Advanced Silicon-Based Reagents for Scalable Asymmetric Synthesis of Pharmaceutical Intermediates
The chemical industry's relentless pursuit of efficient, high-purity chiral building blocks has led to significant advancements in asymmetric synthesis methodologies. Patent CN1639172A introduces a groundbreaking class of cyclic silicon-based reagents designed specifically for asymmetric allylation, aldol, and tandem reactions. These reagents address critical limitations found in conventional methods, such as reagent instability, toxicity of by-products, and the difficulty of separating reaction products. By utilizing stable, storable cyclic silanes derived from readily available chiral auxiliaries like pseudoephedrine or pinacol, this technology enables the formation of chiral alcohols and hydrazines with exceptional enantioselectivity. For R&D directors and procurement specialists in the pharmaceutical sector, this represents a pivotal shift towards more reliable and scalable processes for generating complex pharmaceutical intermediates. The ability to store these reagents for extended periods without decomposition fundamentally changes the logistics of fine chemical manufacturing, allowing for batch consistency and reduced waste.
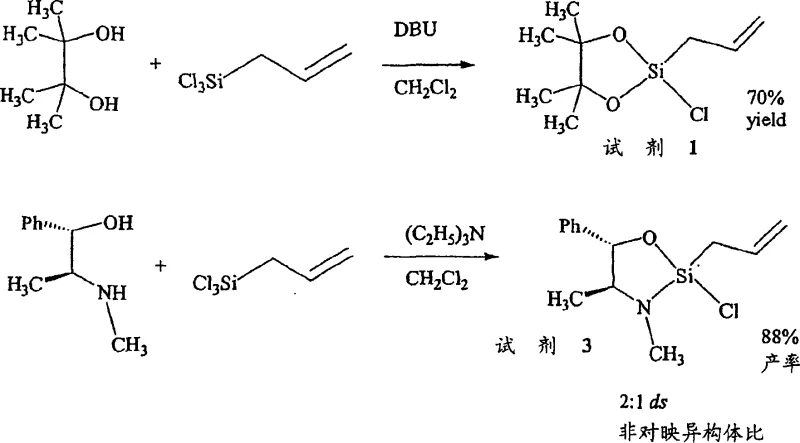
The development of robust synthetic routes for chiral homoallylic alcohols is a cornerstone of modern drug discovery, yet traditional methods often suffer from significant operational drawbacks. Conventional allylation and enolate addition reactions frequently rely on catalysts or reagents that must be prepared in situ and used immediately due to their inherent instability. This requirement for on-site preparation increases operational complexity, introduces variability between batches, and often necessitates the use of toxic heavy metals or difficult-to-remove by-products. Furthermore, the separation of the chiral auxiliary from the final product in older methodologies can be labor-intensive, requiring multiple chromatographic steps that reduce overall yield and increase production costs. These limitations have historically hindered the commercial scale-up of complex pharmaceutical intermediates, forcing manufacturers to accept lower efficiencies or higher safety risks. The inability to store key reagents also creates supply chain vulnerabilities, as any disruption in the immediate preparation workflow can halt production lines entirely.
In stark contrast, the novel approach detailed in the patent utilizes pre-formed, isolable cyclic silane reagents that offer superior stability and handling characteristics. These reagents, such as the oxazasilolidine derivatives, can be distilled and stored for weeks at ambient or reduced temperatures without losing efficacy. This stability allows for quality control testing prior to use, ensuring consistent performance in downstream reactions. The new methodology eliminates the need for transition metal catalysts in many instances, relying instead on the inherent chirality of the silicon-bound auxiliary to direct stereochemistry. This simplifies the reaction profile, reduces the risk of metal contamination in the final API, and streamlines the purification process. By shifting from transient, unstable species to robust, storable reagents, the technology significantly enhances the reliability of cost reduction in pharmaceutical intermediates manufacturing. The process facilitates easier isolation of products through simple aqueous workups, where the chiral auxiliary partitions into the water phase for recovery, leaving the organic product ready for final purification.
