Advanced Palladium-Catalyzed Ring Carbonylation for High-Purity Carvedilol Intermediates
Advanced Palladium-Catalyzed Ring Carbonylation for High-Purity Carvedilol Intermediates
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical active pharmaceutical ingredients (APIs) and their precursors. Patent CN1266131C introduces a transformative methodology for the preparation of heterocyclic indene analogs, specifically targeting the synthesis of 4-hydroxycarbazole and its N-protected derivatives. These compounds serve as indispensable structural units for potent cardiovascular medications such as Carvedilol. The disclosed technology leverages a novel palladium-catalyzed ring carbonylation strategy that fundamentally alters the economic and technical landscape of producing these high-value intermediates. By shifting away from traditional harsh conditions, this process enables the efficient construction of complex fused ring systems directly from accessible allyl ester precursors. 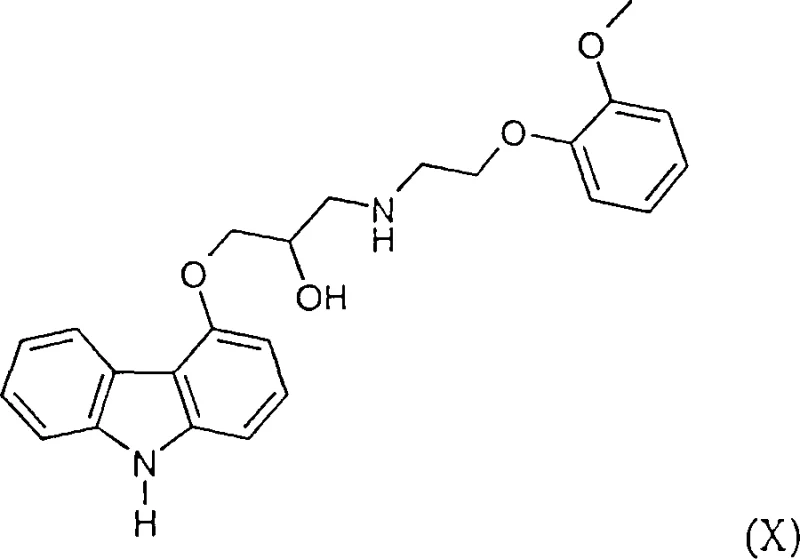 This innovation is particularly relevant for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status, as it addresses long-standing challenges regarding yield, purity, and operational safety. The ability to utilize crude starting materials without extensive pre-purification represents a significant leap forward in process chemistry, reducing both waste generation and processing time. For R&D teams focused on optimizing impurity profiles, this route offers a cleaner reaction trajectory that simplifies downstream isolation and enhances the overall quality of the final API.
This innovation is particularly relevant for manufacturers aiming to secure a reliable pharmaceutical intermediate supplier status, as it addresses long-standing challenges regarding yield, purity, and operational safety. The ability to utilize crude starting materials without extensive pre-purification represents a significant leap forward in process chemistry, reducing both waste generation and processing time. For R&D teams focused on optimizing impurity profiles, this route offers a cleaner reaction trajectory that simplifies downstream isolation and enhances the overall quality of the final API.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pyrrole and indole derivatives via catalytic ring carbonylation has been plagued by significant operational inefficiencies and economic burdens. Prior art, such as the methods described by Hiday et al., typically necessitates extremely high reaction temperatures and substantial catalyst loadings to drive the cyclization to completion. These harsh thermal conditions often lead to moderate selectivities, resulting in complex reaction mixtures that are difficult to separate and purify. Furthermore, the starting materials required for these legacy processes are frequently expensive and not commercially available off-the-shelf, demanding lengthy and costly multi-step preparatory sequences before the actual ring-forming reaction can even commence. This reliance on bespoke, high-cost precursors creates a bottleneck in the supply chain, increasing the lead time for high-purity pharmaceutical intermediates and inflating the overall cost of goods sold. Additionally, the rigorous purification steps often needed to render these precursors suitable for reaction add further layers of complexity, solvent consumption, and waste disposal issues, making the conventional approach less attractive for large-scale commercial manufacturing.
