Advanced One-Pot Synthesis of Isocoumarin-3-yl-acetic Acid Derivatives for Pharmaceutical Applications
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for complex heterocyclic intermediates, particularly those serving as core structures for bioactive molecules. Patent CN1214021C, titled "Process for producing isocoumarins and intermediates for same," presents a significant technological advancement in the synthesis of isocoumarin-3-yl-acetic acid derivatives. These compounds are critical precursors in the development of agents capable of effectively preventing and treating abnormal immune regulation or post-angiogenesis diseases. The core innovation lies in a novel condensation strategy that reacts a homophthalic acid derivative, represented by formula (III), with a malonic acid derivative, represented by formula (IV). This approach not only streamlines the production workflow but also allows for greater structural diversity at the 2-position of the acetic acid chain, which is crucial for tuning biological activity. By leveraging this patented methodology, manufacturers can access a versatile library of isocoumarin scaffolds with improved efficiency.
Historically, the synthesis of isocoumarin derivatives has relied on multi-step sequences that often suffer from low overall yields and limited flexibility regarding substituent introduction. Conventional methods, such as those referenced in international patent WO97/48693, typically start from pre-formed isocoumarin cores like 8-hydroxy-3-methyl-6-methoxy-isocoumarin. While these established routes can provide good yields at individual steps, they are inherently rigid; modifying the substitution pattern on the acetic acid side chain often requires restarting the synthesis from different, sometimes inaccessible, starting materials. Furthermore, the sequential nature of these traditional processes increases the accumulation of impurities and necessitates multiple isolation and purification stages, which drives up both production costs and environmental waste. The inability to easily introduce various functional groups at the 2-position of the acetic acid chain limits the medicinal chemist's ability to optimize the pharmacokinetic and pharmacodynamic profiles of the final drug candidates.
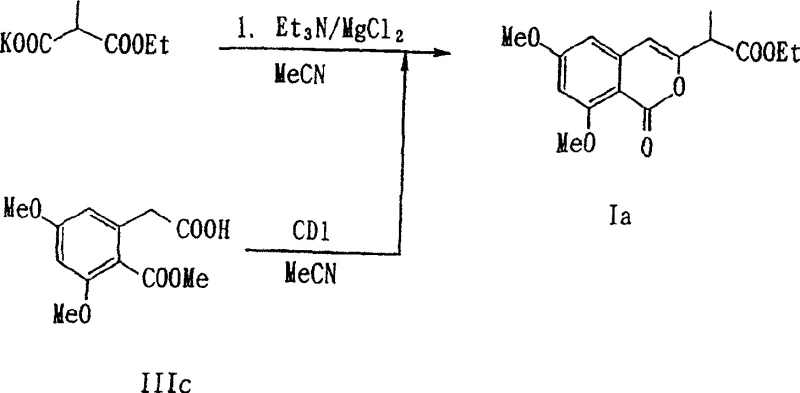
In stark contrast, the novel approach disclosed in CN1214021C utilizes a direct ring condensation reaction that can be executed as a one-pot process. This method involves reacting specific homophthalic acid derivatives with malonic acid derivatives in the presence of a condensing agent within an inert organic solvent. The brilliance of this strategy is its modularity; by simply varying the R group on the malonic acid derivative, manufacturers can efficiently generate a wide array of isocoumarin-3-yl-acetic acid derivatives with different substituents at the biologically critical 2-position. Additionally, the process allows for the optional isolation of the intermediate β-oxycarboxylic acid derivative (Formula II), providing process engineers with the flexibility to optimize the cyclization step independently if necessary. This level of control over the reaction pathway ensures higher purity profiles and facilitates the rapid scale-up of diverse analogues for structure-activity relationship (SAR) studies.
