Advanced Aryl Ether Preparation for Commercial Scale Pharmaceutical Intermediates
Advanced Aryl Ether Preparation for Commercial Scale Pharmaceutical Intermediates
The pharmaceutical industry continuously seeks robust synthetic routes for high-value antidepressant intermediates, and patent CN1636988A presents a transformative approach to preparing aryl ethers useful as active pharmaceutical ingredients. This specific intellectual property outlines a refined methodology for synthesizing compounds of formula (A), notably including the well-known antidepressant Reboxetine, by addressing critical bottlenecks in yield, reagent cost, and purification efficiency that have historically plagued large-scale production. By shifting away from expensive solid oxidants and carcinogenic solvents, this process establishes a new benchmark for cost reduction in API manufacturing while ensuring the high-purity aryl ether standards required by global regulatory bodies. The strategic implementation of selective protection groups and phase transfer catalysis demonstrates a deep understanding of process chemistry, offering a viable pathway for reliable pharmaceutical intermediate supplier partnerships aiming to optimize their supply chains. This report analyzes the technical merits and commercial implications of adopting this improved synthesis route for complex morpholine derivatives.
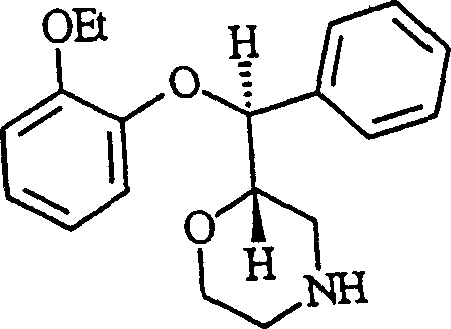
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of aryl ether intermediates like Reboxetine has been hindered by the reliance on inefficient reagents and hazardous solvents that complicate scale-up and drive up operational expenditures. Conventional protocols often utilize meta-chloroperbenzoic acid (m-CPBA) for epoxidation, which is not only significantly more expensive than liquid alternatives but also generates substantial solid waste that requires energy-intensive filtration steps. Furthermore, older methods frequently employ dioxane as a reaction solvent for cyclization steps, a known carcinogen with a high boiling point and freezing point that makes recovery difficult and poses serious safety risks during distillation on a multi-kilogram scale. The lack of selectivity in protecting group chemistry in prior art often leads to the formation of unwanted secondary alcohol esters, with impurity levels reaching up to 13 percent, necessitating complex and yield-reducing purification procedures to meet stringent pharmaceutical specifications. These cumulative inefficiencies result in prolonged production times and increased environmental burdens, making traditional routes economically unviable for modern cost-sensitive supply chains.
The Novel Approach
The innovative process described in the patent data overcomes these historical challenges by introducing a series of chemically elegant and operationally simple modifications that drastically streamline the manufacturing workflow. By substituting solid m-CPBA with liquid peracetic acid for the initial epoxidation of trans-cinnamyl alcohols, the method eliminates the need for filtering solid by-products and reduces reagent costs while maintaining high reaction efficiency at moderate temperatures. The replacement of dioxane with a toluene and water system under phase transfer catalysis conditions allows for rapid cyclization reactions that complete in approximately 45 minutes, with the organic solvent being easily separated and recovered without the risk of freezing in equipment. Crucially, the introduction of selective silylation using chlorotrimethylsilane at low temperatures ensures that only the primary alcohol is protected, virtually eliminating the formation of secondary alcohol ester impurities that plagued previous esterification-based strategies. This holistic optimization of reagents, solvents, and reaction conditions creates a robust platform for the commercial scale-up of complex morpholine derivatives with superior economic and environmental profiles.