The Novel Approach
In stark contrast, the methodology outlined in CN1266131C presents a streamlined and economically superior alternative that mitigates the drawbacks of earlier techniques. The core of this innovation lies in the ability to perform efficient ring carbonylation under remarkably mild conditions, typically around 90°C, using a sophisticated palladium-phosphine catalyst system. A defining feature of this novel approach is its tolerance for impurities; the substrate, specifically compounds of formula (II), can be employed directly as a crude substance without the need for prior purification via crystallization or distillation. 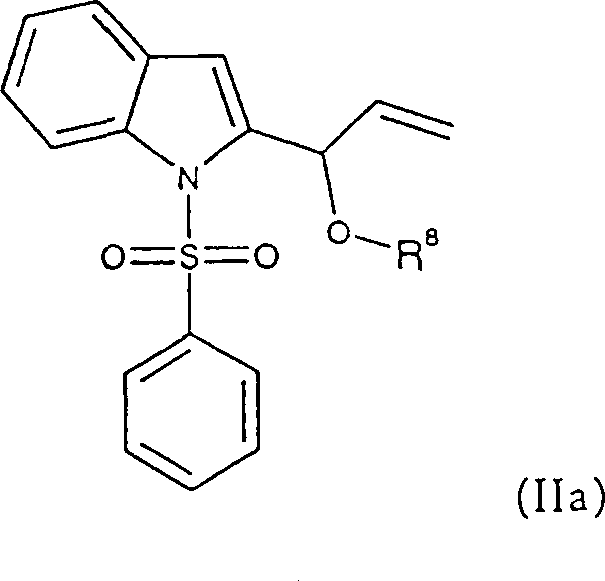 This capability drastically simplifies the upstream process flow, eliminating unit operations that traditionally consume time and resources. The reaction utilizes readily available reagents such as acid anhydrides and bases in common organic solvents like toluene, ensuring that the process remains compatible with existing infrastructure in fine chemical plants. By enabling the use of commercially accessible starting materials and tolerating crude inputs, this method significantly lowers the barrier to entry for production, offering a compelling value proposition for cost reduction in API manufacturing. The result is a more agile supply chain capable of responding rapidly to market demands for critical cardiovascular drug intermediates.
This capability drastically simplifies the upstream process flow, eliminating unit operations that traditionally consume time and resources. The reaction utilizes readily available reagents such as acid anhydrides and bases in common organic solvents like toluene, ensuring that the process remains compatible with existing infrastructure in fine chemical plants. By enabling the use of commercially accessible starting materials and tolerating crude inputs, this method significantly lowers the barrier to entry for production, offering a compelling value proposition for cost reduction in API manufacturing. The result is a more agile supply chain capable of responding rapidly to market demands for critical cardiovascular drug intermediates.
Mechanistic Insights into Pd-Catalyzed Cyclocarbonylation
The success of this synthetic route hinges on the precise orchestration of a transition metal-catalyzed cycle involving palladium. The catalytic system is generated in situ, typically utilizing palladium acetate as the metal source and triphenylphosphine as the preferred ligand. The interaction between the palladium center and the phosphine ligands creates an electron-rich environment that facilitates the oxidative addition and subsequent migratory insertion of carbon monoxide into the organic framework. Critical to the reaction's efficiency is the molar ratio of the phosphine ligand to the transition metal compound, which is optimized between 6:1 and 15:1. This specific stoichiometry ensures that the active catalytic species remains stable throughout the reaction duration, preventing premature catalyst deactivation or the formation of inactive palladium black. The reaction proceeds under a carbon monoxide pressure ranging from 35 to 60 bar, which is sufficient to drive the carbonylation equilibrium towards the desired cyclic ester product without requiring extreme pressures that would compromise safety or equipment integrity.
From an impurity control perspective, the mechanism offers distinct advantages over non-catalytic thermal cyclizations. The specificity of the palladium catalyst minimizes side reactions such as polymerization of the allyl group or decomposition of the sensitive indole nucleus, which are common pitfalls in high-temperature processes. Following the carbonylation step, the resulting ester intermediate (Formula III) undergoes a straightforward saponification. This hydrolysis step, conducted in a biphasic mixture of aqueous sodium hydroxide and toluene or a homogeneous methanolic sodium methoxide solution, cleanly cleaves the ester bond to reveal the free hydroxyl group. The mildness of the saponification conditions preserves the integrity of the newly formed carbazole ring system, ensuring that the final 4-hydroxy product maintains a high degree of chemical purity. This two-stage sequence—carbonylation followed by saponification—provides a robust handle for process chemists to tune the reaction parameters for optimal yield and minimal byproduct formation, thereby securing a consistent supply of high-purity heterocyclic indene analogs.
How to Synthesize 4-Hydroxycarbazole Derivatives Efficiently
The practical execution of this synthesis involves a carefully controlled sequence of reactions designed to maximize throughput while maintaining safety standards. The process begins with the preparation or procurement of the allyl ester precursor, which, as noted, does not require rigorous purification before use. This precursor is then subjected to the carbonylation conditions in a pressure-rated reactor, where the palladium catalyst, base, and acid anhydride are introduced under an inert atmosphere.
- Prepare the starting material, such as 1-(1-benzenesulfonyl-1H-indol-2-yl)-allyl acetate, potentially using crude material without extensive purification.
