Advanced Solvent-Participation Synthesis of 4-Acetyl-1,2,3-Triazoles for Commercial Scale-Up
Advanced Solvent-Participation Synthesis of 4-Acetyl-1,2,3-Triazoles for Commercial Scale-Up
The pharmaceutical and fine chemical industries are constantly seeking robust methodologies to construct nitrogen-containing heterocycles, particularly 1,2,3-triazoles, due to their profound biological activities and widespread application in drug discovery. Patent CN107162991B introduces a groundbreaking approach for synthesizing 4-acetyl-1,2,3-triazole compounds by leveraging solvent participation to provide the necessary carbon source. This innovation represents a significant paradigm shift from traditional multi-step syntheses, offering a streamlined one-pot reaction that utilizes methyl ketones and organic azides in the presence of inexpensive metal salt catalysts. For R&D Directors and Procurement Managers, this technology promises not only enhanced synthetic efficiency but also a substantial reduction in the cost of goods sold (COGS) by eliminating the need for pre-functionalized acetylating agents. The method's ability to operate under mild conditions while maintaining high yields positions it as a critical asset for the reliable pharmaceutical intermediates supplier looking to optimize their production pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1,2,3-triazole derivatives has relied heavily on the Huisgen 1,3-dipolar cycloaddition, which often necessitates harsh reaction conditions such as high temperatures and pressures to proceed at a viable rate. Furthermore, traditional methods frequently suffer from poor regioselectivity, yielding mixtures of 1,4-disubstituted and 1,5-disubstituted isomers that require cumbersome and costly separation processes. The reliance on terminal alkynes and specific activating groups limits the substrate scope, while the use of noble metal catalysts in click chemistry variants drives up the raw material costs significantly. Additionally, the safety concerns associated with handling unstable azide intermediates and the generation of toxic byproducts in older protocols pose significant challenges for industrial scale-up. These limitations collectively hinder the commercial viability of many potential drug candidates, creating a bottleneck in the development of high-purity OLED material and pharmaceutical intermediates.
The Novel Approach
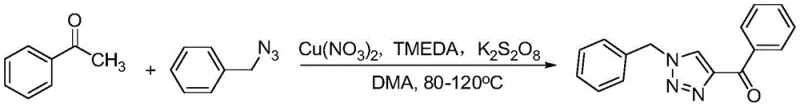
In stark contrast, the method disclosed in CN107162991B utilizes a solvent-participation strategy where solvents like DMA, DMSO, or TMEDA act as the carbon source for the acetyl group, thereby simplifying the reactant list. This novel approach employs inexpensive copper salts as catalysts and TMEDA as a ligand, facilitating a highly efficient one-pot reaction between methyl ketones and organic azides. The reaction conditions are remarkably mild, typically operating between 80-120°C, which significantly reduces energy consumption and enhances operational safety. By avoiding the need for pre-synthesized acetylating reagents, this method drastically simplifies the supply chain logistics and reduces the overall chemical waste generated. The broad substrate scope allows for the synthesis of diverse derivatives, making it an ideal solution for cost reduction in pharmaceutical intermediates manufacturing where flexibility and efficiency are paramount.
Mechanistic Insights into Copper-Catalyzed Solvent Participation
The core of this technological advancement lies in the unique mechanistic pathway where the solvent molecule is actively incorporated into the final product structure. The copper catalyst, in conjunction with the TMEDA ligand, activates the methyl ketone and facilitates the oxidative coupling with the organic azide. The solvent, acting as a carbon source, undergoes a transformation that introduces the acetyl moiety at the 4-position of the triazole ring with high regioselectivity. This mechanism bypasses the need for external acetylating agents, which are often expensive and hazardous. The oxidative conditions, supported by oxidants like K2S2O8 or TBHP, ensure the reaction proceeds to completion without the accumulation of partially reduced intermediates. Understanding this mechanism is crucial for R&D teams aiming to replicate the high yields and purity profiles reported in the patent data.

Furthermore, the impurity control mechanism inherent in this catalytic system is designed to minimize side reactions that typically plague triazole synthesis. The specific coordination of the copper-TMEDA complex stabilizes the transition state, preventing the polymerization of olefins or the decomposition of azides that can occur under thermal stress. This high level of control results in a cleaner reaction profile, reducing the burden on downstream purification processes such as column chromatography. For quality control laboratories, this means fewer impurities to identify and quantify, leading to faster release times for batches. The ability to consistently produce high-purity 4-acetyl-1,2,3-triazole compounds is a significant advantage for suppliers targeting the stringent requirements of the global pharmaceutical market.
