Advanced Chiral Synthesis of 3-Amino-4-Methylpyrrolidine Derivatives for Next-Generation Quinolone Antibiotics
The pharmaceutical landscape for antibacterial agents has shifted dramatically towards third and fourth-generation quinolones, driven by the urgent need to combat resistant Gram-positive pathogens. Patent CN1054980A introduces a pivotal advancement in this domain by disclosing novel optically active 8-methoxyquinolone carboxylic acid derivatives and, more critically, their chiral intermediates. This intellectual property outlines a robust methodology for synthesizing 3-amino-4-methylpyrrolidine derivatives with precise stereochemical control, utilizing natural amino acids as the foundational chiral pool. For R&D directors and procurement strategists, this represents a significant opportunity to secure high-purity intermediates essential for next-generation antibiotics that demonstrate superior efficacy against Staphylococcus aureus compared to traditional agents like ciprofloxacin.
The core innovation lies in the ability to access specific optical isomers, namely the 7-[3(S)-amino-4(S)-methyl-1-pyrrolidinyl] and 7-[3(S)-amino-4(R)-methyl-1-pyrrolidinyl] configurations. Biological data within the patent reveals that the S-configuration at the 3-position of the pyrrolidine ring correlates with a two to three-fold increase in antibacterial potency and a six-fold improvement in preventive action against Gram-positive bacteria. This specificity necessitates a synthesis route that avoids racemic mixtures and costly resolution steps, positioning the disclosed chiral pool synthesis as a commercially superior alternative for large-scale API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of chiral pyrrolidine side chains for quinolone antibiotics has relied heavily on the resolution of racemic mixtures or the use of expensive chiral auxiliaries and catalysts. These conventional approaches often suffer from inherent inefficiencies, primarily the theoretical maximum yield of 50% during resolution, which drastically impacts cost structures and waste generation. Furthermore, methods involving transition metal-catalyzed asymmetric hydrogenation or alkylation require stringent exclusion of oxygen and moisture, along with the removal of trace heavy metals to meet regulatory standards for pharmaceutical ingredients. The complexity of purifying enantiomers from racemates often leads to extended cycle times and variable batch-to-batch consistency, creating bottlenecks in the supply chain for critical antibiotic intermediates.
The Novel Approach
In contrast, the methodology described in CN1054980A leverages the inherent chirality of abundant natural amino acids, specifically Aspartic acid and Malic acid, to dictate the stereochemistry of the final product. This "chiral pool" strategy eliminates the need for resolution steps, theoretically allowing for 100% yield of the desired enantiomer from the starting material. By converting L- or D-Aspartic acid into protected gamma-butyrolactones, or L- or D-Malic acid into protected diols, the synthesis establishes the critical stereocenters early in the sequence. This approach not only simplifies the synthetic route but also ensures that the optical purity is locked in from the outset, significantly reducing the risk of isomeric impurities that could compromise the biological activity of the final quinolone drug substance.
![Reaction scheme showing the synthesis of 3(S)-[(tert-butoxycarbonyl)amino]-gamma-butyrolactone from L-Aspartic acid](/insights/img/optically-active-pyrrolidine-intermediates-pharma-supplier-20260306130909-01.webp)
The versatility of this approach is evident in the multiple pathways provided. One route involves the conversion of L-Aspartic acid into 3(S)-[(benzyloxycarbonyl)amino]succinic acid, followed by cyclization to a lactone and subsequent methylation to introduce the 4-methyl group with high diastereoselectivity. Another parallel route utilizes L-Malic acid, undergoing esterification and protection to form a tetrahydropyranyl ether, followed by selective reduction and cyclization. Both pathways converge on the formation of the pyrrolidine ring through intramolecular nucleophilic substitution, utilizing leaving groups such as tosylates or bromides to close the ring while preserving the established chirality.
