Advanced Synthesis of GABA Analog Prodrugs for Enhanced Bioavailability and Commercial Scalability
The pharmaceutical industry continuously seeks innovative solutions to overcome the bioavailability limitations of potent therapeutic agents, particularly those with polar functional groups that hinder membrane permeability. Patent CN1675165A, filed in September 2005, presents a groundbreaking methodology for synthesizing 1-(acyloxy)-alkyl carbamates of GABA analogs, effectively transforming hydrophilic drugs into lipophilic prodrugs. This technical advancement addresses the critical challenge of delivering GABAergic agents across hydrophobic biological barriers, such as the gastrointestinal tract and the blood-brain barrier, without compromising the safety profile of the parent drug. By masking polar amino and carboxylic acid groups with precursor moieties that are unstable under physiological conditions, this invention ensures rapid and quantitative cleavage in vivo to release the active pharmaceutical ingredient. The strategic design of these prodrugs not only enhances systemic exposure but also reduces local irritation to the gastric mucosa, representing a significant leap forward in oral drug delivery systems for neurological disorders.
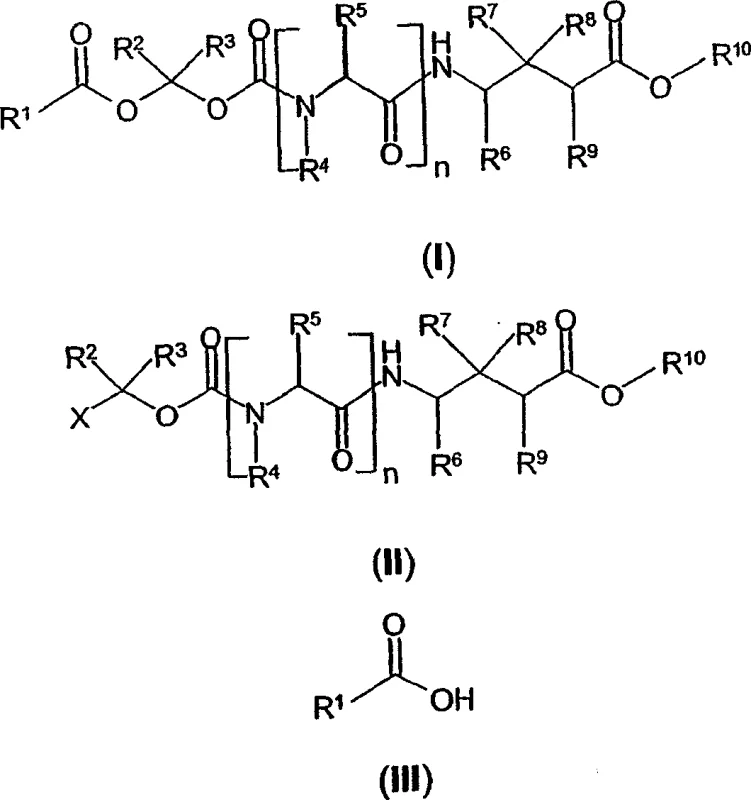
For research and development directors focusing on process chemistry, understanding the structural versatility of these derivatives is paramount. The general formula (I) disclosed in the patent encompasses a broad spectrum of chemical space, allowing for fine-tuning of physicochemical properties such as solubility, stability, and partition coefficients. The ability to vary the acyl group (R1) and the alkyl linker provides a robust platform for optimizing pharmacokinetic profiles. Furthermore, the inclusion of diverse GABA analog backbones, ranging from simple cyclohexyl derivatives to complex bridged bicyclic systems, demonstrates the universal applicability of this synthetic approach. As a reliable pharmaceutical intermediates supplier, recognizing the depth of this chemical library is essential for developing next-generation therapeutics that meet stringent regulatory requirements for purity and efficacy while addressing the unmet medical needs of patients suffering from epilepsy and neuropathic pain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of acyloxyalkyl esters and carbamates has been plagued by significant technical hurdles that impede large-scale manufacturing and commercial viability. Traditional methods described in prior art often rely on multistep synthetic routes that employ unstable intermediates, which are prone to decomposition and difficult to isolate with high purity. These conventional pathways frequently necessitate the use of toxic compounds or hazardous salts, introducing substantial safety risks for operators and complicating waste management protocols in industrial settings. Moreover, the reliance on harsh reaction conditions can lead to the formation of undesirable by-products, such as lactams, which are challenging to remove and can compromise the safety profile of the final drug product. The cumulative effect of these inefficiencies results in low overall yields, increased production costs, and extended lead times, making it difficult for manufacturers to achieve cost reduction in pharmaceutical intermediates manufacturing while maintaining consistent quality standards.
