Advancing Pharmaceutical Stability: High-Purity Crystalline Salts of 3,3-Diphenylpropylamine Derivatives for Commercial Scale-Up
Advancing Pharmaceutical Stability: High-Purity Crystalline Salts of 3,3-Diphenylpropylamine Derivatives for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust solutions to enhance the bioavailability and shelf-life of active pharmaceutical ingredients (APIs), particularly in the treatment of chronic conditions like urinary incontinence. Patent CN1215045C introduces a groundbreaking methodology for producing high-purity, crystalline, and stable compounds of new derivatives of 3,3-diphenylpropylamine in the form of their salts. This innovation addresses critical limitations associated with previous generations of antispasmodic agents, such as Oxybutynin and Tolterodin, by providing prodrugs with improved pharmacokinetic profiles. The core of this technology lies in the transformation of basic components into specific salt forms that avoid the pitfalls of amorphous solids, ensuring consistent performance in drug formulations. By leveraging regio- and chemoselective synthesis pathways, manufacturers can now access intermediates that are not only highly pure but also possess the physical stability required for rigorous pharmaceutical processing and long-term storage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
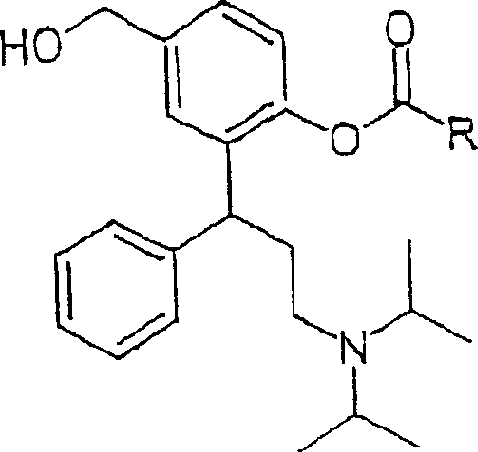
Historically, the development of 3,3-diphenylpropylamine derivatives has been hindered by significant physicochemical challenges that compromise their efficacy as therapeutic agents. Compounds possessing the structure of Formula A, while biologically active, exhibit notoriously low solubility in water, which poses a severe limitation on their oral bioavailability and absorption rates in the human body. Furthermore, these monoesters demonstrate a troubling tendency towards intramolecular ester group transfer, leading to chemical instability during prolonged storage periods. As the content of the desired compound decreases over time, degradation products such as diester compounds and free diols accumulate, rendering the medication less effective or potentially unsafe. Additionally, attempts to convert these basic components into salts using standard acid treatments often result in solid forms that are completely amorphous and highly hygroscopic. Such amorphous salts lack the necessary chemical stability to withstand pharmaceutical processing, making them unsuitable for reliable mass production and commercial distribution.
The Novel Approach
The novel approach detailed in the patent overcomes these historical barriers through a sophisticated strategy of salt formation that yields highly pure, crystalline, and stable compounds. By converting the 3,3-diphenylpropylamine derivatives into salts with physiologically compatible inorganic or organic acids, the invention successfully avoids the defects of amorphous instability and hygroscopicity. The resulting salts, particularly the hydrogen fumarate and hydrochloride hydrate forms, exhibit high melting points and remain stable at room temperature, ensuring they do not absorb moisture from the environment. This crystalline nature allows for direct crystallization from common solvents, facilitating easier purification and handling during the manufacturing process. The ability to produce these compounds in a stable, crystalline state represents a significant leap forward, enabling the creation of pharmaceutical preparations that maintain their potency and safety profile throughout their shelf life, thereby solving the critical problem of chemical degradation in earlier iterations of these drugs.
