Advanced Chiral Sulfone Technology For Commercial Scale Statin Intermediate Manufacturing
The pharmaceutical industry continuously seeks robust synthetic pathways for high-value active pharmaceutical ingredients, particularly for lipid-lowering agents known as HMG CoA reductase inhibitors. Patent CN1656077A discloses a sophisticated process for preparing chiral diol sulfones and dihydroxy acid derivatives that serve as critical intermediates in the synthesis of major statin drugs. This technology leverages the Julia-Kocienski olefination reaction to construct the carbon backbone with exceptional stereochemical control, addressing long-standing challenges in impurity profiles and process efficiency. By starting from readily available chiral pool materials like Kaneka alcohol, the method circumvents the need for expensive enzymatic resolutions or complex asymmetric catalysis in later stages. For R&D directors and procurement specialists, understanding this pathway offers a strategic advantage in sourcing reliable pharmaceutical intermediates with defined impurity profiles. The integration of sulfone chemistry allows for the precise installation of the trans-olefin geometry required for biological activity, ensuring that the final drug substance meets stringent regulatory standards for purity and efficacy.
The evolution of statin intermediate synthesis has historically been plagued by inefficiencies associated with traditional olefination methods and racemic resolutions. Conventional approaches often relied on Wittig reactions or Horner-Wadsworth-Emmons modifications that struggled to maintain high trans-selectivity without extensive purification steps, leading to significant material loss and increased waste generation. Furthermore, older methodologies frequently necessitated the resolution of racemic mixtures, inherently capping the maximum theoretical yield at fifty percent and driving up the cost of goods sold. In contrast, the novel approach detailed in the patent utilizes a chiral sulfone intermediate derived directly from enantiomerically pure starting materials. This strategic design ensures that the chirality is preserved throughout the synthetic sequence, eliminating the need for downstream chiral separation. The process transforms a liquid thioether into a crystalline sulfone, which serves as a pivotal purification point, allowing manufacturers to remove non-volatile impurities before the crucial carbon-carbon bond-forming step. This shift from resolution-dependent strategies to chirality-transfer methodologies represents a paradigm shift in manufacturing efficiency.
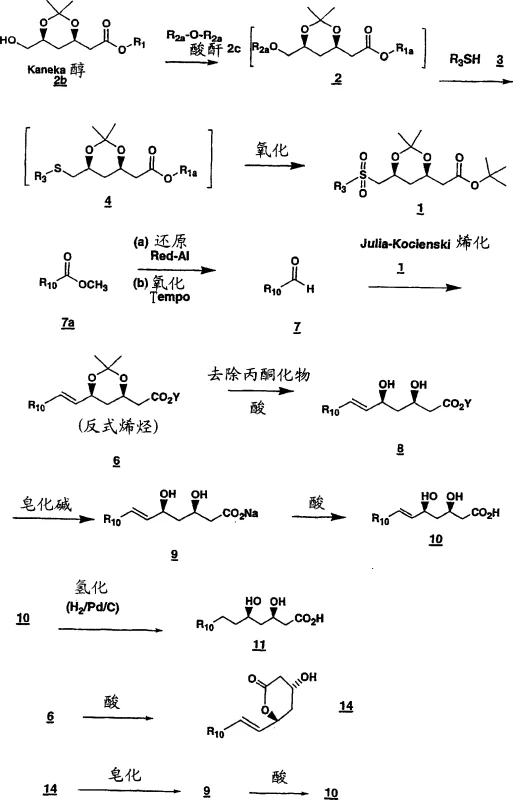
Mechanistically, the success of this process hinges on the precise oxidation of the chiral thioether to the corresponding sulfone and the subsequent base-mediated elimination. The oxidation step, typically catalyzed by ammonium heptamolybdate in the presence of hydrogen peroxide, proceeds under mild conditions that prevent epimerization of the sensitive chiral centers adjacent to the sulfur atom. This careful control of oxidation state is critical, as over-oxidation to sulfones or incomplete reaction to sulfoxides would compromise the reactivity in the subsequent olefination. The Julia-Kocienski reaction itself operates through a distinct mechanism involving the formation of a betaine intermediate that collapses to release sulfur dioxide and generate the alkene. The use of heteroaryl sulfones, such as tetrazole or benzothiazole derivatives, facilitates this fragmentation at lower temperatures compared to traditional phenyl sulfones. This kinetic advantage allows the reaction to proceed with high diastereoselectivity favoring the trans-isomer, which is the biologically active configuration for HMG CoA reductase inhibitors. The ability to tune the electronic properties of the sulfone leaving group provides chemists with a powerful handle to optimize reaction rates and selectivity for different substrate classes.