Mechanistic Insights into Chiral Pool Synthesis from Natural Amino Acids
The mechanistic elegance of this synthesis lies in the preservation of stereochemical integrity throughout the transformation from acyclic amino acids to cyclic pyrrolidines. In the Aspartic acid route, the alpha-amino group is first protected, typically with a benzyloxycarbonyl (Cbz) or tert-butoxycarbonyl (Boc) group, to prevent unwanted side reactions. The carboxylic acid moieties are then manipulated; one is reduced to an alcohol while the other participates in lactonization. The introduction of the methyl group at the 4-position is achieved through enolate alkylation of the lactone. Crucially, the existing chiral center at the 3-position directs the incoming methyl group, favoring the formation of specific diastereomers. Subsequent reduction of the lactone to a diol, followed by activation of the hydroxyl groups as sulfonates or halides, sets the stage for ring closure.
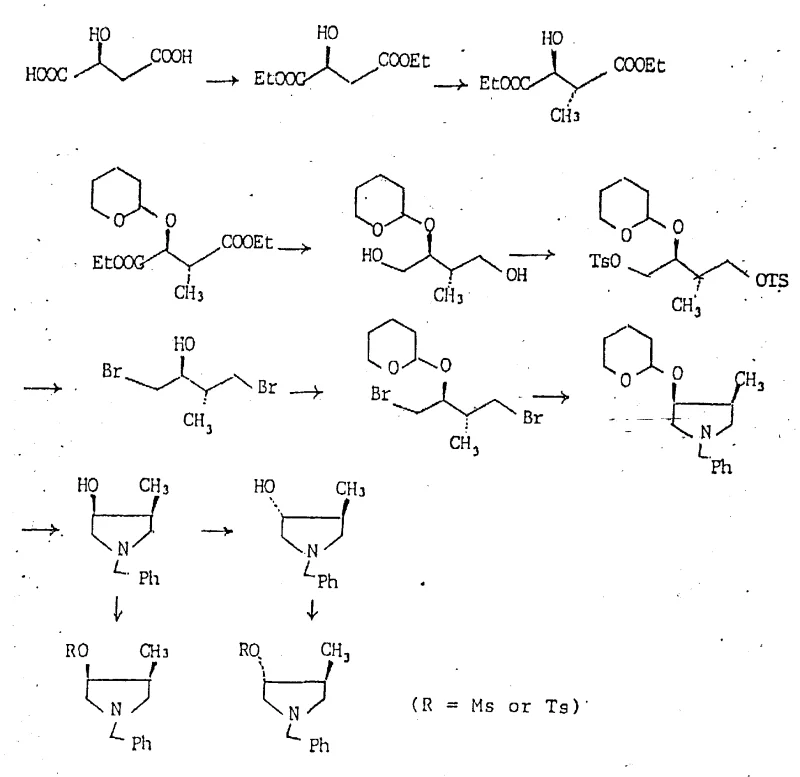
In the Malic acid pathway, the hydroxyl group of the starting material serves as a handle for further functionalization. After esterification to protect the carboxyl groups, the hydroxyl is protected as a tetrahydropyranyl (THP) ether. Alpha-alkylation with methyl iodide introduces the second chiral center. The subsequent reduction of the diester to a diol using lithium aluminum hydride provides the 1,4-diol scaffold necessary for pyrrolidine formation. The cyclization step typically involves converting one or both hydroxyl groups into good leaving groups, such as mesylates or tosylates, followed by displacement with a nitrogen nucleophile like benzylamine. This intramolecular SN2 reaction inverts the configuration at the carbon undergoing substitution, allowing for precise control over the final absolute configuration of the pyrrolidine ring, whether it be the (3S, 4R) or (3S, 4S) isomer required for optimal antibacterial activity.
Impurity control is inherently managed by the choice of starting material. Since L-Aspartic acid and L-Malic acid are available in high optical purity from fermentation processes, the resulting intermediates inherit this purity. The primary impurities arise from incomplete reactions or over-alkylation, which are manageable through standard purification techniques like crystallization or silica gel chromatography. The use of Boc protection groups also facilitates purification, as the protected intermediates often exhibit favorable crystallization properties, allowing for the removal of minor diastereomeric impurities before the final deprotection steps.
