Advanced Transition Metal Catalysis for High-Purity Benzofuran Pharmaceutical Intermediates
Introduction to Advanced Benzofuran Synthesis
The pharmaceutical industry continuously demands more efficient and scalable routes for complex heterocyclic intermediates, particularly those serving as core structures for oncology and CNS active pharmaceutical ingredients. Patent CN1232519C introduces a sophisticated methodology for producing 5-(1-piperazinyl)-benzofuran-2-carboxamide, a critical building block in modern drug discovery. This technology leverages transition metal-catalyzed amination, specifically utilizing palladium complexes to forge carbon-nitrogen bonds under controlled conditions. Unlike legacy processes that often struggle with low yields and difficult purifications due to side reactions, this patented approach offers a versatile platform that can be adapted for various substituted derivatives. By integrating robust catalytic cycles with strategic protection group chemistry, the process ensures high purity profiles essential for regulatory compliance. For a reliable pharmaceutical intermediate supplier, mastering such catalytic transformations is key to delivering consistent quality.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of N-aryl piperazines attached to benzofuran cores relied heavily on classical nucleophilic aromatic substitution (SNAr). These traditional pathways frequently necessitate extreme reaction conditions, such as very high temperatures and strong bases, which can compromise the integrity of sensitive functional groups like esters or amides present on the molecule. Furthermore, SNAr reactions on electron-rich heterocycles often suffer from sluggish kinetics, resulting in incomplete conversions and complex mixtures of by-products that are arduous to separate. The reliance on stoichiometric amounts of harsh reagents also generates significant waste streams, posing environmental challenges and increasing disposal costs. In many cases, the crude product requires extensive chromatographic purification, which is economically unviable for commercial scale-up of complex pharmaceutical intermediates. These inefficiencies create bottlenecks in the supply chain, leading to longer lead times and higher variability in batch-to-batch quality.
The Novel Approach
The methodology outlined in CN1232519C represents a paradigm shift by employing transition metal catalysis to overcome these kinetic and thermodynamic barriers. The process allows for the amination step to occur either before or after the formation of the benzofuran ring, providing strategic flexibility depending on the specific substitution pattern required. 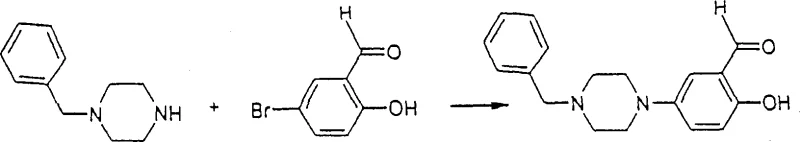 As illustrated in the reaction scheme, the coupling of 5-bromo-salicylaldehyde derivatives with protected piperazines proceeds smoothly using palladium catalysts and specialized ligands. This novel approach operates at moderate temperatures, typically between 60°C and 130°C, significantly reducing energy consumption compared to fusion methods. The use of bulky phosphine ligands enhances the reactivity of the palladium center, facilitating the oxidative addition of the aryl halide and the subsequent reductive elimination to form the C-N bond. This results in cleaner reaction profiles and simplifies the downstream isolation of the target intermediate, thereby enhancing overall process efficiency.
As illustrated in the reaction scheme, the coupling of 5-bromo-salicylaldehyde derivatives with protected piperazines proceeds smoothly using palladium catalysts and specialized ligands. This novel approach operates at moderate temperatures, typically between 60°C and 130°C, significantly reducing energy consumption compared to fusion methods. The use of bulky phosphine ligands enhances the reactivity of the palladium center, facilitating the oxidative addition of the aryl halide and the subsequent reductive elimination to form the C-N bond. This results in cleaner reaction profiles and simplifies the downstream isolation of the target intermediate, thereby enhancing overall process efficiency.
