Advanced Synthesis of Alpha-Hydroxycarboxylic Acid Amides for Scalable Fungicide Production
The chemical industry continuously seeks more efficient pathways for producing high-value agrochemical intermediates, and patent CN100340541C presents a significant breakthrough in the synthesis of alpha-hydroxycarboxylic acid amides. These compounds, specifically those of Formula I, serve as critical precursors for a new class of fungicidally active phenyl-propargyl ether derivatives. The traditional methods for constructing these complex amide bonds often involve multi-step sequences with activated carboxylic acids or isocyanide intermediates, which can introduce significant operational complexity and waste. This patent discloses a novel preparation method that streamlines the coupling of 2-(3-alkoxy-4-hydroxyphenyl)-ethylamines with alpha-hydroxycarboxylates or dioxolanones, offering a robust alternative for industrial scale-up. By optimizing reaction conditions to include solvent-free melting techniques or specific solution-phase protocols, the process achieves virtually quantitative conversion, addressing key pain points in yield and throughput for manufacturers of fine chemical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of structurally similar amide intermediates has relied on pathways that are chemically inefficient and operationally burdensome. Prior art, such as that described in WO 01/87822, often necessitates the activation of carboxylic acid groups prior to amidation or the use of unstable isocyanide species. These conventional routes typically require the use of large volumes of organic solvents to manage heat dissipation and solubility, leading to substantial downstream processing costs associated with solvent recovery and waste treatment. Furthermore, reactions involving activated esters or acid chlorides can be sensitive to moisture and require strict anhydrous conditions, increasing the risk of batch failure and impurity formation. The reliance on multi-step sequences, such as converting N-formyl groups to isocyanides before coupling, extends the overall lead time and reduces the overall mass balance efficiency of the manufacturing process. These factors collectively contribute to higher production costs and a larger environmental footprint, which are increasingly unacceptable in modern sustainable chemistry practices.
The Novel Approach
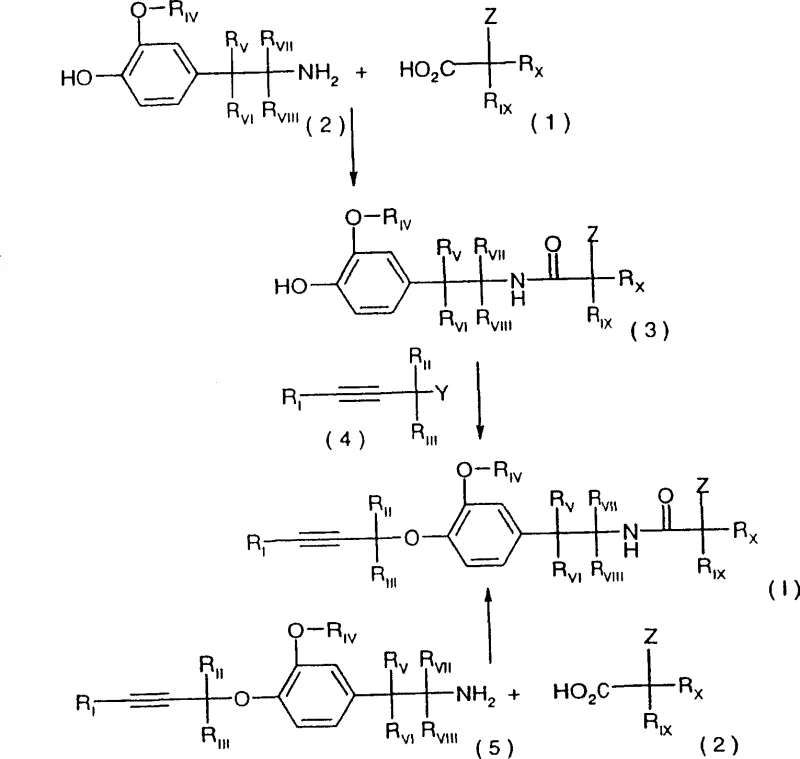
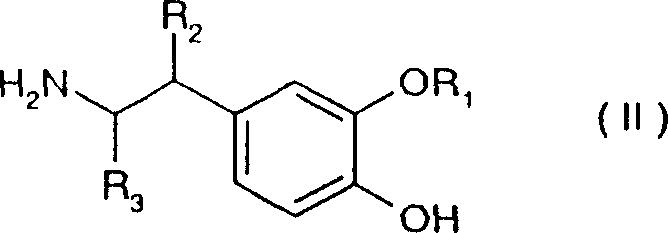
The methodology outlined in CN100340541C fundamentally shifts the paradigm by enabling the direct reaction of Formula II amines with Formula III alpha-hydroxycarboxylates or Formula IIIa dioxolanones. A particularly innovative aspect of this invention is the embodiment where the reaction is conducted in the absence of a solvent, utilizing the melting point of the reaction mixture itself to drive the kinetics. By heating the thoroughly mixed reactants to a temperature range between the melting point and up to +100°C above it, preferably within +20°C to +50°C, the process eliminates the need for bulk solvents entirely. This solvent-free approach not only drastically reduces the volume of waste generated but also significantly enhances the yield per unit volume of the reactor. Alternatively, for substrates requiring solution-phase processing, the patent describes effective protocols using inert solvents and bases at temperatures ranging from -80°C to +200°C, providing flexibility for different substrate profiles while maintaining high conversion rates.