Mechanistic Insights into Condensation and Cyclization
The mechanistic pathway of this synthesis is driven by the activation of the carboxylic acid moiety on the homophthalic acid derivative. Typically, a condensing agent such as carbonyldiimidazole (CDI) is employed to activate the carboxyl group, forming a reactive acyl imidazole intermediate in situ. Simultaneously, the malonic acid derivative, often used as an alkali metal salt (e.g., potassium salt), acts as a nucleophile. The reaction proceeds through a nucleophilic attack on the activated carbonyl carbon, leading to the formation of a β-keto ester or β-oxycarboxylic acid intermediate (Formula II). This step is highly sensitive to reaction conditions, with the patent specifying temperatures ranging from 0°C to 40°C for the initial mixing to prevent side reactions. The presence of additives like magnesium chloride (MgCl2) plays a pivotal role in stabilizing the enolate species and facilitating the subsequent cyclization. Following the formation of the linear intermediate, the system undergoes an intramolecular cyclization, often promoted by bases such as triethylamine, potassium tert-butoxide, or sodium hydride. This cyclization closes the lactone ring to form the isocoumarin core. The ability to perform this sequence in a single vessel (one-pot) minimizes the exposure of reactive intermediates to atmospheric moisture and reduces the handling of hazardous materials, thereby enhancing operational safety and reproducibility.
Impurity control is another critical aspect addressed by the mechanistic design of this process. The patent highlights a specific challenge in the deprotection of the 8-position alkoxy group (R2), where conventional methods using boron tribromide (BBr3) can be too violent, leading to the elimination of other protecting groups or degradation of the sensitive isocoumarin ring. Conversely, using magnesium iodide (MgI2) offers better selectivity but is prohibitively expensive for large-scale manufacturing. The inventors discovered that a combination of an alkali metal iodide (such as potassium iodide) and a magnesium halide (such as magnesium chloride) in a solvent like THF or acetonitrile at moderate temperatures (60-80°C) achieves quantitative deprotection. This specific reagent system selectively cleaves the methyl ether at the 8-position while leaving other protecting groups, such as the 6-methoxy group or ester functionalities, intact. This selective deprotection mechanism is vital for maintaining the integrity of the molecule and ensuring that the final API intermediate meets stringent purity specifications without requiring complex chromatographic separations.
How to Synthesize Isocoumarin-3-yl-acetic Acid Derivatives Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for producing high-purity isocoumarin intermediates suitable for pharmaceutical development. The process begins with the preparation of the requisite homophthalic acid monoester and the malonic acid derivative salt. These components are then combined in a dry, inert solvent system under a nitrogen atmosphere to exclude moisture. The reaction is initiated by the addition of a condensing agent, followed by controlled heating to drive the cyclization to completion. Workup procedures involve standard aqueous extractions and acidification to isolate the product, often yielding crystalline solids that can be further purified by recrystallization. This straightforward operational sequence makes the technology highly attractive for contract development and manufacturing organizations (CDMOs) looking to offer reliable pharmaceutical intermediate supplier services. For detailed procedural specifics regarding stoichiometry, temperature profiles, and workup parameters, please refer to the standardized guide below.
- Prepare a homophthalic acid derivative (Formula III) and a malonic acid derivative (Formula IV) in an inert organic solvent such as THF or acetonitrile.
- React the mixture in the presence of a condensing agent like carbonyldiimidazole (CDI) and a base such as triethylamine or potassium tert-butoxide.
- Perform a one-pot cyclization reaction, optionally heating to reflux, followed by selective deprotection using magnesium chloride and potassium iodide to yield the final isocoumarin derivative.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of the synthesis method described in CN1214021C offers substantial strategic benefits for procurement managers and supply chain directors. The transition from multi-step linear syntheses to a convergent one-pot condensation strategy fundamentally alters the cost structure of manufacturing these complex intermediates. By reducing the number of unit operations, the process significantly lowers labor costs, energy consumption, and solvent usage. Furthermore, the elimination of expensive and specialized deprotection reagents like magnesium iodide in favor of commodity chemicals like potassium iodide and magnesium chloride results in drastic raw material cost savings. This cost reduction in API manufacturing is not merely theoretical; it is derived from the tangible simplification of the chemical workflow, which reduces the burden on waste treatment facilities and shortens the overall production cycle time. Consequently, companies adopting this technology can achieve a more competitive pricing structure while maintaining healthy margins.