Mechanistic Insights into Pd-Catalyzed Amination
The core of this synthetic strategy lies in the intricate catalytic cycle driven by palladium species. The reaction initiates with the generation of an active Pd(0) species, often derived from precursors like Pd(OAc)2 or Pd(DBA)2 in the presence of reducing agents or excess ligand. 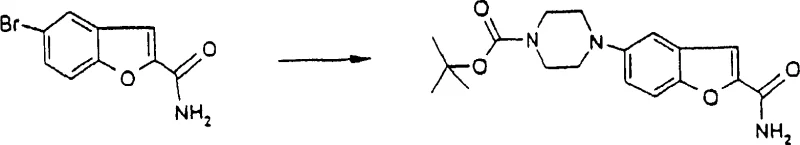 This active catalyst undergoes oxidative addition with the aryl halide substrate, forming an aryl-palladium(II) complex. The choice of ligand is paramount; bulky, electron-rich phosphines such as tri-tert-butylphosphine (P(t-Bu)3) or bidentate ligands like BINAP and DPPF stabilize the palladium center and accelerate the rate-determining steps. Following oxidative addition, the piperazine nitrogen coordinates to the metal center and undergoes deprotonation by a base, such as sodium tert-butoxide, to form an amido-palladium complex. The final step involves reductive elimination, which releases the coupled product and regenerates the Pd(0) catalyst. Understanding this mechanism allows for precise tuning of reaction parameters to minimize side reactions like dehalogenation or homocoupling, ensuring high selectivity.
This active catalyst undergoes oxidative addition with the aryl halide substrate, forming an aryl-palladium(II) complex. The choice of ligand is paramount; bulky, electron-rich phosphines such as tri-tert-butylphosphine (P(t-Bu)3) or bidentate ligands like BINAP and DPPF stabilize the palladium center and accelerate the rate-determining steps. Following oxidative addition, the piperazine nitrogen coordinates to the metal center and undergoes deprotonation by a base, such as sodium tert-butoxide, to form an amido-palladium complex. The final step involves reductive elimination, which releases the coupled product and regenerates the Pd(0) catalyst. Understanding this mechanism allows for precise tuning of reaction parameters to minimize side reactions like dehalogenation or homocoupling, ensuring high selectivity.
Impurity control is another critical aspect addressed by this mechanistic understanding. The patent details how specific reaction conditions mitigate the formation of des-halo by-products, which are common in palladium-catalyzed reactions. By optimizing the ratio of ligand to metal and selecting appropriate solvents like toluene, xylene, or NMP, the process maximizes the turnover number of the catalyst. Furthermore, the use of protected piperazines prevents unwanted side reactions at the secondary amine during the cyclization steps. The subsequent removal of protecting groups, whether via hydrogenolysis for benzyl groups or acidolysis for BOC groups, is designed to be orthogonal and high-yielding. This meticulous control over the chemical pathway ensures that the final 5-(1-piperazinyl)-benzofuran-2-carboxamide meets stringent purity specifications required for GMP manufacturing.
How to Synthesize 5-(1-Piperazinyl)-Benzofuran-2-Carboxamide Efficiently
The synthesis of this valuable intermediate can be achieved through multiple convergent routes, offering flexibility for process chemists to select the most cost-effective pathway based on raw material availability. The patent describes a robust sequence where the amination is performed on a salicylaldehyde derivative, followed by etherification and cyclization to close the furan ring. Alternatively, the benzofuran core can be constructed first, followed by the palladium-catalyzed introduction of the piperazine moiety. Both routes utilize common industrial reagents and avoid exotic or hazardous chemicals, facilitating easier technology transfer. The detailed standardized synthesis steps involve precise control of temperature, stoichiometry, and workup procedures to ensure reproducibility. For a comprehensive guide on executing these reactions at scale, please refer to the technical documentation below.
- Perform transition metal-catalyzed amination of 5-halo-salicylaldehyde derivatives with protected piperazines using Pd catalysts and bulky phosphine ligands.
- Execute etherification with halo-acetates followed by cyclization with formamide or chloroacetamide to form the benzofuran core.
- Remove amino protecting groups (e.g., BOC or Benzyl) via acidolysis or hydrogenolysis to yield the final free base or salt.