Mechanistic Insights into Direct Amidation and Dioxolanone Coupling
The core chemical transformation involves the nucleophilic attack of the primary amine group of the 2-(3-alkoxy-4-hydroxyphenyl)-ethylamine on the carbonyl carbon of the alpha-hydroxycarboxylate ester or the dioxolanone ring. In the solvent-free embodiment, the thermal energy provided by heating the mixture above its melting point facilitates the molecular mobility required for this bimolecular reaction without the dilution effect of a solvent. This high-concentration environment favors the forward reaction, driving the equilibrium towards the amide product with virtually quantitative conversion. The use of dioxolanones (Formula IIIa) as acylating agents is particularly advantageous because the cyclic structure can offer enhanced reactivity or stability compared to linear esters, and the byproduct of the ring-opening amidation is a stable diol or ketone derivative that does not interfere with the product integrity. The patent specifies that the molar ratio of the alpha-hydroxycarboxylate to the amine can range from 1:2 to equimolar, with a 1:1.2 ratio being preferred to ensure complete consumption of the amine while minimizing excess reagent waste.
Impurity control is meticulously addressed through the optional addition of bases such as triethylamine or inorganic carbonates. Trace acidic impurities, which might originate from the hydrolysis of the ester starting materials, can catalyze side reactions or inhibit the amidation; the addition of a base neutralizes these acids, ensuring the reaction proceeds to completion efficiently. Furthermore, the process allows for the preparation of optically active intermediates by employing chiral alpha-hydroxycarboxylic acids. The stereochemical integrity of the chiral center is maintained during the amidation, allowing for the production of specific R- or S-enantiomers of the final fungicide intermediates. This capability is crucial for agrochemical applications where biological activity is often highly dependent on the specific spatial arrangement of the molecule. The resulting molten product can be used immediately for further conversion or purified via crystallization, offering a flexible workflow that adapts to the purity requirements of the subsequent synthetic steps.
How to Synthesize Alpha-Hydroxycarboxylic Acid Amides Efficiently
The synthesis protocol described in the patent provides a clear roadmap for producing these valuable intermediates with high efficiency. The process begins with the precise weighing and mixing of the amine and ester components, followed by controlled heating or dissolution in a selected solvent system. The choice between the solvent-free melt method and the solution-based method depends on the physical properties of the specific substituents involved, such as the melting points of the reactants and their thermal stability. For the melt method, careful temperature control is essential to prevent decomposition while ensuring the mixture remains fluid enough for effective mixing. For solution-based reactions, the selection of solvents like dichloromethane, toluene, or alcohols, combined with appropriate bases, allows for milder conditions suitable for more sensitive substrates. Detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- Mix Formula II 2-(3-alkoxy-4-hydroxyphenyl)-ethylamine with Formula III alpha-hydroxycarboxylate in equimolar amounts.
- Heat the mixture to a temperature range between the melting point and +100°C above the melting point for solvent-free reaction.
