Advanced Manufacturing Of Beta-Lactamase Inhibitor Intermediates Via Selective Hydrolysis
The global demand for effective antibacterial therapies continues to drive innovation in the synthesis of beta-lactamase inhibitors, critical components in combating resistant bacterial strains. Patent CN1823074A introduces a transformative methodology for preparing bicyclic heteroaryl carboxaldehydes, specifically targeting the synthesis of intermediates like 5,6-dihydro-4H-pyrrolo[1,2-b]pyrazole-2-carbaldehyde. This technical breakthrough addresses the longstanding inefficiencies associated with traditional routes, particularly the reliance on resource-intensive chromatographic separations. By leveraging a novel selective hydrolysis strategy, this process enables the direct isolation of key carboxylate salts from crude ester mixtures. For R&D directors and procurement specialists, this represents a significant opportunity to streamline supply chains for high-purity pharmaceutical intermediates. The following analysis details the mechanistic advantages and commercial viability of this route.
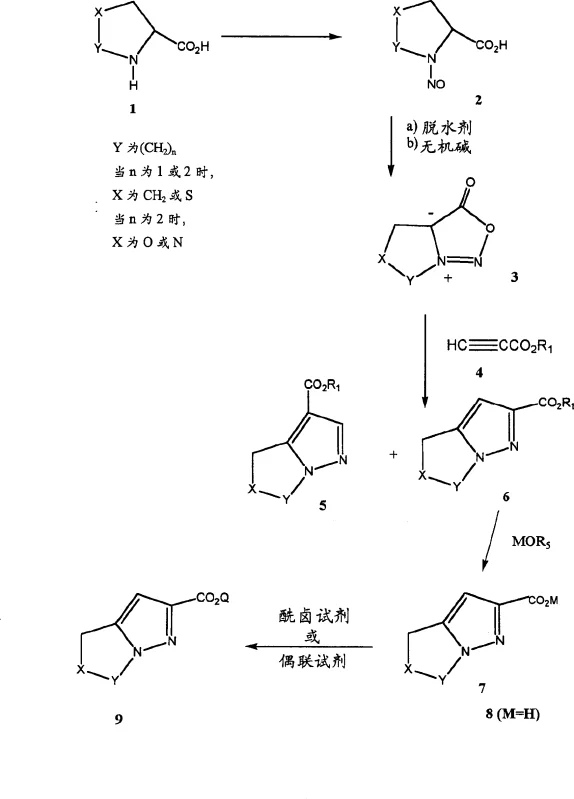
The limitations of conventional methods for synthesizing bicyclic heteroaryl aldehydes often stem from the difficulty in separating positional isomers generated during ring construction. Traditional approaches typically involve the formation of ester mixtures, requiring tedious chromatographic purification to isolate the desired 2-substituted isomer from the 3-substituted byproduct. This reliance on chromatography not only escalates manufacturing costs due to solvent consumption and silica gel usage but also severely limits scalability, making it unsuitable for multi-kilogram or ton-scale production required by the pharmaceutical industry. Furthermore, the multiple handling steps increase the risk of product degradation and impurity accumulation, compromising the stringent purity specifications needed for antibiotic intermediates.
In stark contrast, the novel approach disclosed in the patent circumvents these bottlenecks through a clever exploitation of differential hydrolysis rates. Instead of separating the ester isomers prior to hydrolysis, the process subjects the crude mixture of 2-ester and 3-ester directly to controlled alkaline hydrolysis conditions. Under optimized parameters, the desired 2-ester is selectively converted into its corresponding carboxylate salt, which possesses distinct solubility properties allowing it to crystallize directly from the reaction medium. This eliminates the need for chromatography entirely, replacing it with a robust crystallization unit operation. The result is a drastic simplification of the workflow, enhancing both throughput and environmental compliance by reducing solvent waste and hazardous solid waste generation.
Mechanistic Insights Into Meso-Ionic Synthon Cycloaddition
The core of this synthetic strategy relies on the generation of a reactive meso-ionic synthon, specifically a 1,3-dipole derived from the dehydration of a nitrosated amino acid. As illustrated in the reaction scheme, the initial nitrosylation of proline derivatives followed by dehydration with agents like trifluoroacetic anhydride yields a stable ylide intermediate. This meso-ionic species then undergoes a 1,3-dipolar cycloaddition with electron-deficient alkynes, such as ethyl propiolate. The regioselectivity of this cycloaddition is paramount, as it dictates the ratio of the resulting 2-carboxylate versus the 3-carboxylate isomers. The patent data indicates that solvent choice and temperature play critical roles, with 1,2-diethoxyethane or chlorobenzene at 120-125°C favoring the desired 2-isomer in ratios ranging from 1.5:1 to 3:1.
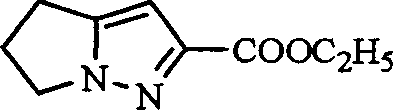
Following the cycloaddition, the control of impurities shifts to the hydrolysis phase. The mechanism involves the nucleophilic attack of hydroxide ions on the ester carbonyls. Due to steric and electronic differences between the 2-position and 3-position esters on the pyrrolo-pyrazole ring, the 2-ester hydrolyzes more rapidly or forms a less soluble salt under specific conditions. By carefully tuning the stoichiometry of the base (e.g., potassium hydroxide) and the solvent system (e.g., ethanol with controlled water content), the process ensures that the 2-carboxylate salt precipitates while the 3-ester remains in solution. This chemoselective crystallization is the key to achieving high purity without chromatography. Subsequent conversion to the Weinreb amide and reduction with lithium aluminum hydride provides the final aldehyde with excellent fidelity, minimizing over-reduction to the alcohol.