Mechanistic Insights into Selective Silylation and Epoxide Ring Opening
The core chemical innovation of this synthesis lies in the precise control of stereochemistry and functional group reactivity during the intermediate stages, particularly in the protection of the diol intermediate formed after epoxide ring opening. In conventional esterification approaches, the reactivity difference between primary and secondary hydroxyl groups is often insufficient to prevent the acylation of the secondary alcohol, leading to diastereomeric impurities that are difficult to separate. The novel method leverages the steric bulk of silylating agents like chlorotrimethylsilane combined with strict temperature control between minus 15 and minus 25 degrees Celsius to achieve near-perfect selectivity for the primary hydroxyl group. This selective protection is critical because it prevents the formation of sulfonate esters on the secondary alcohol in subsequent steps, which would otherwise lead to the generation of unwanted diastereomers during the cyclization to the epoxide. The mechanism ensures that the stereochemical integrity of the molecule is preserved throughout the sequence, resulting in a final product with a significantly cleaner impurity profile and higher overall yield.
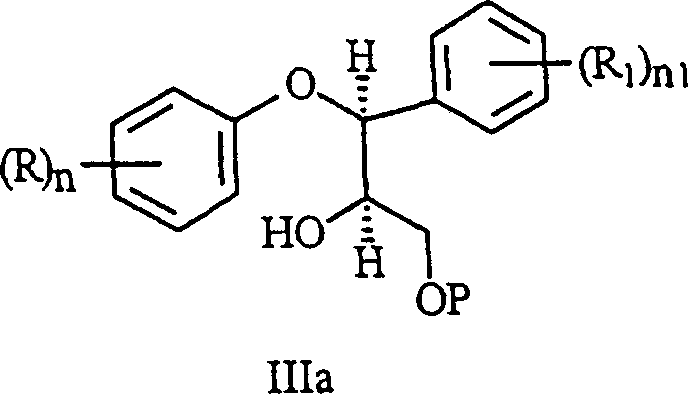
Furthermore, the final reduction step of the morpholinone intermediate to the target morpholine is optimized to minimize the formation of dimeric impurities that typically arise from over-reaction or incomplete reduction. The patent data highlights that using a stoichiometric excess of reducing agent, specifically at least 4 equivalents of sodium bis(2-methoxyethoxy)aluminum hydride, drives the reaction to completion while suppressing side reactions that generate difficult-to-remove by-products. In previous methods, the presence of these impurities often necessitated a controlled pH extraction step that resulted in a product loss of 20 to 30 percent, a significant economic drain in large-scale manufacturing. By adjusting the stoichiometry and addition order, the new process directly produces the free base with less than 0.1 percent of the critical dimeric impurity, thereby eliminating the need for the yield-reducing extraction step. This mechanistic refinement not only improves the mass balance of the process but also simplifies the downstream processing requirements, making it highly attractive for high-purity antidepressant intermediates production.
How to Synthesize Reboxetine Efficiently
The synthesis of this high-value aryl ether involves a multi-step sequence that begins with the epoxidation of substituted trans-cinnamyl alcohols and proceeds through selective protection, cyclization, and reduction to yield the final morpholine structure. Each step has been engineered to maximize throughput and minimize waste, starting with the use of peracetic acid in dichloromethane to generate the initial epoxide intermediate under mild conditions. The subsequent ring opening with substituted phenols is conducted under phase transfer conditions to ensure high conversion rates, followed by the critical low-temperature silylation that sets the stage for high-purity cyclization. The detailed standardized synthesis steps see the guide below for specific reaction parameters and workup procedures that ensure reproducibility and safety.
- Oxidize substituted trans-cinnamyl alcohols using peracetic acid to form intermediate epoxides efficiently.
- React the epoxide with substituted phenols under phase transfer conditions to generate diols with high stereocontrol.
- Protect primary alcohols selectively using chlorotrimethylsilane at low temperatures to prevent secondary alcohol esterification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented process translates into tangible benefits regarding cost stability, lead time reduction, and regulatory compliance across the global manufacturing network. The shift from expensive solid oxidants to commodity liquid reagents like peracetic acid significantly lowers the raw material cost base, while the elimination of difficult solvent removal steps reduces energy consumption and processing time per batch. By avoiding the use of carcinogenic solvents like dioxane, the process simplifies environmental health and safety protocols, reducing the regulatory burden and potential liability associated with hazardous waste disposal in various international jurisdictions. The improved impurity profile means less material is lost during purification, effectively increasing the throughput of existing manufacturing assets without the need for capital investment in new equipment. These factors combine to create a more resilient supply chain capable of meeting fluctuating demand for high-quality pharmaceutical intermediates with greater flexibility and lower total cost of ownership.