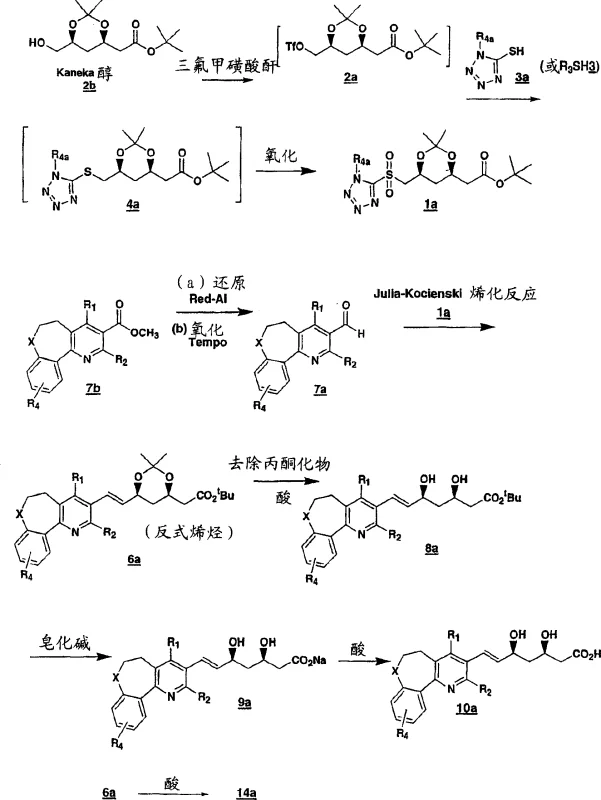
Impurity control is another cornerstone of this synthetic strategy, particularly regarding the removal of heavy metal residues and sulfur-containing byproducts. The crystallization of the chiral sulfone intermediate acts as a potent purge step for trace metals introduced during the oxidation phase, ensuring that the final API meets ICH guidelines for elemental impurities. Furthermore, the high stereoselectivity of the olefination minimizes the formation of cis-isomer impurities, which are structurally similar and difficult to separate by standard chromatography. By designing the synthesis to favor the desired isomer kinetically, the process reduces the burden on downstream purification units such as preparative HPLC or recrystallization trains. This inherent purity by design is highly valued by quality assurance teams who must validate cleaning procedures and demonstrate control over genotoxic impurities. The robustness of the reaction conditions also means that minor fluctuations in temperature or stoichiometry do not lead to catastrophic failures in product quality, enhancing the overall reliability of the manufacturing campaign.
How to Synthesize Chiral Diol Sulfone Efficiently
The implementation of this synthetic route requires careful attention to reaction parameters, particularly temperature control during the triflation and olefination steps. The initial activation of Kaneka alcohol to the triflate must be conducted at cryogenic temperatures to prevent decomposition, followed by immediate displacement with the thiol nucleophile. Detailed standardized synthesis steps are provided below to guide process chemists in replicating this high-yielding pathway.
- Preparation of triflate intermediate from Kaneka alcohol using triflic anhydride and organic base at low temperature.
- Nucleophilic substitution with tetrazole thiol to form chiral thioether followed by oxidation to chiral sulfone.
