Advanced Catalytic Synthesis of Metabolically Stable Beta-Carboline Derivatives for Psychotropic Applications
Advanced Catalytic Synthesis of Metabolically Stable Beta-Carboline Derivatives for Psychotropic Applications
The pharmaceutical industry continuously seeks novel scaffolds that offer improved pharmacokinetic profiles, particularly for central nervous system (CNS) agents where metabolic stability is paramount. Patent CN1033391C presents a significant technological breakthrough in the preparation of new beta-carboline derivatives, specifically addressing the historical limitations of metabolic instability found in earlier generations of chlordiazepoxide-receptor ligands. This intellectual property outlines a versatile suite of synthetic methodologies, ranging from transition metal-catalyzed cross-couplings to innovative cycloaddition strategies, enabling the precise installation of aryl, aralkyl, and alkynyl substituents on the A-ring of the beta-carboline core. By leveraging these advanced catalytic protocols, manufacturers can access a diverse library of high-purity intermediates that exhibit superior binding affinity and metabolic resistance, positioning them as ideal candidates for next-generation anxiolytic and psychotropic drug development.
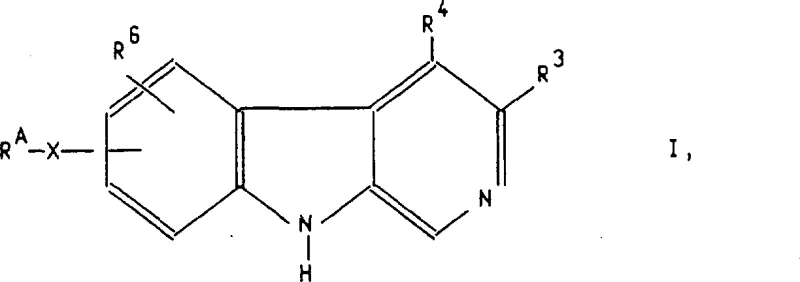
For procurement specialists and supply chain directors evaluating reliable pharmaceutical intermediate supplier options, the versatility of the routes described in CN1033391C offers substantial strategic advantages. The ability to modulate the substitution pattern at the 5, 6, 7, or 8 positions of the A-ring allows for fine-tuning of the molecule's lipophilicity and electronic properties without compromising the core pharmacophore. This flexibility is crucial for optimizing drug candidates during the lead optimization phase, ensuring that the final active pharmaceutical ingredient (API) meets stringent regulatory requirements for purity and performance. Furthermore, the reliance on well-established catalytic cycles involving palladium and nickel ensures that the technology is transferable to large-scale production environments, facilitating the commercial scale-up of complex heterocycles needed for global market supply.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations detailed in this patent, the synthesis of beta-carboline derivatives often suffered from significant drawbacks regarding metabolic stability and structural diversity. As noted in the background art, earlier compounds such as those disclosed in EP-54507 and EP-A-137390, while possessing receptor affinity, failed to demonstrate the metabolic stability required for viable pharmaceutical products. Conventional synthetic routes frequently relied on harsh electrophilic aromatic substitution conditions that offered poor regioselectivity, leading to difficult-to-separate isomer mixtures and reduced overall yields. Additionally, traditional methods for introducing side chains onto the indole nucleus were often limited to simple alkylation or acylation, restricting the chemical space available for medicinal chemists to explore. These limitations resulted in higher development costs and longer timelines, as extensive purification was required to remove impurities that could pose toxicity risks in final drug formulations.
The Novel Approach
The methodology presented in CN1033391C overcomes these historical barriers through the application of modern organometallic chemistry, specifically utilizing nickel and palladium-catalyzed cross-coupling reactions. This novel approach enables the direct coupling of pre-functionalized beta-carboline precursors, such as triflates or halides, with a wide array of organometallic reagents including boronic acids, stannanes, and zinc derivatives. By shifting the functionalization strategy to late-stage cross-coupling, the process achieves exceptional regiocontrol, ensuring that substituents are installed exclusively at the desired positions on the A-ring. This precision not only enhances the metabolic profile of the resulting molecules by blocking vulnerable metabolic sites but also streamlines the purification process. The inclusion of alkynylation pathways further expands the structural diversity, allowing for the creation of rigidified analogues that may possess improved receptor selectivity and bioavailability compared to their flexible counterparts.