The Novel Approach
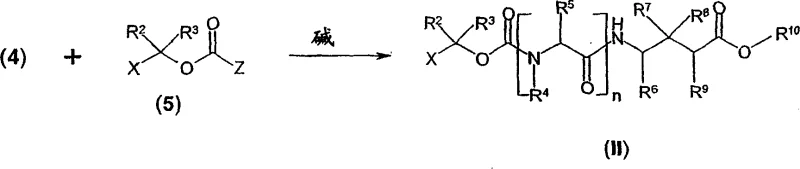
In stark contrast, the methodology disclosed in CN1675165A offers a streamlined and efficient alternative that directly addresses the shortcomings of legacy processes. This novel approach involves the direct reaction of a 1-haloalkyl carbamate of a GABA analog with a carboxylic acid in the presence of a metal salt or an organic base. This single-step transformation eliminates the need for isolating unstable intermediates, thereby simplifying the workflow and reducing the potential for material loss. The reaction conditions are remarkably mild, typically proceeding at temperatures between -25°C and 120°C, which allows for precise control over the reaction kinetics and minimizes thermal degradation of sensitive functional groups. By utilizing readily available synthetic precursors such as simple carboxylic acids and stable haloalkyl carbamates, this process facilitates the commercial scale-up of complex pharmaceutical intermediates. The versatility of the method is further enhanced by the option to use the carboxylic acid itself as the reaction solvent, which not only drives the equilibrium towards product formation but also simplifies the downstream processing and purification steps.
Mechanistic Insights into Base-Promoted Nucleophilic Substitution
The core chemical transformation underpinning this invention is a nucleophilic substitution reaction where the carboxylate anion attacks the electrophilic carbon of the 1-haloalkyl carbamate moiety. In the presence of a base, the carboxylic acid is deprotonated to generate a nucleophilic carboxylate species, which then displaces the halide leaving group (X = F, Cl, Br, or I) from the alpha-position of the carbamate. This mechanism is highly dependent on the nature of the base and the solvent system employed. When metal salts such as cesium carbonate or silver salts are used, they act to sequester the halide ion, driving the reaction forward through the formation of insoluble precipitates or stable complexes. Alternatively, organic bases like triethylamine or DBU facilitate the generation of the reactive nucleophile in situ. The choice of base can significantly influence the reaction rate and the impurity profile, with stronger bases potentially leading to faster conversion but requiring careful control to avoid side reactions such as elimination or hydrolysis of the carbamate linkage.
Controlling the impurity profile is a critical aspect of this synthesis, particularly for GABA analogs which are prone to cyclization. The patent highlights that the 1-haloalkyl carbamate intermediates must be handled carefully to prevent the formation of lactams, which can occur if the free amine is exposed to the carboxylic acid moiety within the same molecule under certain conditions. The use of protecting groups, such as the tert-butoxycarbonyl (Boc) group, during the initial stages of synthesis helps to stabilize the amine functionality and prevent premature cyclization. Furthermore, the selection of reaction solvents plays a pivotal role in managing solubility and reaction homogeneity. Solvents like dichloromethane, toluene, or N-methylpyrrolidone are preferred for their ability to dissolve both the organic substrates and the inorganic salts, ensuring efficient mass transfer. The rigorous control of water content is also essential, as moisture can lead to the hydrolysis of the reactive haloalkyl carbamate, generating the corresponding alcohol and carbon dioxide, which would reduce the overall yield and complicate the purification process.