- Julia-Kocienski olefination with aldehyde using LiHMDS base to yield trans-alkene with high stereoselectivity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers substantial benefits for procurement managers and supply chain heads focused on cost reduction and continuity of supply. The reliance on Kaneka alcohol, a commercially established chiral building block, mitigates the risk associated with sourcing custom chiral catalysts or enzymes that may have limited supplier bases. This commoditization of the starting material ensures that multiple vendors can supply the necessary input, fostering a competitive pricing environment and reducing the risk of single-source bottlenecks. Additionally, the generation of crystalline intermediates throughout the sequence simplifies logistics, as solids are generally more stable and easier to transport than oils or solutions. This physical stability reduces the need for specialized cold-chain shipping or inert atmosphere handling, further lowering the total landed cost of the intermediate. For supply chain planners, the predictability of crystallization processes allows for more accurate forecasting of cycle times and inventory levels.
- Cost Reduction in Manufacturing: The elimination of chiral resolution steps fundamentally alters the economic model of statin intermediate production by removing the fifty percent yield penalty associated with separating enantiomers. By transferring chirality from the starting material rather than creating it de novo or resolving it later, the process maximizes atom economy and reduces the volume of solvent and reagents required per kilogram of product. Furthermore, the avoidance of expensive transition metal catalysts for asymmetric hydrogenation or coupling reduces the cost of raw materials and eliminates the need for costly metal scavenging resins. These cumulative efficiencies translate into a significantly lower cost of goods sold, allowing pharmaceutical companies to maintain healthy margins even in a generic pricing environment.
- Enhanced Supply Chain Reliability: Utilizing a synthetic route based on robust, non-proprietary reagents such as hydrogen peroxide and common organic bases enhances the resilience of the supply chain against raw material shortages. Unlike processes dependent on bespoke ligands or biocatalysts that require long lead times for production, the reagents for this Julia-Kocienski protocol are available from multiple global chemical distributors. This redundancy ensures that production schedules are not disrupted by vendor-specific quality issues or capacity constraints. Moreover, the high yield and purity of the intermediates reduce the frequency of batch failures and re-processing, leading to more consistent output volumes that align with demand forecasts. This reliability is critical for maintaining uninterrupted supply of life-saving cardiovascular medications to the global market.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing unit operations such as filtration and crystallization that are easily transferred from pilot plant to commercial scale without significant re-engineering. The use of aqueous work-ups and the generation of water-soluble byproducts simplify waste stream management, reducing the environmental footprint of the manufacturing facility. By minimizing the use of chlorinated solvents in the final isolation steps and employing greener oxidants like hydrogen peroxide, the process aligns with modern sustainability goals and regulatory expectations for green chemistry. This environmental compliance reduces the risk of regulatory fines and enhances the corporate social responsibility profile of the manufacturing partner, which is increasingly important for multinational pharmaceutical clients.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral sulfone technology in statin manufacturing. These insights are derived directly from the patent specifications and are intended to clarify feasibility for potential licensing or outsourcing partners.
Q: What is the primary advantage of the Julia-Kocienski olefination in this patent?
A: The primary advantage is the high trans-selectivity and optical purity achieved without requiring chromatographic separation, which significantly simplifies downstream processing.
Q: How does this process improve supply chain reliability for statin production?
A: By utilizing commercially available Kaneka alcohol as a starting material and generating crystalline intermediates, the process reduces dependency on custom chiral resolution and ensures consistent batch quality.
Q: Can this method be scaled for industrial production of Atorvastatin intermediates?
A: Yes, the patent describes robust conditions using common reagents like ammonium heptamolybdate and standard solvents, which are amenable to large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Diol Sulfone Supplier
NINGBO INNO PHARMCHEM stands at the forefront of fine chemical manufacturing, possessing the technical expertise to adapt and scale complex synthetic pathways like the one described in CN1656077A. Our engineering teams have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory successes are translated into efficient industrial realities. We maintain stringent purity specifications across all our product lines, supported by rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and potency. Our commitment to quality ensures that every batch of chiral intermediate meets the exacting standards required for GMP pharmaceutical synthesis, providing peace of mind to our global partners.
We invite procurement leaders and technical decision-makers to engage with us for a Customized Cost-Saving Analysis tailored to your specific production volumes and quality requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing capabilities can optimize your supply chain. Contact us today to discuss how we can support your next project with high-quality, cost-effective pharmaceutical intermediates.