- Alternatively, react in an inert solvent with an organic or inorganic base at temperatures ranging from -80°C to +200°C.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this novel synthesis route offers compelling advantages for procurement managers and supply chain directors looking to optimize their sourcing strategies for agrochemical intermediates. The elimination of solvents in the preferred embodiment translates directly into substantial cost savings by removing the expenses associated with solvent purchase, storage, recovery, and disposal. This reduction in material usage also simplifies the regulatory compliance burden related to volatile organic compound (VOC) emissions, making the manufacturing process more environmentally sustainable and easier to permit in strict jurisdictions. Additionally, the high yield per volume means that existing reactor infrastructure can produce significantly more product in the same amount of time, effectively increasing capacity without capital expenditure on new equipment. These efficiencies contribute to a more resilient supply chain capable of meeting fluctuating market demands with greater agility.
- Cost Reduction in Manufacturing: The solvent-free nature of the primary embodiment removes the need for expensive organic solvents and the energy-intensive processes required to distill and recover them. This leads to a drastic simplification of the downstream processing workflow, as there is no solvent to strip off before crystallization or further reaction. The near-quantitative conversion rates minimize the loss of valuable starting materials, ensuring that the raw material input is maximized in the final product output. By reducing the number of unit operations and the complexity of waste treatment, the overall cost of goods sold (COGS) for these intermediates is significantly lowered, providing a competitive edge in pricing for the final fungicide formulations.
- Enhanced Supply Chain Reliability: The starting materials, including substituted phenethylamines and alpha-hydroxy esters, are derived from readily available commodity chemicals, reducing the risk of supply bottlenecks associated with exotic reagents. The robustness of the reaction conditions, which tolerate a range of temperatures and do not require ultra-dry environments in the melt method, ensures consistent batch-to-batch quality and reduces the likelihood of production delays due to technical failures. This reliability is critical for maintaining continuous production schedules for downstream agrochemical manufacturers who depend on a steady flow of high-purity intermediates to meet seasonal planting demands. The ability to scale the process from laboratory to commercial tonnage without fundamental changes to the chemistry further secures the long-term availability of these key building blocks.
- Scalability and Environmental Compliance: The process is inherently scalable, as the heat transfer dynamics in a melt reaction are manageable with standard industrial agitation and heating systems. The reduction in solvent waste aligns with green chemistry principles, significantly lowering the environmental impact of the manufacturing process. This eco-friendly profile is increasingly valued by global agrochemical companies seeking to reduce their carbon footprint and meet corporate sustainability goals. The simplified workup procedures also mean less hazardous waste is generated, lowering disposal costs and minimizing the environmental liability associated with chemical production. This combination of scalability and compliance makes the technology an attractive option for long-term strategic partnerships in the fine chemical sector.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from the specific embodiments and experimental data provided in the patent documentation, offering clarity on reaction conditions, substrate scope, and product handling. Understanding these details is essential for R&D teams evaluating the feasibility of integrating this process into their existing manufacturing lines. The answers reflect the versatility of the method across different substituents and its compatibility with various purification strategies.
Q: What are the primary advantages of the solvent-free melting method?
A: The solvent-free method offers significantly shorter reaction times and higher yield per volume compared to conventional solvent-based processes, with virtually quantitative conversion to the desired amide.
Q: Can this process produce enantiomerically enriched intermediates?
A: Yes, optical isomers can be prepared by reacting the amine with optically active R- or S-isomers of alpha-hydroxycarboxylic acids, allowing for the production of specific enantiomers required for biological activity.
Q: What types of bases are suitable for the solution-based variation?
A: Suitable bases include tertiary amines like triethylamine and N,N-diisopropylethylamine, as well as inorganic bases such as sodium carbonate and potassium carbonate, depending on the specific solvent system used.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Hydroxycarboxylic Acid Amides Supplier
NINGBO INNO PHARMCHEM stands at the forefront of custom synthesis and contract manufacturing, leveraging advanced technologies like the one described in CN100340541C to deliver superior agrochemical intermediates. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet your volume requirements with consistency and precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of alpha-hydroxycarboxylic acid amides meets the highest industry standards. Our commitment to technical excellence allows us to navigate the complexities of chiral synthesis and solvent-free processing, delivering products that facilitate the efficient manufacture of next-generation fungicides.
We invite you to collaborate with us to explore how this innovative synthesis route can optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific project needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our capabilities align with your strategic goals. Let us be your partner in transforming complex chemical challenges into commercial successes.