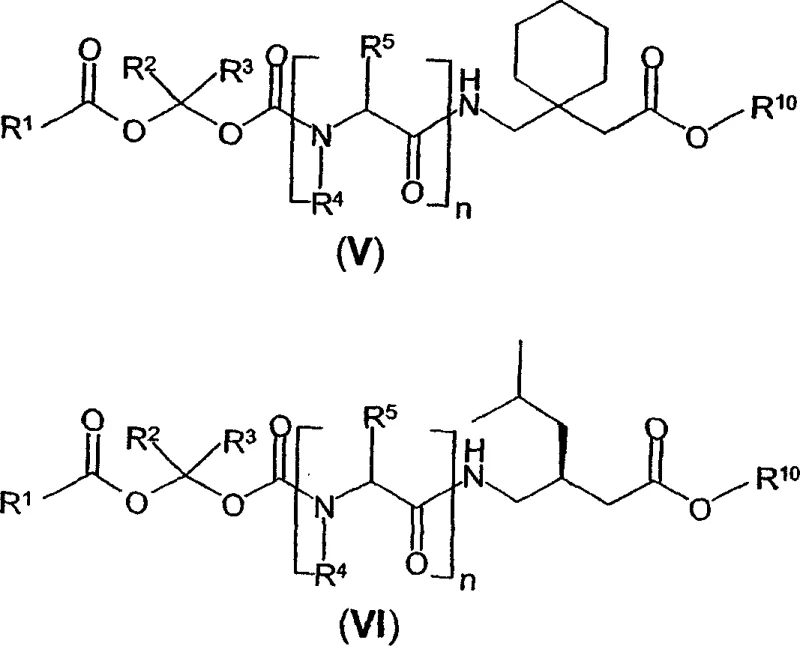
To illustrate the practical application of this chemistry, the patent provides specific examples involving well-known GABA analogs such as gabapentin and pregabalin. For instance, the synthesis of benzyl 1-{[(α-isobutanoyloxyethoxy)carbonyl]aminomethyl}-1-cyclohexane acetate demonstrates the successful coupling of a branched carboxylic acid with a gabapentin-derived haloalkyl carbamate. The resulting prodrug exhibits improved lipophilicity compared to the parent acid, facilitating better absorption. Similarly, derivatives of pregabalin are synthesized using allyl or trimethylsilyl protecting groups, which can be removed under mild conditions to yield the free acid prodrug. These specific embodiments underscore the flexibility of the platform technology, allowing medicinal chemists to explore a wide array of prodrug candidates to optimize the therapeutic index of GABAergic drugs. The structural diversity achievable through this method is vast, encompassing various acyl chains and ester protecting groups that can be tailored to specific delivery requirements.
How to Synthesize 1-(Acyloxy)-alkyl Carbamates Efficiently
The synthesis of these high-value prodrugs follows a logical sequence that begins with the protection of the GABA analog amine, followed by esterification and subsequent acylation to introduce the haloalkyl carbamate functionality. The detailed standardized synthetic steps involve precise stoichiometric control and temperature management to ensure high conversion and minimal by-product formation. The process is designed to be robust and reproducible, making it suitable for transfer from laboratory scale to pilot plant and eventually to full commercial production. Operators must adhere to strict safety protocols when handling reactive halides and strong bases, ensuring that all reactions are conducted under inert atmosphere to prevent moisture ingress. The following guide outlines the critical operational parameters required to execute this synthesis successfully.
- Preparation of the 1-haloalkyl carbamate intermediate via acylation of the protected GABA analog amine.
- Reaction of the haloalkyl carbamate with a carboxylic acid in the presence of a metal salt or organic base.