Supply chain reliability is another major advantage conferred by this technology. The starting materials, specifically homophthalic acid derivatives and malonic acid derivatives, are readily available from bulk chemical suppliers, reducing the risk of raw material shortages that often plague specialized synthetic routes. The robustness of the one-pot reaction conditions, which tolerate a range of temperatures and solvent systems (THF, acetonitrile, DMF), ensures consistent batch-to-batch quality even when scaling up from pilot plants to commercial production. This scalability is essential for reducing lead time for high-purity pharmaceutical intermediates, allowing drug developers to accelerate their clinical timelines without being bottlenecked by supply constraints. Additionally, the simplified purification protocols, which often rely on crystallization rather than column chromatography, enhance the throughput capacity of manufacturing facilities, ensuring a steady flow of materials to meet global demand.
Environmental compliance and sustainability are increasingly critical factors in vendor selection, and this process aligns well with green chemistry principles. The reduction in solvent volume and the avoidance of hazardous reagents like boron tribromide minimize the generation of toxic waste streams. The use of safer deprotection conditions also lowers the risk of workplace exposure incidents, contributing to a safer operational environment. These factors collectively support the commercial scale-up of complex pharmaceutical intermediates in a manner that is both economically viable and environmentally responsible. For organizations prioritizing ESG (Environmental, Social, and Governance) goals, partnering with a manufacturer utilizing this patented process demonstrates a commitment to sustainable innovation and regulatory compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of isocoumarin derivatives via this patented route. These insights are derived directly from the experimental data and claims within CN1214021C, providing clarity on the feasibility and advantages of the technology. Understanding these details is crucial for R&D teams evaluating the route for new drug candidates and for procurement teams assessing supplier capabilities. The answers below reflect the specific chemical innovations regarding condensation efficiency and selective deprotection that distinguish this method from prior art.
Q: What is the primary advantage of the process described in CN1214021C?
A: The primary advantage is the ability to perform the ring condensation reaction of homophthalic acid derivatives with malonic acid derivatives in a one-pot reaction, significantly simplifying the synthesis of various isocoumarin-3-yl-acetic acid derivatives compared to conventional multi-step methods.
Q: How does this method handle protecting group removal efficiently?
A: The patent discloses a cost-effective deprotection method using a combination of alkali metal iodides (like potassium iodide) and magnesium halides (like magnesium chloride) at moderate temperatures (60-80°C), avoiding the use of expensive reagents like magnesium iodide or harsh conditions associated with boron tribromide.
Q: What are the potential biological applications of these compounds?
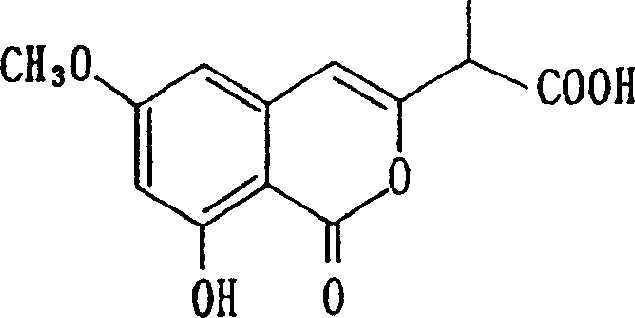
A: The resulting isocoumarin-3-yl-acetic acid derivatives are effective for the prevention and treatment of diseases related to abnormal immunomodulatory effects or post-angiogenesis, similar to known compounds like 2-(8-hydroxy-6-methoxy-1-oxo-1H-2-benzopyran-3-yl)propionic acid.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Isocoumarin-3-yl-acetic Acid Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the synthesis technology detailed in CN1214021C for the development of next-generation immunomodulatory and anti-angiogenic therapies. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project can seamlessly transition from laboratory discovery to global market supply. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications required for clinical and commercial grade intermediates. We understand that the consistency and quality of the isocoumarin core are paramount to the efficacy of the final drug product, and our process engineering team is dedicated to optimizing every parameter of this condensation and cyclization sequence to maximize yield and minimize impurities.
We invite you to engage with our technical procurement team to discuss how we can support your specific supply chain needs. Whether you require a Customized Cost-Saving Analysis for your existing routes or need to validate the feasibility of this one-pot synthesis for your pipeline, we are ready to assist. Please contact us to request specific COA data and route feasibility assessments tailored to your project milestones. By leveraging our expertise in complex heterocyclic synthesis, we can help you secure a reliable supply of high-quality isocoumarin intermediates, accelerating your path to commercialization.
