Advanced Manufacturing of Trans-4-Amino-1-Cyclohexanecarboxylic Acid Derivatives for Pharma
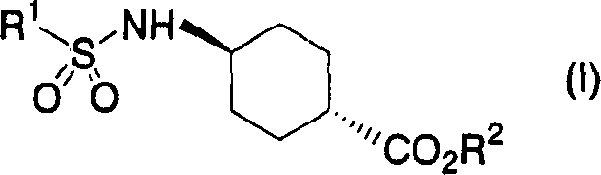
The pharmaceutical industry constantly seeks robust synthetic routes for key intermediates, particularly those required for novel receptor antagonists. Patent CN1639096A introduces a groundbreaking methodology for the preparation of trans-4-amino-1-cyclohexanecarboxylic acid derivatives, which serve as critical building blocks for NPYY5 receptor antagonists. Historically, the synthesis of these trans-isomers has been plagued by low yields and difficult separation from their cis-counterparts. This patent discloses a novel approach that leverages specific solvent systems and base-catalyzed isomerization to dramatically improve both the isomerization rate and the isolated yield. By shifting the equilibrium towards the thermodynamically more stable trans-configuration through selective crystallization in non-polar aprotic solvents, this technology offers a reliable pharmaceutical intermediate supplier pathway that addresses long-standing efficiency bottlenecks in metabolic disease drug development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to the innovations described in CN1639096A, the production of trans-4-amino-1-cyclohexanecarboxylic acid derivatives faced significant hurdles that impacted both cost and timeline for drug development programs. Conventional methods often resulted in a mixture of cis and trans isomers where the desired trans-isomer was difficult to isolate in high purity. Specifically, existing literature indicated that even after prolonged reaction times, the cis-isomer remained stubborn and resistant to isomerization, leading to isolated yields of the trans-isomer as low as 40 percent. This inefficiency necessitated extensive purification steps, such as repeated recrystallizations or chromatography, which not only increased the cost reduction in API manufacturing but also generated substantial chemical waste. Furthermore, the inability to effectively convert the cis-form meant that nearly half of the starting material was essentially wasted, creating a supply chain vulnerability for manufacturers relying on these intermediates for large-scale production of obesity and diabetes therapeutics.
The Novel Approach
The methodology outlined in the patent represents a paradigm shift by introducing a base-catalyzed isomerization performed in a non-polar aprotic solvent, such as toluene. Unlike previous attempts that may have used polar solvents hindering crystallization, this new process exploits the solubility differences between the isomers. As the reaction proceeds, the trans-isomer selectively crystallizes out of the solution, driving the equilibrium forward according to Le Chatelier's principle. This allows for isomerization rates exceeding 90 percent, with some embodiments achieving nearly 100 percent conversion. The process is versatile, accommodating various protecting groups on the amino and carboxyl functionalities, and can be integrated into a one-pot sequence that includes subsequent hydrolysis. This streamlined workflow eliminates the need for isolating intermediate species, thereby reducing processing time and enhancing the overall throughput for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Base-Catalyzed Isomerization and Oxidation
The core of this technological advancement lies in the precise control of stereochemistry through thermodynamic equilibration. The reaction mechanism involves the deprotonation of the alpha-carbon adjacent to the carbonyl group by a strong base, such as sodium methoxide or potassium tert-butoxide. This generates an enolate intermediate that allows for the inversion of configuration at the chiral center. In a non-polar environment like toluene, the trans-isomer, being less soluble than the cis-isomer, precipitates from the reaction mixture as it forms. This continuous removal of the product from the liquid phase prevents the reverse reaction, effectively locking the stereochemistry in the desired trans-configuration. Following the isomerization, the process typically involves an oxidation step where a sulfinyl group is converted to a sulfonyl group using hydrogen peroxide in the presence of a catalytic amount of ammonium molybdate. This oxidation is highly selective and proceeds under mild conditions, preserving the integrity of the cyclohexane ring and the ester functionality.
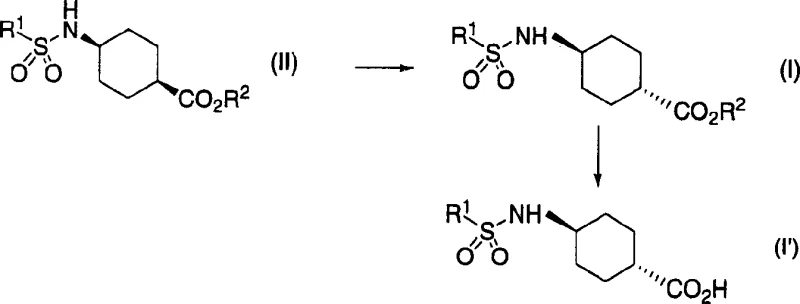
Impurity control is inherently built into this mechanism due to the crystallization-driven nature of the isomerization. Since the trans-isomer crystallizes preferentially, impurities and the remaining cis-isomer stay in the mother liquor, resulting in a crude product of exceptionally high purity without the need for column chromatography. The subsequent hydrolysis step, which converts the ester to the free carboxylic acid, can be performed directly on the crude isomerized product or after a simple filtration. This minimizes the exposure of the intermediate to harsh conditions that might lead to racemization or degradation. The use of common reagents like sodium methoxide and hydrogen peroxide ensures that the process remains economically viable and environmentally manageable, as the byproducts are generally benign salts and water. This mechanistic elegance translates directly into a robust manufacturing process capable of delivering high-purity OLED material or pharmaceutical grades with consistent quality.