Mechanistic Insights into Silicon-Mediated Asymmetric Allylation and Tandem Reactions
The exceptional stereocontrol observed in these reactions stems from the rigid cyclic structure of the silicon reagent, which locks the chiral auxiliary in a specific conformation relative to the reactive allyl group. When the reagent reacts with an electrophilic aldehyde, the silicon center acts as a Lewis acid, coordinating with the carbonyl oxygen to form a highly organized transition state. This coordination minimizes the degrees of freedom for the incoming nucleophile, forcing the addition to occur from a specific face of the carbonyl group. In the case of tandem aldol-allylation reactions, the mechanism becomes even more sophisticated. The reagent first undergoes an aldol-type addition where an enol moiety attacks the aldehyde, forming an intermediate that subsequently undergoes an intramolecular allylation. This cascade process establishes multiple chiral centers in a single operation with high diastereoselectivity. Understanding this mechanistic pathway is crucial for R&D teams aiming to optimize reaction conditions for specific substrates, as the electronic nature of the aldehyde and the steric bulk of the auxiliary directly influence the energy landscape of the transition state.
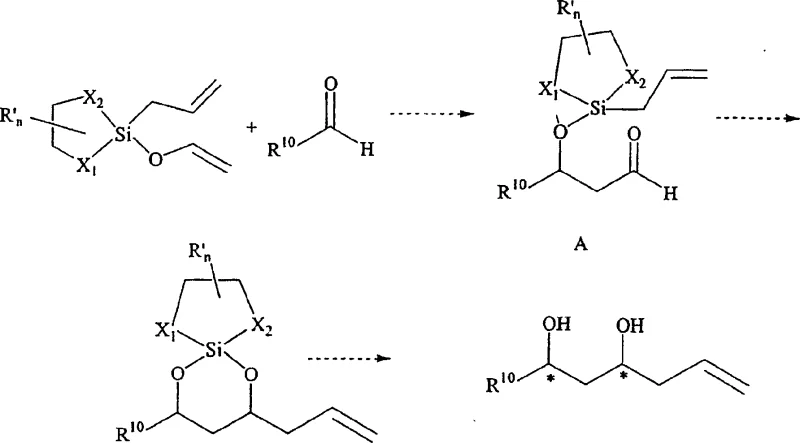
Impurity control is inherently built into the design of these cyclic silane reagents due to their high chemoselectivity and the clean nature of the hydrolysis step. Unlike traditional methods that might generate polymeric by-products or require harsh quenching conditions, the silicon-oxygen bonds in the intermediate are cleaved smoothly under mild acidic conditions. This specificity ensures that side reactions, such as polymerization of the aldehyde or over-alkylation, are minimized. The patent data indicates that by-products are primarily the regenerated chiral auxiliary and silanol derivatives, both of which are easily separated from the desired organic product. For quality assurance teams, this means a cleaner impurity profile in the crude material, reducing the burden on downstream purification units. The ability to predict and control the formation of diastereomers is particularly valuable when synthesizing complex molecules where a specific stereoisomer is required for biological activity. The mechanistic rigidity provided by the five-membered or six-membered silacycles ensures that the high-purity pharmaceutical intermediates produced meet stringent regulatory standards with minimal need for extensive recrystallization or chromatography.
How to Synthesize Chiral Homoallylic Alcohols Efficiently
The synthesis of chiral homoallylic alcohols using these novel reagents follows a streamlined protocol that balances high yield with operational simplicity. The process begins with the preparation of the cyclic silane reagent, typically achieved by reacting allyltrichlorosilane with a chiral diol or aminoalcohol in the presence of a non-nucleophilic base. Once the reagent is isolated and purified, it is reacted with the target aldehyde in a solvent like toluene or dichloromethane at controlled temperatures, often ranging from -78°C to room temperature depending on the substrate reactivity. The detailed standardized synthesis steps for implementing this technology in a GMP environment are outlined below.
- Preparation of the cyclic silane reagent by reacting allyltrichlorosilane with a chiral aminoalcohol or diol auxiliary in the presence of a base like triethylamine or DBU.
- Reaction of the purified silane reagent with the target aldehyde electrophile in a non-polar solvent such as toluene or dichloromethane at controlled low temperatures.