How To Synthesize 5,6-Dihydro-4H-Pyrrolo[1,2-B]Pyrazole-2-Carbaldehyde Efficiently
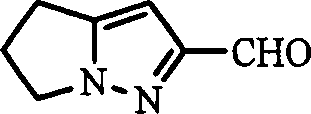
The synthesis of this critical beta-lactamase inhibitor intermediate follows a streamlined sequence designed for industrial scalability. The process begins with the nitrosation of the amino acid precursor, followed by cyclization to form the bicyclic core. The pivotal step involves the selective hydrolysis of the ester mixture to isolate the potassium salt of the acid, which is then converted to the Weinreb amide and finally reduced to the aldehyde. This route is specifically engineered to maximize yield and minimize purification steps, making it ideal for commercial manufacturing. The detailed standardized synthesis steps are provided in the guide below.
- Nitrosylation of amino acid precursors followed by dehydration to form a meso-ionic synthon.
- 1,3-Dipolar cycloaddition with propiolates to generate a mixture of regioisomeric esters.
- Selective hydrolysis and crystallization of the desired 2-carboxylate salt, avoiding chromatographic separation.
Commercial Advantages For Procurement And Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this chromatography-free process offers substantial strategic benefits. The elimination of column chromatography translates directly into significant cost reduction in pharmaceutical intermediate manufacturing. Chromatography is notoriously expensive due to the high cost of stationary phases, large volumes of high-purity solvents, and the labor-intensive nature of the operation. By replacing this with crystallization, the process drastically reduces the cost of goods sold (COGS) and shortens the production cycle time. Additionally, the ability to crystallize the intermediate salt directly from the crude reaction mixture enhances supply chain reliability by reducing dependency on specialized purification equipment and consumables that may face supply constraints.
Enhanced supply chain reliability is further supported by the robustness of the reagents and conditions employed. The use of common industrial solvents like ethanol and chlorobenzene, along with standard inorganic bases like potassium hydroxide, ensures that raw material sourcing is stable and geographically diverse. This reduces the risk of production delays caused by specialty chemical shortages. Furthermore, the process demonstrates excellent scalability, moving seamlessly from laboratory bench scale to multi-ton commercial production. The crystallization steps are inherently scalable and easier to control in large reactors compared to chromatographic columns, ensuring consistent quality and batch-to-batch reproducibility which is critical for regulatory compliance in antibiotic production.
Scalability and environmental compliance are also markedly improved. The reduction in solvent usage and the avoidance of silica gel waste contribute to a greener manufacturing profile, aligning with increasingly strict environmental regulations. The process generates less hazardous waste, simplifying disposal and lowering environmental compliance costs. Moreover, the high purity achieved through crystallization reduces the burden on downstream processing, ensuring that the final aldehyde meets the rigorous specifications required for coupling with beta-lactam cores. This efficiency supports the rapid scale-up of complex pharmaceutical intermediates, ensuring a steady supply of high-quality materials for the production of life-saving antibiotics.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the synthesis and application of these bicyclic heteroaryl intermediates. The answers are derived directly from the experimental data and process descriptions found in the patent literature, ensuring accuracy and relevance for technical decision-makers. Understanding these details is crucial for evaluating the feasibility of integrating this route into existing manufacturing workflows.
Q: How does this process avoid chromatography?
A: The process utilizes selective hydrolysis conditions where the desired 2-ester is hydrolyzed to a salt that crystallizes directly from the reaction mixture, leaving the 3-ester isomer in solution.
Q: What is the key intermediate formed before the aldehyde?
A: The key intermediate is the Weinreb amide (N-methoxy-N-methyl amide), which allows for controlled reduction to the aldehyde without over-reduction to the alcohol.
Q: What solvents are preferred for the cycloaddition step?
A: Preferred solvents include 1,2-diethoxyethane (DEE) or chlorobenzene, operated at temperatures between 120-125°C to optimize the isomer ratio.
Partnering With NINGBO INNO PHARMCHEM: Your Reliable 5,6-Dihydro-4H-Pyrrolo[1,2-B]Pyrazole-2-Carbaldehyde Supplier
As the demand for advanced beta-lactamase inhibitors grows, securing a reliable 5,6-dihydro-4H-pyrrolo[1,2-b]pyrazole-2-carbaldehyde supplier is essential for maintaining uninterrupted antibiotic production. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our technical team is adept at optimizing the selective hydrolysis and crystallization protocols described in patent CN1823074A to ensure maximum efficiency and yield. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the highest standards required for pharmaceutical applications.
We invite you to collaborate with us to optimize your supply chain for beta-lactamase inhibitor synthesis. Our experts can provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this chromatography-free route can lower your overall manufacturing expenses. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to a stable, high-quality supply of critical intermediates, ensuring your antibiotic development programs proceed without delay.