- Perform ring carbonylation in an autoclave using a palladium catalyst (e.g., Pd(OAc)2), triphenylphosphine ligand, base, and acid anhydride under CO pressure (35-60 bar) at 90°C.
- Subject the resulting ester intermediate to saponification using aqueous sodium hydroxide or sodium methoxide to yield the final 4-hydroxy heterocyclic product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic benefits that extend beyond simple chemical yield. The primary advantage lies in the drastic simplification of the raw material supply chain. Since the process tolerates crude starting materials and utilizes commercially available reagents like acrolein and indole derivatives, manufacturers are not locked into sourcing expensive, custom-synthesized precursors from limited vendors. This flexibility enhances supply chain reliability by diversifying the potential supplier base and reducing the risk of production stoppages due to raw material shortages. Furthermore, the elimination of intermediate purification steps, such as distillation or recrystallization of the starting material, leads to substantial cost savings in terms of solvent usage, energy consumption, and labor hours. These operational efficiencies directly contribute to a lower cost of goods, allowing for more competitive pricing in the global marketplace for pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The ability to bypass the purification of the formula (II) starting material represents a significant reduction in processing costs. Traditional methods often require multiple crystallization or chromatography steps to remove impurities that would otherwise poison the catalyst or lower the yield. By accepting crude inputs, this process removes these unit operations entirely, thereby reducing solvent waste and energy demand associated with heating and cooling cycles. Additionally, the use of a highly active palladium catalyst system allows for lower catalyst loadings relative to older technologies, minimizing the expense associated with precious metal recovery and disposal. The overall effect is a leaner manufacturing process that delivers the same high-quality output with a significantly reduced operational footprint and expenditure.
- Enhanced Supply Chain Reliability: Reliance on commercially available building blocks such as 1-(phenylsulfonyl)indole and acrolein ensures a stable and predictable supply chain. Unlike proprietary intermediates that may have long lead times or single-source dependencies, these commodity chemicals are produced by multiple global suppliers, mitigating the risk of supply disruptions. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in raw material quality, further stabilizing production schedules. For supply chain heads, this translates to improved on-time delivery performance and the ability to scale production volumes rapidly in response to fluctuating market demand for cardiovascular medications without facing raw material bottlenecks.
- Scalability and Environmental Compliance: The reaction conditions, operating at moderate temperatures around 90°C and pressures up to 60 bar, are well within the capabilities of standard industrial autoclaves, facilitating easy scale-up from pilot plant to commercial tonnage. The use of common solvents like toluene and ethyl acetate simplifies solvent recovery and recycling programs, aligning with modern environmental compliance standards. Moreover, the high atom economy of the carbonylation step, where carbon monoxide is incorporated directly into the product structure, minimizes the generation of stoichiometric byproducts. This green chemistry profile reduces the burden on waste treatment facilities and lowers the environmental impact of the manufacturing process, making it an attractive option for companies committed to sustainable production practices.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this ring carbonylation technology. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the optimal reaction conditions for the ring carbonylation step?
A: The process operates optimally at temperatures between 60-120°C, most preferably around 90°C, with a carbon monoxide pressure of 35 to 60 bar. A palladium-to-phosphine ligand ratio of approximately 1:6 to 1:15 is recommended for maximum catalytic efficiency.
Q: Does the starting material require high purity before the carbonylation reaction?
A: No, one of the significant advantages of this method is that the starting material, such as formula (II) compounds, can be used in a crude state without prior purification by crystallization or distillation, which streamlines the overall production workflow.
Q: Which catalyst system is preferred for this synthesis?
A: The preferred catalyst system utilizes palladium acetate (Pd(OAc)2) combined with triphenylphosphine as the ligand. This combination provides high selectivity and allows the reaction to proceed under milder conditions compared to traditional methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Hydroxycarbazole Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes in the modern pharmaceutical landscape. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of essential intermediates. We are fully equipped to implement advanced catalytic processes such as the palladium-mediated ring carbonylation described in CN1266131C, leveraging our rigorous QC labs to meet stringent purity specifications required for API synthesis. Our commitment to technical excellence allows us to navigate the complexities of heterocyclic chemistry, delivering high-quality 4-hydroxycarbazole derivatives that serve as the foundation for life-saving cardiovascular therapies.
We invite potential partners to engage with our technical procurement team to discuss how this innovative technology can be tailored to your specific production needs. By collaborating with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to this streamlined process. We encourage you to request specific COA data and route feasibility assessments to verify the compatibility of our capabilities with your project requirements. Together, we can optimize the supply chain for high-purity pharmaceutical intermediates, driving down costs and accelerating the availability of critical medicines to patients worldwide.