Mechanistic Insights into Pd/Ni-Catalyzed Cross-Coupling and Cyclization
The core of this technological advancement lies in the sophisticated manipulation of transition metal catalytic cycles to construct the substituted beta-carboline framework. In the preferred embodiments, the reaction initiates with the oxidative addition of a palladium(0) or nickel(0) species into the carbon-halogen or carbon-oxygen (triflate) bond of the beta-carboline precursor. This step is critical and is facilitated by electron-rich phosphine ligands, such as triphenylphosphine or tri-o-tolylphosphine, which stabilize the active metal center and promote the formation of the organometallic intermediate. Following oxidative addition, the transmetallation step occurs, where the organic group from the coupling partner (e.g., a boronic acid or organotin compound) is transferred to the metal center. The cycle concludes with reductive elimination, which forge the new carbon-carbon bond and regenerate the active catalyst. This mechanistic pathway is highly efficient and tolerant of various functional groups, allowing for the synthesis of complex molecules without the need for extensive protecting group strategies.
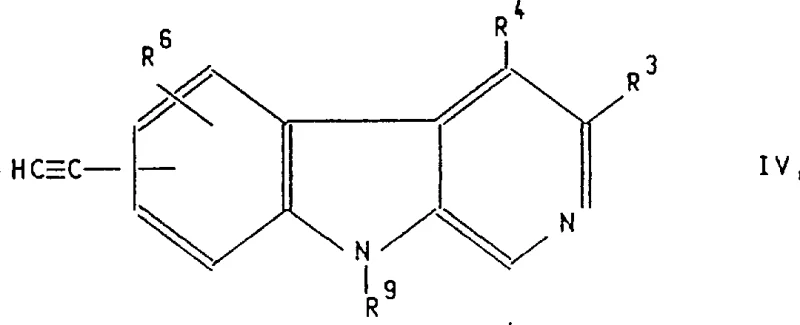
Impurity control is inherently built into this catalytic design through the high specificity of the metal-ligand complexes. Unlike radical-based or acid-catalyzed processes that can lead to scrambling of substituents or polymerization, the concerted nature of the cross-coupling mechanism minimizes side reactions. For instance, in the alkynylation of Formula IV compounds, the use of copper(I) iodide as a co-catalyst facilitates the formation of the copper-acetylide species, which then participates in the transmetallation with the palladium complex. This dual-catalyst system ensures that the triple bond is formed cleanly without affecting other sensitive moieties like esters or ethers present on the molecule. Furthermore, the patent describes dehydrogenation and cycloaddition routes that proceed through well-defined transition states, reducing the formation of byproducts. This high level of chemical fidelity translates directly to downstream processing benefits, as the crude reaction mixtures require less intensive chromatographic separation, thereby reducing solvent consumption and waste generation.
How to Synthesize Substituted Beta-Carboline Derivatives Efficiently
The synthesis of these high-value intermediates follows a logical progression starting from readily available indole precursors, moving through cyclization to form the beta-carboline core, and concluding with the critical A-ring functionalization. The patent provides detailed experimental examples demonstrating the robustness of these methods across a range of substrates, confirming their practical utility in a laboratory setting. Operators must pay close attention to the exclusion of oxygen and moisture during the catalytic steps, as the active palladium and nickel species are sensitive to oxidation. The following guide outlines the standardized operational framework derived from the patent's preferred embodiments, ensuring reproducibility and safety during scale-up operations.
- Preparation of the triflate leaving group precursor from the corresponding hydroxy-beta-carboline using trifluoromethanesulfonic anhydride.
- Execution of the cross-coupling reaction using organometallic reagents (boronic acids or stannanes) with palladium or nickel catalysts in inert solvents.