Mechanistic Insights into Regioselective Acylation and Salt Crystallization
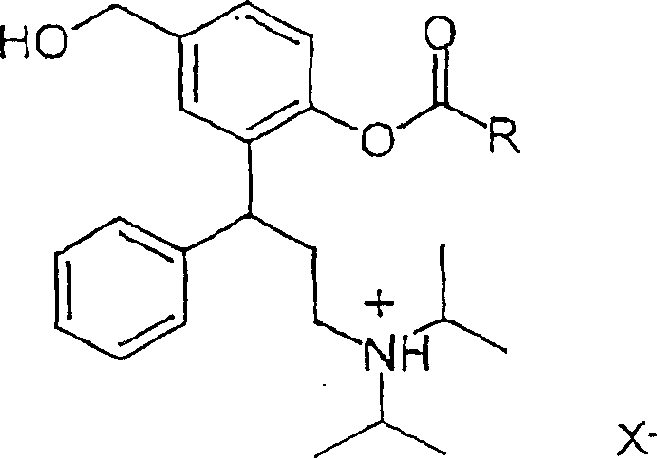
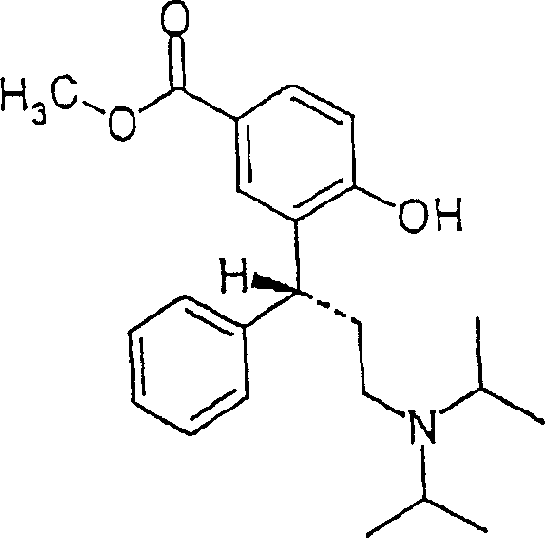
The chemical elegance of this process is rooted in its precise control over regioselectivity and stereochemistry, ensuring the production of the desired R-configuration compounds with minimal impurities. The synthesis begins with the preparation of key intermediates, such as R-(-)-3-(3-diisopropylamino-phenyl-propyl)-4-hydroxy-benzoic acid methyl ester, which is obtained through catalytic hydrogenation using Raney nickel. This step is crucial for removing protecting groups without affecting the sensitive amine functionality. Subsequent reduction of the ester to the corresponding phenol alcohol is performed under strictly controlled low-temperature conditions using reducing agents like lithium aluminum hydride. The final acylation step is where the true mechanistic precision is displayed; the reaction with acid chlorides, such as isobutyryl chloride, occurs with exceptional regio- and chemoselectivity at the phenolic hydroxyl group. This selectivity prevents unwanted side reactions at the primary alcohol or amine sites, resulting in a clean product profile that simplifies downstream purification efforts significantly.
Furthermore, the mechanism of salt formation is engineered to promote spontaneous crystallization, a phenomenon that is rare and highly valuable in industrial chemistry. When the acylated base is treated with acids like fumaric acid in specific solvent systems, such as 2-butanol mixed with cyclohexane, the solution reaches a supersaturated state that triggers the immediate nucleation of stable crystals. This spontaneous crystallization is not merely a physical change but a thermodynamic stabilization of the molecule into a lattice structure that resists degradation. The process effectively locks the molecule into its most stable conformation, preventing the intramolecular ester transfers that plague the free base forms. By understanding and controlling these mechanistic nuances, manufacturers can consistently produce batches with uniform particle size and purity, which are critical parameters for ensuring the reproducibility of drug dissolution rates and therapeutic outcomes in the final dosage forms.
How to Synthesize R-(+)-2-(3-Diisopropylamino-1-phenylpropyl)-4-hydroxymethylphenyl Isobutyrate Efficiently
The synthesis of these high-value intermediates requires a meticulously orchestrated sequence of reactions that balance yield, purity, and operational safety. The process typically initiates with the preparation of the benzyloxy precursor, followed by a catalytic deprotection step to reveal the phenolic hydroxyl group essential for subsequent functionalization. Once the core scaffold is established, the regioselective acylation is conducted in solvents like dichloromethane or ethyl acetate, utilizing tertiary amines as scavengers for the generated acid byproducts. The final and perhaps most critical stage involves the conversion of the free base into its stable salt form, where careful control of solvent composition and temperature induces the formation of the desired crystalline polymorph. Detailed standardized synthesis steps for this complex pathway are provided in the guide below to assist technical teams in replicating these results.