Commercial Advantages for Procurement and Supply Chain Teams
Adopting this patented synthesis route offers substantial strategic benefits for procurement and supply chain management within the pharmaceutical sector. The primary advantage lies in the significant reduction of processing complexity, which directly translates to lower manufacturing costs. By eliminating the need for extreme reaction conditions and reducing the number of purification steps, the overall resource intensity of the production process is drastically diminished. This efficiency gain allows for a more competitive pricing structure without compromising on the quality of the high-purity pharmaceutical intermediate. Moreover, the versatility of the catalytic system means that the same equipment train can be utilized for a variety of analogous compounds, maximizing asset utilization and reducing capital expenditure requirements for dedicated production lines.
- Cost Reduction in Manufacturing: The transition metal-catalyzed approach eliminates the need for stoichiometric amounts of expensive copper salts or harsh forcing conditions often associated with traditional Ullmann-type couplings. The ability to use catalytic amounts of palladium, potentially with recovery and recycling protocols, significantly lowers the raw material cost per kilogram. Additionally, the simplified workup procedures, often involving straightforward crystallization rather than column chromatography, reduce solvent consumption and waste disposal fees. These cumulative efficiencies result in substantial cost savings in API manufacturing, making the final drug product more economically viable in competitive markets.
- Enhanced Supply Chain Reliability: The starting materials for this synthesis, such as 5-bromo-salicylaldehyde and various protected piperazines, are commercially available from multiple global vendors, reducing the risk of single-source dependency. The robustness of the reaction conditions ensures high batch-to-batch consistency, minimizing the risk of production delays due to failed batches or out-of-specification results. This reliability is crucial for maintaining continuous supply chains for critical medications. Furthermore, the scalability of the process from laboratory to pilot and commercial plant scales has been demonstrated, ensuring that supply can be ramped up quickly to meet market demand fluctuations without lengthy re-validation periods.
- Scalability and Environmental Compliance: The process is designed with green chemistry principles in mind, utilizing solvents that can be recovered and recycled effectively. The reduction in reaction steps and the avoidance of hazardous reagents contribute to a lower environmental footprint, aligning with increasingly strict global environmental regulations. The one-pot nature of certain variations described in the patent further minimizes solvent usage and energy consumption associated with intermediate isolations. This sustainable approach not only aids in regulatory compliance but also enhances the corporate social responsibility profile of the supply chain, appealing to environmentally conscious stakeholders and partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 5-(1-piperazinyl)-benzofuran-2-carboxamide. These insights are derived directly from the experimental data and claims within the patent literature, providing a factual basis for decision-making. Understanding these nuances helps R&D and procurement teams evaluate the feasibility of integrating this intermediate into their development pipelines. The answers cover catalyst selection, purification strategies, and scalability considerations.
Q: What are the critical catalyst systems for this amination reaction?
A: The patent highlights the efficacy of Palladium sources such as Pd(OAc)2 or Pd(DBA)2 combined with bulky phosphine ligands like tri-tert-butylphosphine (P(t-Bu)3), BINAP, or DPPF to facilitate the coupling of sterically hindered amines.
Q: How does this method improve upon traditional nucleophilic substitution?
A: Traditional methods often require harsh conditions leading to decomposition. This transition metal-catalyzed approach allows for milder temperatures (60°C-130°C) and better tolerance of functional groups, significantly simplifying product isolation and purification.
Q: Can the protecting groups be removed efficiently on a large scale?
A: Yes, the patent describes robust deprotection strategies, including hydrogenolysis for benzyl groups using Pd/C and acidolysis for BOC groups using TFA or HCl, both of which are scalable and utilize common industrial reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-(1-Piperazinyl)-Benzofuran-2-Carboxamide Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of new therapeutic agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 5-(1-piperazinyl)-benzofuran-2-carboxamide meets the highest industry standards. Our commitment to excellence extends beyond mere supply; we act as a strategic partner, offering technical support to optimize your downstream synthesis and formulation processes.
We invite you to contact our technical procurement team to discuss your specific requirements and explore how our advanced manufacturing capabilities can support your pipeline. Request a Customized Cost-Saving Analysis to understand the economic benefits of switching to our optimized synthetic route. We are ready to provide specific COA data and route feasibility assessments to accelerate your project timelines. Let us collaborate to bring your innovative medicines to market faster and more efficiently.
