluating this route for their specific projects. Understanding these nuances is essential for assessing the feasibility of integrating this chemistry into existing production lines or new drug development pipelines. We encourage stakeholders to review these insights to fully appreciate the transformative potential of this synthetic approach.
Q: Why is the copper-catalyzed method safer than traditional azide methods?
A: Traditional methods often rely on sodium azide (NaN3) and fuming sulfuric acid, which pose severe explosion and toxicity risks. The novel copper-catalyzed protocol utilizes stable iminoiodinane reagents under mild conditions, eliminating the handling of hazardous azides and significantly improving operational safety.
Q: What are the primary applications of difluoromethyl sulfoximines?
A: These compounds serve as versatile intermediates for introducing the difluoromethyl (CF2H) group into organic molecules. They are particularly valuable in medicinal chemistry for synthesizing bioisosteres of hydroxymethyl groups, enhancing metabolic stability and lipophilicity in drug candidates.
Q: Can this synthesis be scaled for industrial production?
A: Yes, the process uses commercially available catalysts and solvents with manageable exotherms compared to azide chemistry. The absence of highly explosive intermediates makes it highly suitable for commercial scale-up and continuous manufacturing processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Difluoromethyl Sulfoximine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of accessing high-quality fluorinated intermediates to accelerate drug discovery and development timelines. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory innovation to industrial reality is seamless and efficient. We are committed to delivering products that meet stringent purity specifications through our rigorous QC labs, which utilize state-of-the-art analytical instrumentation to verify every batch against the highest industry standards. By leveraging the advanced copper-catalyzed synthesis described in CN101328143B, we can offer a superior supply solution that balances performance, safety, and cost-effectiveness for your most challenging fluorination projects.
We invite you to engage with our technical procurement team to discuss how our capabilities can support your specific supply chain needs and drive value for your organization. By requesting a Customized Cost-Saving Analysis, you can gain a deeper understanding of the economic benefits of switching to this safer, more efficient manufacturing route. We are ready to provide specific COA data and route feasibility assessments tailored to your target molecules, ensuring that you have all the necessary information to make informed sourcing decisions. Contact us today to explore a partnership that combines cutting-edge chemistry with reliable commercial execution.