- Final purification via chromatography and recrystallization to achieve high-purity pharmaceutical intermediates suitable for further drug development.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the synthetic routes described in CN1033391C offers compelling economic and logistical benefits that extend beyond simple yield improvements. The shift towards catalytic cross-coupling eliminates the need for stoichiometric amounts of hazardous reagents often used in classical substitution chemistry, thereby reducing the environmental footprint and associated disposal costs. Moreover, the modularity of the approach means that a single batch of the beta-carboline core can be diversified into multiple derivatives simply by changing the coupling partner, allowing for agile response to changing R&D demands without retooling the entire production line. This flexibility is a key driver for cost reduction in API manufacturing, as it maximizes asset utilization and minimizes inventory holding costs for specialized intermediates.
- Cost Reduction in Manufacturing: The implementation of palladium and nickel-catalyzed processes significantly lowers the overall cost of goods sold by improving atom economy and reducing the number of synthetic steps required to reach the target molecule. By avoiding multi-step protection and deprotection sequences typical of older methods, the process flow is drastically simplified, leading to substantial cost savings in labor and raw materials. Additionally, the ability to recover and recycle expensive noble metal catalysts through established scavenging technologies further enhances the economic viability of the process, making it competitive even for large-volume production runs where catalyst cost is a major factor.
- Enhanced Supply Chain Reliability: The starting materials required for these reactions, such as aryl boronic acids, halogenated indoles, and standard phosphine ligands, are commodity chemicals available from a broad network of global suppliers. This widespread availability mitigates the risk of supply chain disruptions that often plague processes relying on exotic or proprietary reagents. Furthermore, the reaction conditions are compatible with standard stainless steel reactors and do not require specialized high-pressure or cryogenic equipment, ensuring that production can be easily transferred between different manufacturing sites globally without significant capital investment, thus securing supply continuity.
- Scalability and Environmental Compliance: The processes described operate in common industrial solvents like toluene, DMF, and THF, which have well-established recovery and recycling protocols in modern chemical plants. The high selectivity of the catalytic reactions results in cleaner reaction profiles with fewer byproducts, simplifying wastewater treatment and reducing the load on effluent processing facilities. This alignment with green chemistry principles not only ensures compliance with increasingly stringent environmental regulations but also enhances the corporate sustainability profile of the manufacturing operation, a factor that is becoming increasingly important for partnerships with major pharmaceutical companies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these beta-carboline derivatives. The answers are derived directly from the experimental data and technical specifications provided in the patent documentation, ensuring accuracy and relevance for potential partners. Understanding these details is essential for evaluating the feasibility of integrating this technology into existing drug development pipelines.
Q: What distinguishes these beta-carboline derivatives from prior art compounds?
A: Unlike earlier compounds disclosed in EP-54507 which lacked sufficient metabolic stability, the derivatives in CN1033391C feature specific A-ring aralkylation or alkynylation that significantly enhances metabolic stability while maintaining high affinity for chlordiazepoxide receptors.
Q: Which catalytic systems are preferred for the A-ring functionalization?
A: The patent highlights the efficacy of nickel and palladium catalysts, specifically complexes like tetrakis(triphenylphosphine)palladium(0) and bis(triphenylphosphine)palladium(II) chloride, often utilized in conjunction with copper(I) iodide co-catalysts for alkynylation reactions.
Q: Are the synthesis conditions suitable for large-scale manufacturing?
A: Yes, the described methods utilize standard industrial solvents such as toluene, DMF, and THF, and operate at moderate temperatures ranging from 0°C to reflux, making the process amenable to commercial scale-up without requiring extreme cryogenic or high-pressure equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Beta-Carboline Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of having a dependable partner who can translate complex patent chemistry into commercial reality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from the bench to the plant. We are committed to delivering high-purity beta-carboline derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our expertise in handling sensitive catalytic reactions guarantees that the metabolic stability and receptor affinity inherent in the CN1033391C designs are preserved in the final bulk material.
We invite you to engage with our technical procurement team to discuss how we can tailor our manufacturing capabilities to your specific needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can reduce your overall development budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that accelerate your path to market with confidence and efficiency.