- Purification and isolation of the final 1-(acyloxy)-alkyl carbamate prodrug through standard extraction and crystallization.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, the adoption of this novel synthesis route offers transformative benefits that extend beyond mere technical feasibility. The elimination of complex multistep sequences and the avoidance of unstable intermediates significantly streamline the manufacturing process, leading to substantial cost savings in raw materials and labor. By utilizing readily available carboxylic acids and simple haloalkyl chloroformates, manufacturers can reduce their dependency on specialized or exotic reagents that are subject to supply chain volatility. This simplification of the bill of materials enhances supply chain reliability, ensuring consistent availability of key intermediates even during periods of market disruption. Furthermore, the ability to perform the reaction in a single pot with minimal workup reduces the consumption of solvents and energy, aligning with modern sustainability goals and reducing the environmental footprint of pharmaceutical production.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the drastic simplification of the synthetic route. By avoiding the use of heavy metal catalysts in certain embodiments and eliminating the need for cryogenic conditions often required for unstable intermediates, the operational expenditure is significantly lowered. The high yields reported in the patent examples mean that less starting material is wasted, directly improving the atom economy of the process. Additionally, the use of common organic solvents and bases reduces the cost of goods sold, allowing for more competitive pricing in the global market. The removal of expensive purification steps, such as extensive chromatography, in favor of crystallization or simple extraction further contributes to the economic viability of scaling this technology.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals such as isobutyric acid, benzyl alcohol, and thionyl chloride ensures that the supply chain is robust and resilient. These materials are produced in large volumes by multiple suppliers worldwide, mitigating the risk of single-source dependency. The stability of the 1-haloalkyl carbamate intermediates, when properly stored, allows for the strategic stocking of key precursors, enabling manufacturers to respond quickly to fluctuations in demand. This flexibility is crucial for maintaining continuous production schedules and meeting the just-in-time delivery requirements of major pharmaceutical clients. The reduced complexity of the process also lowers the barrier to entry for contract manufacturing organizations, increasing the number of qualified suppliers and fostering a more competitive sourcing environment.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of highly toxic reagents make this process inherently safer and easier to scale. The ability to conduct reactions at near-ambient temperatures reduces the energy load on heating and cooling systems, contributing to lower utility costs. From an environmental standpoint, the reduction in solvent usage and the potential for solvent recycling align with green chemistry principles. The waste streams generated are less hazardous compared to those from traditional methods involving heavy metals or azide chemistry, simplifying waste treatment and disposal. This compliance with stringent environmental regulations not only avoids potential fines but also enhances the corporate social responsibility profile of the manufacturing entity, which is increasingly important for stakeholders and investors.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and application of these GABA analog prodrugs. These answers are derived directly from the technical specifications and experimental data provided in the patent documentation, ensuring accuracy and relevance for industry professionals. Understanding these nuances is critical for making informed decisions about process adoption and product development strategies.
Q: What are the primary advantages of this synthesis method over conventional routes?
A: This method avoids the use of unstable intermediates and toxic compounds often found in traditional multistep routes, offering higher yields and better scalability for commercial production.
Q: Which GABA analogs are suitable for this prodrug strategy?
A: The process is applicable to a wide range of GABA analogs including gabapentin, pregabalin, and various cyclohexyl or bicyclic amino acid derivatives.
Q: Does this process require heavy metal catalysts?
A: While metal salts can be used, the patent also describes embodiments using organic bases, allowing for metal-free synthesis which simplifies purification and regulatory compliance.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable GABA Analogs Supplier
The technological potential of the synthesis method described in CN1675165A is immense, offering a pathway to superior drug delivery systems for a wide range of neurological indications. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market launch is seamless and efficient. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of GABA analog intermediates meets the highest international standards. We understand the critical importance of consistency in pharmaceutical manufacturing and are committed to delivering products that support your regulatory filings and clinical trials with unwavering quality.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain valuable insights into the economic benefits of switching to this novel methodology. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your specific project needs. Let us partner with you to accelerate the development of next-generation therapeutics and bring life-changing medications to patients faster and more cost-effectively.