- Cost Reduction in Manufacturing: The replacement of high-cost reagents such as m-CPBA with economically favorable alternatives like peracetic acid directly reduces the bill of materials for each production batch. Additionally, the selective silylation strategy prevents the formation of impurities that previously required extensive chromatographic purification or recrystallization, thereby saving on solvent usage and labor costs associated with complex workups. The elimination of the yield-reducing pH extraction step in the final reduction phase further enhances the overall mass efficiency, ensuring that more of the starting material is converted into saleable product. These cumulative savings contribute to substantial cost savings in API manufacturing without compromising on the quality or purity of the final active ingredient.
- Enhanced Supply Chain Reliability: Utilizing liquid reagents that are readily available in bulk quantities reduces the risk of supply disruptions compared to specialized solid oxidants that may have longer lead times or limited supplier bases. The simplified solvent system, which avoids high-freezing-point solvents like dioxane, ensures that production can continue uninterrupted even in varying climatic conditions, preventing equipment damage or process delays due to solvent solidification. The robustness of the phase transfer catalysis system allows for consistent reaction times and yields, enabling more accurate production planning and inventory management for reliable pharmaceutical intermediate supplier operations. This reliability is crucial for maintaining continuous supply to downstream drug product manufacturers who depend on just-in-time delivery models.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing reaction conditions that are easily managed in large reactors without exothermic runaways or difficult heat transfer issues. The reduction in solid waste generation from filtration steps and the avoidance of carcinogenic solvents align with green chemistry principles, facilitating easier permitting and compliance with increasingly strict environmental regulations worldwide. The ability to produce high-purity intermediates with minimal impurity load reduces the environmental impact of waste streams, as less solvent is required for purification and less hazardous waste is generated for disposal. This sustainability profile enhances the corporate social responsibility standing of the manufacturing entity and future-proofs the supply chain against tightening environmental legislation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved aryl ether synthesis route, based on the specific advantages and data points outlined in the patent documentation. These answers are derived from the comparative analysis of the novel method against prior art, focusing on the practical implications for process development and manufacturing teams. Understanding these details is essential for evaluating the feasibility of technology transfer and the potential return on investment for adopting this new chemistry.
Q: How does the new process improve impurity profiles compared to conventional methods?
A: The novel method utilizes selective silylation to prevent secondary alcohol esterification, reducing impurity formation significantly compared to the up to 13 percent impurity levels seen in older esterification protocols.
Q: What are the solvent advantages in the cyclization step?
A: The process replaces carcinogenic dioxane with a toluene and water mixture under phase transfer conditions, facilitating easier solvent removal and enhancing environmental compliance.
Q: Is this method suitable for industrial scale production?
A: Yes, the use of liquid peracetic acid instead of solid m-CPBA and the elimination of difficult filtration steps make this route highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Reboxetine Supplier
As a leading CDMO expert, NINGBO INNO PHARMCHEM possesses the technical capability to translate this sophisticated patent chemistry into a robust commercial manufacturing process that meets the rigorous demands of the global pharmaceutical market. Our facilities are equipped to handle the specific temperature controls and reagent handling requirements necessary for the selective silylation and phase transfer steps, ensuring that the theoretical benefits of the patent are fully realized in production. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, with a commitment to maintaining stringent purity specifications and operating rigorous QC labs to verify every batch. Our team understands the critical nature of impurity control in antidepressant intermediates and is prepared to implement the advanced reduction protocols that eliminate dimeric by-products effectively.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can be integrated into your supply chain to achieve significant operational efficiencies. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic impact of switching to this improved method for your specific volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments that demonstrate our capability to deliver high-quality aryl ethers consistently. Let us help you reduce lead time for high-purity aryl ethers and secure a competitive advantage in the marketplace through superior process chemistry.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