How to Synthesize Trans-4-Amino-1-Cyclohexanecarboxylic Acid Derivatives Efficiently
The synthesis protocol detailed in the patent provides a clear roadmap for laboratory and pilot-scale production. It begins with the preparation of the cis-sulfinyl or sulfonyl precursor, which is then subjected to the isomerization conditions. The key operational parameter is the choice of solvent; toluene is identified as the optimal medium for maximizing yield through precipitation. The reaction temperature is typically maintained between 50°C and 150°C, with reflux conditions often preferred to ensure complete conversion. Following the isomerization, the mixture is cooled to induce further crystallization, and the solid is collected by filtration. For the final acid form, the ester is hydrolyzed using an aqueous base such as sodium hydroxide, followed by acidification to precipitate the final product. Detailed standardized synthetic steps see the guide below.
- React cis-4-amino-1-cyclohexanecarboxylic acid derivatives with a base such as sodium methoxide in a non-polar aprotic solvent like toluene to induce isomerization.
- Oxidize the resulting sulfinyl intermediate to the corresponding sulfonyl compound using hydrogen peroxide and a molybdenum catalyst.
- Hydrolyze the ester group under basic conditions to obtain the final trans-4-amino-1-cyclohexanecarboxylic acid derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the process described in CN1639096A offers tangible strategic benefits beyond mere chemical yield. The shift from low-yielding, separation-intensive methods to a crystallization-driven isomerization fundamentally alters the cost structure of producing these intermediates. By eliminating the need for expensive and time-consuming chromatographic purifications, the process significantly reduces the consumption of silica gel and organic solvents associated with column chromatography. This simplification of the downstream processing directly contributes to substantial cost savings in raw materials and waste disposal. Furthermore, the ability to perform the isomerization and hydrolysis in a telescoped manner reduces the number of unit operations, lowering labor costs and equipment occupancy time, which is critical for optimizing facility utilization rates in multi-purpose manufacturing plants.
- Cost Reduction in Manufacturing: The primary economic driver of this technology is the dramatic improvement in material efficiency. By converting the unwanted cis-isomer into the desired trans-isomer with high efficiency, the effective yield of the process is nearly doubled compared to traditional methods. This means that for every kilogram of starting material purchased, the output of valuable intermediate is significantly higher, effectively lowering the cost per kilogram of the final API intermediate. Additionally, the use of commodity chemicals like toluene, methanol, and hydrogen peroxide ensures that reagent costs remain stable and predictable, avoiding the volatility associated with specialized catalysts or exotic reagents. The reduction in purification steps also lowers the energy consumption required for solvent evaporation and drying, contributing to a leaner and more cost-effective manufacturing profile.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness and scalability of the disclosed chemistry. The reliance on standard industrial solvents and reagents means that sourcing risks are minimized, as these materials are readily available from multiple global suppliers. The process does not depend on single-source proprietary catalysts that could create bottlenecks. Moreover, the high selectivity of the reaction reduces the risk of batch failures due to impurity buildup, ensuring a more consistent and reliable supply of the intermediate to downstream drug substance manufacturers. This reliability is crucial for maintaining uninterrupted production schedules for chronic disease medications where patient demand is continuous and predictable.
- Scalability and Environmental Compliance: From an environmental and regulatory perspective, this process aligns well with green chemistry principles. The avoidance of heavy metal catalysts in the isomerization step and the use of hydrogen peroxide as a clean oxidant generate water as the primary byproduct, simplifying wastewater treatment. The high atom economy resulting from the isomerization of the cis-waste stream into useful product reduces the overall E-factor of the synthesis. Scalability is further enhanced because the crystallization mechanism works effectively at larger volumes, where heat and mass transfer can be managed efficiently. This makes the transition from kilogram-scale development to multi-ton commercial production smoother and less prone to the unforeseen complications that often arise when scaling complex purification processes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this isomerization technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing clarity on the operational parameters and expected outcomes. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing workflows.
Q: What is the primary advantage of the isomerization method in CN1639096A?
A: The primary advantage is the significantly improved isomerization rate, achieving over 90% conversion of the cis-isomer to the thermodynamically stable trans-isomer, compared to only 40% in prior art methods.
Q: Which solvents are preferred for the isomerization step?
A: Non-polar aprotic solvents are preferred, with toluene being the most optimal choice due to its ability to facilitate the selective crystallization of the trans-isomer during the reaction.
Q: Can this process be scaled for commercial production?
A: Yes, the process utilizes common industrial solvents like toluene and ethyl acetate and avoids complex chromatographic separations, making it highly suitable for large-scale commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Trans-4-Amino-1-Cyclohexanecarboxylic Acid Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the successful development of next-generation therapeutics. Our technical team has thoroughly analyzed the process described in CN1639096A and possesses the expertise to implement this advanced isomerization strategy effectively. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab bench to commercial reactor is seamless. Our facilities are equipped with state-of-the-art rigorous QC labs capable of monitoring stereochemical purity and impurity profiles with precision, guaranteeing that every batch meets stringent purity specifications required by global regulatory agencies.
We invite pharmaceutical partners to collaborate with us to leverage this efficient synthetic route for your NPYY5 antagonist programs. By partnering with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Let us help you optimize your supply chain and reduce your time-to-market with our reliable supply of high-purity pharmaceutical intermediates manufactured using cutting-edge process chemistry.
