Advanced Synthesis of Dihalomethyl Pyrazole Intermediates for Scalable Agrochemical Production
Advanced Synthesis of Dihalomethyl Pyrazole Intermediates for Scalable Agrochemical Production
The development of efficient synthetic routes for heterocyclic compounds is a cornerstone of modern agrochemical innovation, particularly for the production of potent fungicides. Patent CN1871204A introduces a groundbreaking methodology for the preparation of 2-dihaloacyl-3-amino-acrylic acid esters and their subsequent conversion into 3-dihalomethyl-pyrazole-4-carboxylic acid esters. This technology addresses long-standing challenges in the industry, specifically regarding yield optimization, impurity control, and the avoidance of hazardous reagents like phosphorus oxychloride. For R&D directors and procurement specialists seeking reliable agrochemical intermediate suppliers, this patent represents a significant leap forward in process chemistry. By enabling the high-yield production of key precursors under mild conditions, it facilitates the commercial scale-up of complex polymer additives and active pharmaceutical ingredients that rely on similar pyrazole scaffolds. The strategic value of this intellectual property lies in its ability to streamline the supply chain while ensuring the high purity required for downstream biological activity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of trihaloacylated aminoacrylates has been plagued by significant inefficiencies and safety concerns. Traditional pathways often rely on the Vilsmeier reaction, which necessitates the use of phosphorus oxychloride, a corrosive and hazardous reagent that complicates waste management and increases operational costs. Furthermore, alternative methods involving the reaction of chloroacrolein with substituted amines have demonstrated unsatisfactory overall yields on an industrial scale. Another prevalent issue in prior art, such as the processes described in US5,498,624, is the formation of substantial quantities of undesired isomeric pyrazoles during the cyclization step. In protic solvents like ethanol, the ratio of desired to undesired isomers can be as poor as 65:35, necessitating energy-intensive distillation or crystallization steps to isolate the target molecule. Additionally, the instability of dihaloketenes formed during certain acylation attempts leads to polymerization, further degrading product quality and reducing the economic viability of these legacy processes.
The Novel Approach
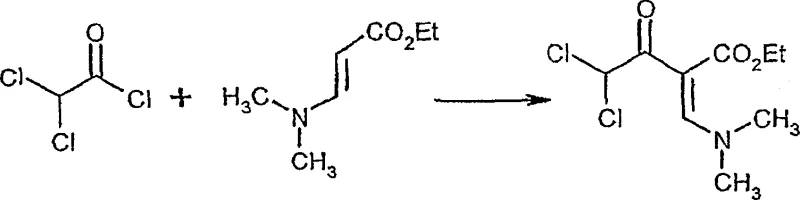
The methodology disclosed in CN1871204A offers a robust solution by reacting acid halides directly with dialkylamino-acrylic acid esters in a water-immiscible organic solvent. This approach eliminates the need for hazardous phosphorus reagents and avoids the instability issues associated with dihaloketene intermediates. A critical innovation is the specific selection of bases; the use of pyridine or pyridine derivatives, rather than trialkylamines like triethylamine, effectively suppresses the elimination of hydrogen chloride and the subsequent formation of unstable dihaloketenes. This results in exceptionally high yields and purity profiles for the 2-dihaloacyl-3-amino-acrylic acid esters. Moreover, the subsequent cyclization step is optimized by employing aprotic solvents and precise temperature control, which drastically minimizes the formation of unwanted isomers. This streamlined two-step sequence not only enhances safety but also provides a clear pathway for cost reduction in fungicide manufacturing by simplifying the purification workflow.
Mechanistic Insights into Base-Mediated Acylation and Regioselective Cyclization
The mechanistic superiority of this process begins with the acylation step, where the choice of base plays a pivotal role in determining the reaction outcome. In conventional acylation reactions involving alpha-hydrogen containing amines, the presence of strong bases like triethylamine often promotes the elimination of hydrogen chloride from the intermediate, leading to the generation of highly reactive dihaloketenes. These ketenes are prone to rapid polymerization, which consumes the starting material and generates difficult-to-remove tarry by-products. The patent elucidates that by utilizing pyridine or its derivatives, the basicity is tuned to facilitate the nucleophilic attack of the aminoacrylate on the acid halide without triggering the elimination pathway. This subtle yet profound adjustment in reaction conditions ensures that the 2-dihaloacyl-3-amino-acrylic acid ester remains stable throughout the reaction, allowing for isolation yields that approach theoretical maximums. The stability of this intermediate is crucial for downstream processing, as it allows for direct use in the next step without extensive purification.
Following the acylation, the cyclization mechanism with hydrazine derivatives is governed by kinetic control to achieve high regioselectivity. The reaction between the 2-dihaloacyl-3-amino-acrylic acid ester and the hydrazine can theoretically produce two isomeric pyrazoles: the desired 3-dihalomethyl isomer and the undesired 5-dihalomethyl isomer. Prior art methods operating in protic solvents or at elevated temperatures favor thermodynamic mixtures rich in the unwanted isomer. However, this invention demonstrates that conducting the reaction in aprotic solvents like toluene at low temperatures, specifically between -50°C and 0°C, kinetically favors the formation of the 3-isomer. The low temperature suppresses the equilibration to the more stable but undesired 5-isomer. Experimental data within the patent shows that optimizing these parameters can shift the isomer ratio from a problematic 68:32 to an impressive 93.8:6.2. This level of control over the impurity profile is essential for meeting the stringent purity specifications required in the production of active fungicidal components.
