Advanced MPO Inhibitor Intermediates: Scalable Synthesis for Neuroinflammatory Therapies
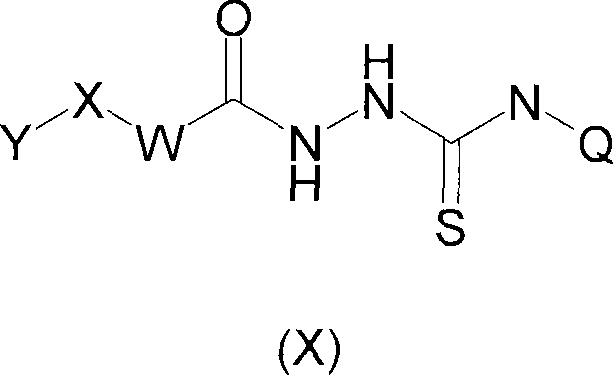
The pharmaceutical landscape is continuously evolving with the discovery of novel therapeutic targets, and patent CN101048392A represents a significant advancement in the field of neuroinflammatory disease treatment. This patent discloses the use of specific 2,4-dihydro-[1,2,4]triazole-3-thione derivatives as potent inhibitors of the enzyme myeloperoxidase (MPO). MPO is a heme-containing enzyme primarily found in polymorphonuclear leukocytes, and its overactivity is strongly implicated in the pathogenesis of various neuroinflammatory disorders including Alzheimer's disease, Parkinson's disease, and multiple sclerosis. The core innovation lies in the structural flexibility of Formula (I), which allows for extensive modification of the Q, X, W, and Y substituents to optimize pharmacokinetic properties and binding affinity. For R&D directors and procurement specialists, understanding the chemical architecture of these intermediates is crucial for developing robust supply chains for next-generation neurological therapeutics. The ability to modulate MPO activity without the severe side effects associated with broad-spectrum anti-inflammatories offers a compelling value proposition for drug development pipelines focused on chronic neurodegenerative conditions.
![General Formula I of 2,4-dihydro-[1,2,4]triazole-3-thione derivatives showing variable substituents Q, X, W, and Y for MPO inhibition](/insights/img/mpo-inhibitor-triazole-thione-pharma-supplier-20260313073645-01.png)
The development of effective MPO inhibitors has historically faced challenges related to selectivity and metabolic stability. Conventional methods for treating inflammatory conditions often rely on non-steroidal anti-inflammatory drugs (NSAIDs) or corticosteroids, which can lead to significant gastrointestinal and cardiovascular adverse events when used long-term. Furthermore, earlier generations of peroxidase inhibitors often lacked the necessary specificity, leading to off-target effects that compromised patient safety. The novel approach detailed in this patent utilizes a triazole-thione scaffold that demonstrates unexpectedly potent properties as an MPO inhibitor. This scaffold provides a rigid structural framework that facilitates precise interaction with the MPO active site, potentially reducing the required dosage and minimizing systemic exposure. By focusing on the thione functionality at the 3-position of the triazole ring, the chemistry enables strong coordination with the heme iron of the enzyme, thereby effectively blocking the oxidation of chloride to hypochlorous acid which is a key mediator of tissue damage in neuroinflammation.
The structural versatility of the novel approach allows for the incorporation of diverse cyclic and acyclic groups at the Q position, ranging from saturated heterocycles to benzo-fused cycloalkyl systems. This modularity is critical for medicinal chemists aiming to fine-tune the lipophilicity and solubility profiles of the final drug candidate. For instance, the inclusion of pyridyl or phenyl groups at the Y position can enhance pi-stacking interactions within the enzyme pocket, while variations in the X and W linkers provide control over the spatial orientation of the pharmacophore. Such detailed structure-activity relationship (SAR) opportunities mean that the synthesis process must be highly adaptable to accommodate different building blocks without requiring complete process redevelopment. This flexibility is a key advantage for contract development and manufacturing organizations (CDMOs) looking to support clients through the lead optimization phase and into clinical trials, ensuring that the manufacturing route remains viable as the molecule evolves.
Mechanistic Insights into MPO Inhibition and Triazole-Thione Cyclization
The mechanism of action for these compounds involves the direct inhibition of myeloperoxidase, preventing the formation of reactive oxygen species that contribute to neuronal damage. From a synthetic chemistry perspective, the formation of the triazole-thione ring is a critical step that determines the overall yield and purity of the intermediate. The patent outlines several pathways, including the reaction of thiosemicarbazide derivatives with esters, acid chlorides, or isothiocyanates. A key intermediate in this process is the acyl thiosemicarbazide species, which undergoes cyclization to form the heterocyclic core. Understanding the stability and reactivity of this intermediate is essential for process chemists, as it dictates the choice of solvents, bases, and reaction temperatures. For example, the use of sodium methoxide in methanol promotes cyclization under mild conditions, whereas coupling agents like EDC may be required for more sterically hindered substrates. This mechanistic understanding allows for the prediction of potential impurities and the design of effective purification strategies.