How to Synthesize 3-Amino-4-Methylpyrrolidine Derivatives Efficiently
The synthesis of these high-value chiral intermediates requires careful attention to reaction conditions, particularly during the alkylation and cyclization stages where stereochemistry is determined. The process begins with the selection of the appropriate enantiomer of the starting amino acid to match the desired final configuration. Protection strategies must be robust to withstand the basic conditions of alkylation and the reducing conditions of ester conversion. Detailed standardized synthesis steps see the guide below.
- Start with chiral pool materials such as L- or D-Aspartic acid or L- or D-Malic acid to establish the initial stereocenter.
- Perform protection and cyclization steps to form key intermediates like gamma-butyrolactones or protected diols.
- Execute stereoselective alkylation and ring closure reactions to construct the pyrrolidine core with defined absolute configuration.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift towards chiral pool synthesis offers tangible strategic benefits beyond mere technical feasibility. The reliance on commodity chemicals like Aspartic and Malic acid decouples the production of these critical intermediates from the volatility of the fine chemical catalyst market. These amino acids are produced on a massive scale for the food and feed industries, ensuring a stable and abundant supply base that is less susceptible to the disruptions often seen with specialized reagents. This stability translates directly into improved lead time predictability and reduced risk of production stoppages due to raw material shortages.
- Cost Reduction in Manufacturing: The elimination of resolution steps and expensive chiral catalysts fundamentally alters the cost structure of the intermediate. By avoiding the 50% yield loss inherent in racemic resolution, the effective cost per kilogram of the active isomer is drastically reduced. Furthermore, the removal of transition metals from the process simplifies the downstream purification train, reducing the consumption of scavengers and specialized filtration media. This streamlined workflow lowers both direct material costs and operational expenses related to waste disposal and quality control testing for heavy metals.
- Enhanced Supply Chain Reliability: Utilizing naturally derived starting materials enhances the sustainability profile of the supply chain, aligning with the increasing environmental, social, and governance (ESG) mandates of major pharmaceutical companies. The robustness of the chemistry, which employs standard unit operations such as esterification, reduction, and nucleophilic substitution, allows for flexible manufacturing across multiple sites. This flexibility mitigates the risk of single-source dependency, enabling procurement teams to qualify multiple suppliers who can replicate the chiral pool methodology without requiring proprietary catalyst licenses.
- Scalability and Environmental Compliance: The synthetic routes described are amenable to large-scale production, with reactions like lactonization and amine alkylation being well-understood in industrial settings. The avoidance of hazardous chiral reagents reduces the environmental footprint of the manufacturing process. Waste streams are primarily composed of organic salts and solvents which are easier to treat compared to heavy metal-contaminated waste. This compliance ease accelerates regulatory approvals for new manufacturing sites and reduces the long-term liability associated with environmental remediation, making the process attractive for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these chiral pyrrolidine intermediates. Understanding these nuances is critical for integrating this technology into existing API manufacturing workflows and ensuring the consistent quality of the final antibiotic product.
Q: What are the primary starting materials for this chiral synthesis?
A: The process utilizes readily available natural amino acids, specifically L- or D-Aspartic acid and L- or D-Malic acid, serving as the chiral pool foundation.
Q: Why is the 3(S)-amino configuration preferred for antibacterial activity?
A: Biological testing indicates that isomers with the 3(S)-amino configuration exhibit significantly higher in vitro antibacterial potency, particularly against Gram-positive bacteria like Staphylococcus aureus.
Q: How does this method improve supply chain reliability?
A: By relying on commodity-grade natural amino acids rather than expensive chiral catalysts or resolution agents, the process ensures consistent raw material availability and reduces dependency on specialized reagents.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-Amino-4-Methylpyrrolidine Supplier
The synthesis of optically active 3-amino-4-methylpyrrolidine derivatives represents a cornerstone in the production of advanced 8-methoxyquinolone antibiotics. At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including detailed chiral HPLC analysis to guarantee the optical purity required for high-efficacy antibacterial agents. We understand the critical nature of these intermediates in the global fight against antibiotic resistance and are committed to delivering quality that meets international regulatory standards.
We invite you to engage with our technical procurement team to discuss how our manufacturing capabilities can support your pipeline. By requesting a Customized Cost-Saving Analysis, you can gain insights into how our optimized chiral pool synthesis can reduce your overall COGS. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from development to commercial supply.