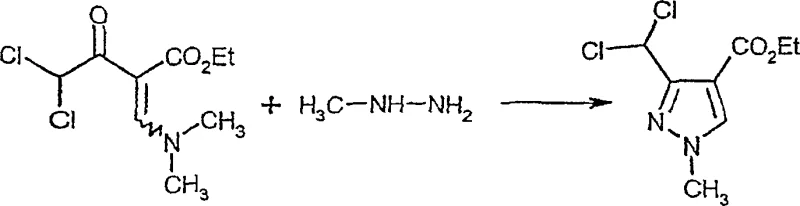
How to Synthesize 3-Dihalomethyl-pyrazole-4-carboxylic Acid Esters Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing these valuable intermediates with high efficiency. The process begins with the preparation of the acyclic precursor through the controlled addition of acid halides to aminoacrylates in a biphasic system, followed by a低温 cyclization step. This sequence is designed to maximize throughput while minimizing waste generation. The detailed standardized synthesis steps below outline the specific operational parameters required to replicate these results in a pilot or production setting. Adhering to these guidelines ensures that the benefits of the novel chemistry, such as improved isomer ratios and simplified workups, are fully realized.
- React acid halides of formula (II) with dialkylamino-acrylic acid esters of formula (III) in a water-immiscible organic solvent like toluene, using a base such as sodium hydroxide or pyridine at temperatures between -20°C and +50°C.
- Isolate the resulting 2-dihaloacyl-3-amino-acrylic acid ester intermediate, optionally drying the organic phase without further purification for immediate use.
- React the intermediate with a hydrazine derivative of formula (IV) in an aprotic solvent at low temperatures (-50°C to 0°C) to achieve high regioselectivity for the 3-dihalomethyl isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical elegance. The primary advantage lies in the substantial cost savings achieved through yield optimization and the elimination of expensive purification steps. By avoiding the formation of polymeric by-products and minimizing isomeric impurities, the process reduces the load on downstream separation units, thereby lowering energy consumption and solvent usage. Furthermore, the use of readily available starting materials such as dichloroacetyl chloride and dimethylaminoacrylates ensures a stable supply chain, reducing the risk of raw material shortages that often plague specialty chemical manufacturing. The robustness of the reaction conditions also translates to enhanced supply chain reliability, as the process is less sensitive to minor fluctuations in operating parameters compared to the finicky Vilsmeier reactions of the past.
- Cost Reduction in Manufacturing: The elimination of phosphorus oxychloride removes the need for specialized corrosion-resistant equipment and complex waste neutralization protocols, leading to significant capital and operational expenditure reductions. Additionally, the high selectivity of the cyclization step means that less product is lost during recrystallization or distillation, directly improving the overall mass balance and reducing the cost per kilogram of the final active ingredient. The ability to use the intermediate crude directly in the next step without drying or further purification further accelerates the production cycle and reduces utility costs.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like toluene, sodium hydroxide, and common acid halides mitigates the risk associated with sourcing exotic or regulated reagents. This accessibility ensures that production schedules can be maintained consistently, even in volatile market conditions. The simplified workup procedure, which relies on standard phase separation techniques, allows for faster batch turnover times, enabling manufacturers to respond more agilely to fluctuating demand from the agrochemical sector without compromising on quality or delivery timelines.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from laboratory benchtop to multi-ton commercial production without the need for re-engineering the core chemistry. The reduction in hazardous waste generation, particularly the avoidance of phosphorus-containing effluents, aligns with increasingly strict environmental regulations globally. This compliance not only avoids potential fines but also enhances the corporate sustainability profile, a key factor for multinational corporations seeking responsible partners for their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation of this patented technology. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on how this method outperforms existing state-of-the-art techniques. Understanding these nuances is critical for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this novel process improve regioselectivity compared to conventional methods?
A: Conventional methods often result in high percentages of unwanted 5-haloalkylpyrazole isomers (up to 88%). This patent demonstrates that by controlling temperature (-50°C to 0°C) and using aprotic solvents, the ratio of desired 3-isomer to unwanted 5-isomer can be optimized to over 93:7, significantly reducing purification costs.
Q: What are the critical reaction conditions for the acylation step?
A: The acylation requires a water-immiscible organic solvent such as toluene or xylene and a base. Crucially, using pyridine or pyridine derivatives instead of triethylamine suppresses the formation of unstable dihaloketenes, leading to higher yields and preventing polymerization side reactions.
Q: Is this process suitable for large-scale industrial manufacturing?
A: Yes, the process avoids the use of hazardous phosphorus oxychloride required in Vilsmeier reactions and utilizes readily available starting materials. The workup involves simple phase separation and distillation, making it highly scalable and economically viable for commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Dihalomethyl-pyrazole-4-carboxylic Acid Esters Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthetic strategies outlined in CN1871204A for the agrochemical industry. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are translated into real-world supply security. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the precise monitoring of isomer ratios critical for fungicide efficacy. We are committed to delivering high-purity agrochemical intermediates that meet the exacting standards of global pharmaceutical and crop protection companies.
We invite you to collaborate with us to leverage this advanced chemistry for your next project. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can optimize your supply chain and reduce your time to market for new active ingredients.