Impurity control is another vital aspect of the mechanistic profile, particularly given the intended use of these compounds in pharmaceuticals. The cyclization step can potentially generate regioisomers or incomplete reaction products if not carefully monitored. The patent suggests that depending on the conditions and the nature of the substituents, the intermediates can be isolated or subjected to in situ cyclization. In situ processing can reduce the number of unit operations and minimize the exposure of reactive intermediates to moisture or oxygen, thereby improving overall process robustness. However, isolation may be necessary to ensure high purity before the final ring closure, especially when dealing with complex substituents on the Q ring. The ability to purify intermediates via crystallization or chromatography provides an additional layer of quality control, ensuring that the final API intermediate meets stringent regulatory specifications. This level of control is paramount for supply chain heads who must guarantee batch-to-batch consistency for clinical and commercial production.
How to Synthesize 2,4-Dihydro-[1,2,4]triazole-3-thione Derivatives Efficiently
The synthesis of these high-value pharmaceutical intermediates can be achieved through multiple validated routes, offering flexibility based on raw material availability and equipment constraints. The patent describes general methods involving the condensation of hydrazides with isothiocyanates or the reaction of thiosemicarbazides with activated carboxylic acid derivatives. These methods are scalable and utilize common laboratory and plant equipment, making them suitable for technology transfer from R&D to manufacturing. The choice of route often depends on the specific substitution pattern required for the target molecule, with some pathways offering better atom economy or milder reaction conditions. Detailed standard operating procedures for these synthesis steps are critical for ensuring reproducibility and safety during scale-up operations.
- Prepare thiosemicarbazide derivatives and react with appropriate esters or acid chlorides in organic solvents.
- Facilitate cyclization under basic conditions or using coupling agents like EDC to form the triazole ring.
- Purify the final 2,4-dihydro-[1,2,4]triazole-3-thione product via crystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the synthetic routes described in patent CN101048392A offer significant advantages in terms of cost reduction and supply chain reliability. The avoidance of precious metal catalysts, which are often required in cross-coupling reactions for similar heterocyclic systems, eliminates the need for expensive metal scavenging steps and reduces the risk of heavy metal contamination in the final product. This simplification of the downstream processing directly translates to lower manufacturing costs and shorter production cycles. Furthermore, the starting materials, such as substituted hydrazides and isothiocyanates, are often commercially available or can be synthesized from commodity chemicals, reducing dependency on specialized suppliers and mitigating supply chain risks. This accessibility is crucial for procurement managers looking to secure long-term contracts for clinical and commercial supply.
- Cost Reduction in Manufacturing: The synthetic methodology relies on standard organic transformations that do not require exotic reagents or extreme reaction conditions. By utilizing common solvents like methanol, ethanol, and dichloromethane, the process minimizes solvent recovery costs and waste disposal expenses. The elimination of transition metal catalysts not only reduces raw material costs but also simplifies the regulatory filing process by removing the need for extensive residual metal testing. This streamlined approach allows for substantial cost savings in the overall manufacturing budget, making the development of MPO inhibitor therapies more economically viable for pharmaceutical companies.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for the sourcing of building blocks from multiple vendors, reducing the risk of supply disruptions. Since the core chemistry is based on well-established reactions, there is a lower risk of technical failure during scale-up compared to novel biocatalytic or photochemical processes. This reliability ensures that production timelines can be met consistently, which is critical for maintaining clinical trial schedules and meeting market demand upon product launch. Procurement teams can leverage this flexibility to negotiate better terms with suppliers and build a resilient supply network.
- Scalability and Environmental Compliance: The processes described are amenable to large-scale production, with reaction conditions that can be safely managed in standard chemical reactors. The waste streams generated are primarily organic solvents and salts, which can be treated using conventional waste management protocols. This compliance with environmental regulations reduces the burden on EHS teams and minimizes the risk of regulatory penalties. Additionally, the high yields and purity achievable with these methods reduce the volume of waste generated per kilogram of product, contributing to a more sustainable manufacturing footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these MPO inhibitor intermediates. The answers are derived from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry stakeholders. Understanding these details helps in making informed decisions regarding process development and sourcing strategies.
Q: What is the primary therapeutic application of these triazole-thione derivatives?
A: These compounds are designed as inhibitors of the enzyme myeloperoxidase (MPO), making them highly relevant for treating neuroinflammatory disorders such as Alzheimer's disease, Parkinson's disease, and multiple sclerosis.
Q: Are the starting materials for this synthesis commercially available?
A: Yes, the patent indicates that many starting materials like thiosemicarbazide derivatives and specific esters are known in literature or can be prepared using standard methods, ensuring supply chain reliability.
Q: How does this synthesis route impact manufacturing costs?
A: The synthetic methods described avoid the use of expensive transition metal catalysts and utilize standard organic solvents and reagents, which significantly reduces raw material costs and simplifies waste treatment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,4-Dihydro-[1,2,4]triazole-3-thione Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from laboratory bench to industrial plant. Our technical team is well-versed in the complexities of heterocyclic chemistry and can optimize the synthesis of 2,4-dihydro-[1,2,4]triazole-3-thione derivatives to meet your specific purity and cost targets. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate complies with global regulatory standards. Our commitment to quality and reliability makes us an ideal partner for pharmaceutical companies seeking to advance their neuroinflammatory drug candidates.
We invite you to contact our technical procurement team to request specific COA data and route feasibility assessments tailored to your project needs. By collaborating with us, you can benefit from a Customized Cost-Saving Analysis that identifies opportunities to optimize your supply chain and reduce overall development costs. Let us help you accelerate your drug development timeline with our proven expertise in fine chemical manufacturing and supply chain management.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