- Prepare the key intermediate R-(-)-3-(3-diisopropylamino-phenyl-propyl)-4-hydroxy-benzoic acid methyl ester via catalytic hydrogenation of the benzyloxy precursor using Raney nickel.
- Reduce the ester intermediate to the corresponding phenol alcohol using lithium aluminum hydride or similar reducing agents under inert atmosphere at low temperatures.
- Perform regioselective acylation with isobutyryl chloride followed by salt formation with fumaric acid or hydrochloric acid in specific solvent systems to induce spontaneous crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented technology translates into tangible strategic advantages that extend far beyond simple chemical specifications. The shift from unstable, amorphous materials to robust, crystalline salts fundamentally alters the risk profile of the supply chain by eliminating the variability associated with hygroscopic degradation. This stability ensures that raw materials can be stored for extended periods without special environmental controls, reducing warehousing costs and minimizing the risk of inventory write-offs due to quality failures. Moreover, the high purity achieved through spontaneous crystallization reduces the need for extensive chromatographic purification, which is often a bottleneck in large-scale manufacturing. This streamlining of the production process leads to a drastic simplification of the workflow, allowing for faster turnaround times and more reliable delivery schedules to downstream pharmaceutical partners.
- Cost Reduction in Manufacturing: The elimination of complex purification steps and the ability to use standard solvents for crystallization significantly lower the operational expenditure associated with producing these intermediates. By avoiding the need for expensive transition metal catalysts or energy-intensive drying processes required for amorphous solids, the overall cost of goods sold is substantially reduced. The high yield and selectivity of the acylation step further contribute to cost efficiency by maximizing the utilization of raw materials and minimizing waste generation. These factors combine to create a more economically viable production model that enhances profit margins while maintaining competitive pricing structures for the final API.
- Enhanced Supply Chain Reliability: The inherent stability of the crystalline salts ensures a consistent supply of high-quality intermediates, mitigating the risks of production delays caused by material degradation. Since the compounds do not require stringent moisture control during transport or storage, logistics become simpler and less prone to disruption. This reliability is crucial for maintaining continuous manufacturing lines and meeting the demanding timelines of global pharmaceutical clients. The ability to stockpile stable intermediates without fear of quality loss provides a buffer against market fluctuations and raw material shortages, ensuring business continuity even in volatile supply environments.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing common reagents and solvents that are readily available in bulk quantities for industrial applications. The spontaneous crystallization phenomenon scales linearly from laboratory to commercial production, reducing the technical risks associated with technology transfer. Additionally, the high atom economy and reduced waste generation align with modern green chemistry principles, facilitating compliance with increasingly strict environmental regulations. This eco-friendly profile not only reduces disposal costs but also enhances the corporate sustainability image of the manufacturing entity, appealing to environmentally conscious stakeholders.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation and benefits of this stable salt technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for decision-making. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this pathway into their existing portfolios and for procurement specialists assessing the long-term value proposition.
Q: Why are crystalline salts preferred over amorphous forms for 3,3-diphenylpropylamine derivatives?
A: Crystalline salts offer superior chemical stability, non-hygroscopic properties, and higher melting points compared to amorphous forms, which are prone to degradation and intramolecular ester transfer during storage.
Q: What specific acids are compatible for forming stable salts in this process?
A: The patent highlights physiologically compatible inorganic or organic acids such as fumaric acid, hydrochloric acid, methanesulfonic acid, and various carboxylic acids like citric or tartaric acid.
Q: How does this technology impact the scalability of urology drug manufacturing?
A: The ability to achieve spontaneous crystallization without complex seeding or extreme cooling significantly simplifies downstream processing, reducing production time and enhancing batch-to-batch consistency for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3,3-Diphenylpropylamine Derivatives Supplier
As the demand for high-performance urology medications continues to grow, securing a supply of stable and pure intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging the insights from patent CN1215045C to deliver superior 3,3-diphenylpropylamine derivatives. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements without compromising on quality. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch meets the highest standards of consistency and safety required for clinical and commercial applications.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced crystallization technology can enhance the stability and efficacy of your pharmaceutical products.