- Quenching the reaction mixture with aqueous acid to hydrolyze the silyl intermediate, followed by extraction and purification to isolate the chiral homoallylic alcohol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this silicon-based reagent technology offers substantial strategic benefits beyond mere chemical efficiency. The primary advantage lies in the decoupling of reagent preparation from the final coupling reaction. Because the cyclic silane reagents are stable and storable, they can be manufactured in large batches during off-peak times and held in inventory, smoothing out production schedules and mitigating the risk of raw material shortages. This inventory capability drastically simplifies logistics, as there is no longer a need to coordinate the simultaneous availability of sensitive catalysts and substrates. Furthermore, the recovery and reuse of the chiral auxiliary, such as pseudoephedrine, represent a significant opportunity for cost reduction in fine chemical manufacturing. Since the auxiliary is not consumed but rather acts as a temporary scaffold, its recovery from the aqueous waste stream allows for multiple cycles of use, effectively amortizing the cost of this expensive starting material over many tons of product. This circular economy approach within the synthesis loop reduces the overall material cost per kilogram of the final intermediate.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the ability to recover chiral auxiliaries lead to significant optimization of the bill of materials. By avoiding the need for specialized metal scavengers and reducing the number of purification steps, the overall processing cost is lowered. The high yields reported in the patent, often exceeding 80% and reaching up to 99% in crude forms, mean that less raw material is wasted, directly improving the gross margin of the manufacturing process. Additionally, the use of common solvents like dichloromethane and toluene, rather than exotic or highly regulated solvents, further contributes to a more economical process footprint.
- Enhanced Supply Chain Reliability: The stability of the reagents at ambient temperatures removes the need for complex cold-chain logistics during storage and transport. This robustness ensures that the supply of key intermediates remains continuous even in the face of logistical disruptions. The ability to produce the reagent independently of the final coupling step allows for a more flexible supply chain model, where reagent stockpiles can be maintained to buffer against fluctuations in aldehyde availability. This reliability is critical for maintaining the production schedules of downstream API manufacturing, preventing costly downtime and ensuring timely delivery to customers.
- Scalability and Environmental Compliance: The process is inherently scalable, as demonstrated by the successful execution of reactions on multi-gram scales in the patent examples without loss of selectivity. The absence of heavy metals simplifies waste treatment and disposal, aligning with increasingly stringent environmental regulations. The aqueous workup generates waste streams that are easier to treat compared to those containing metal residues. This environmental compatibility reduces the regulatory burden and potential liability associated with hazardous waste management, making the process more sustainable and attractive for long-term commercial production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this asymmetric synthesis technology. These answers are derived directly from the experimental data and specifications provided in the patent documentation to ensure accuracy and relevance for technical decision-makers.
Q: What are the stability characteristics of the novel cyclic silane reagents?
A: According to the patent data, reagents such as the oxazasilolidine derivatives are stable compounds that can be stored at or below 25°C for several weeks, such as two months, without significant decomposition, addressing a major limitation of prior art reagents.
Q: How is the chiral auxiliary recovered after the reaction?
A: The process allows for the regeneration of the chiral auxiliary, such as pseudoephedrine, upon addition of aqueous acid during the workup phase. The auxiliary remains in the aqueous phase and can be recovered and reused for preparing additional amounts of the reagent.
Q: What level of enantioselectivity can be achieved with these reagents?
A: The patent examples demonstrate high enantioselectivity, with specific reactions yielding products with enantiomeric excess (ee) values ranging from 81% up to 98%, depending on the specific aldehyde substrate and reagent configuration used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Asymmetric Allylation Reagents Supplier
NINGBO INNO PHARMCHEM stands at the forefront of translating advanced academic research into commercial reality. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the promising results seen in laboratory settings are faithfully reproduced on an industrial scale. Our facility is equipped with rigorous QC labs and stringent purity specifications to guarantee that every batch of chiral intermediates meets the exacting standards required by the global pharmaceutical industry. We understand that the transition from bench-scale chemistry to tonnage production involves unique challenges, and our team of expert engineers is dedicated to optimizing these silicon-mediated processes for maximum efficiency and safety.
We invite you to collaborate with us to leverage this cutting-edge technology for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific molecule. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our capabilities can accelerate your development timeline and reduce your overall manufacturing costs. Let us be your partner in navigating the complexities of asymmetric synthesis and delivering high-quality intermediates for your critical drug candidates.