How to Synthesize 4-Acetyl-1,2,3-Triazole Efficiently
The practical implementation of this synthesis route involves a straightforward procedure that can be easily adapted for both laboratory and pilot plant scales. The process begins with the precise weighing of methyl ketone compounds and organic azides, which are then combined with the metal salt catalyst and oxidant in the chosen solvent. The reaction mixture is subjected to magnetic stirring at controlled temperatures, typically ranging from 80°C to 120°C, for a duration of 3 to 8 hours depending on the specific substrate reactivity. Detailed standardized synthesis steps see the guide below, which outlines the exact molar ratios and workup procedures to ensure optimal yield and reproducibility. This simplicity in execution is a key factor in the technology's potential for widespread adoption in the fine chemical industry.
- Prepare the reaction mixture by combining methyl ketone compounds, organic azides, metal salt catalyst (e.g., Cu(NO3)2), TMEDA ligand, and oxidant in a solvent like DMA.
- Heat the mixture to 80-120°C under magnetic stirring for 3-8 hours to facilitate the one-pot cycloaddition and oxidation reaction.
- Perform workup by extraction with ethyl acetate, wash with saturated brine, dry over anhydrous sodium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement perspective, this synthesis method offers compelling economic benefits driven by the use of commodity chemicals and the elimination of expensive reagents. The substitution of noble metal catalysts with abundant copper salts results in a direct reduction in raw material costs, which is a critical metric for purchasing managers evaluating vendor proposals. Moreover, the use of common solvents like DMA and DMSO as reactants simplifies inventory management, as these chemicals are already standard in most chemical manufacturing facilities. The one-pot nature of the reaction reduces the number of unit operations required, leading to lower labor costs and decreased equipment occupancy time. These factors collectively contribute to a more competitive pricing structure for the final intermediates, enhancing the supplier's value proposition in a cost-sensitive market.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and specialized acetylating agents significantly lowers the direct material costs associated with production. By utilizing the solvent as a reactant, the process reduces the total number of chemicals required, thereby minimizing waste disposal costs and environmental compliance burdens. The high yields reported in the patent examples indicate efficient atom economy, which further translates to substantial cost savings per kilogram of product. This economic efficiency allows manufacturers to offer more competitive pricing without compromising on quality or margin.
- Enhanced Supply Chain Reliability: The reliance on readily available raw materials such as methyl ketones and organic azides ensures a stable supply chain that is less susceptible to market fluctuations. Unlike methods requiring rare or specialized reagents, this protocol can be sustained even during periods of global supply chain disruption. The robustness of the reaction conditions also means that production can be maintained across different manufacturing sites with minimal requalification effort. This reliability is crucial for supply chain heads who need to guarantee continuous availability of critical intermediates for downstream drug manufacturing.
- Scalability and Environmental Compliance: The mild reaction temperatures and atmospheric pressure conditions make this process inherently safer and easier to scale up compared to high-pressure alternatives. The simplified workup procedure, involving standard extraction and washing steps, reduces the generation of hazardous waste and facilitates compliance with increasingly strict environmental regulations. The ability to scale from grams to tons without significant process modifications ensures that the technology can meet growing market demand efficiently. This scalability supports the commercial scale-up of complex pharmaceutical intermediates, enabling rapid response to market needs.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the patent specifications and are intended to provide clarity on the method's capabilities and limitations. Understanding these details is essential for stakeholders evaluating the feasibility of integrating this process into their existing manufacturing workflows. The information provided here serves as a foundational reference for further technical discussions and feasibility studies.
Q: What are the primary advantages of this solvent-participation method over traditional click chemistry?
A: This method eliminates the need for expensive noble metal catalysts and harsh conditions typical of traditional click chemistry. By utilizing the solvent as a carbon source, it simplifies the reaction setup to a one-pot process, significantly reducing raw material costs and operational complexity while maintaining high yields.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the process utilizes readily available raw materials and mild reaction conditions (80-120°C), which are highly favorable for scale-up. The simple workup procedure involving standard extraction and chromatography ensures that the method can be adapted for commercial scale-up of complex pharmaceutical intermediates without requiring specialized high-pressure equipment.
Q: How does this method impact the purity and impurity profile of the final triazole product?
A: The use of specific copper salts and TMEDA ligands enhances regioselectivity, minimizing the formation of unwanted isomers often seen in thermal cycloadditions. The one-pot nature reduces intermediate handling, thereby lowering the risk of contamination and ensuring high-purity 4-acetyl-1,2,3-triazole compounds suitable for sensitive biological applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4-Acetyl-1,2,3-Triazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the solvent-participation synthesis method described in CN107162991B for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory methods are successfully translated into robust industrial processes. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against the highest industry standards. We are equipped to handle the specific challenges of triazole synthesis, offering tailored solutions that meet the exacting requirements of global pharmaceutical clients.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis that demonstrates how this method can optimize your specific supply chain. We encourage you to contact us to request specific COA data and route feasibility assessments for your target molecules. By partnering with us, you gain access to a wealth of chemical expertise and manufacturing capacity dedicated to driving your projects forward efficiently and cost-effectively.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
