Advanced Manufacturing of 2,3,5,6-Tetrachlorobenzoyl Chloride for Global Pharma Supply Chains
The landscape of fine chemical manufacturing is constantly evolving, driven by the need for higher purity intermediates and more robust supply chains. A recent breakthrough detailed in patent CN111689852A introduces a sophisticated preparation method for 2,3,5,6-tetrachlorobenzoyl chloride, a critical building block for various antibacterial drugs and agrochemical agents. This innovation addresses long-standing challenges in the synthesis of highly chlorinated aromatic systems, specifically targeting the issues of isomeric impurities and low yields that have plagued traditional methods. By shifting from a direct carbonation strategy to a Grignard exchange followed by Boc-anhydride trapping, the new process achieves a final product purity exceeding 99.0% with a total yield of 62-64%. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technological leap represents a significant opportunity to optimize production costs and ensure consistent quality for downstream applications in veterinary drugs and pesticides.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2,3,5,6-tetrachlorobenzoyl chloride has relied on the direct formation of a Grignard reagent from pentachlorobenzene and magnesium, followed by carboxylation with carbon dioxide. As illustrated in the legacy pathway below, this conventional route suffers from inherent inefficiencies. The direct insertion of magnesium into the carbon-chlorine bond of pentachlorobenzene is often sluggish and non-selective, leading to the formation of multiple isomeric Grignard species. Furthermore, the subsequent reaction with dry ice or gaseous CO2 requires stringent anhydrous conditions and often results in moderate yields, typically hovering around 58-60% for the acid formation step alone. The final conversion to the acid chloride using thionyl chloride adds another layer of complexity, often requiring pyridine catalysts and generating significant waste. These factors combined create a bottleneck for cost reduction in pharmaceutical intermediate manufacturing, as the purification of isomeric byproducts becomes increasingly difficult and expensive at scale.
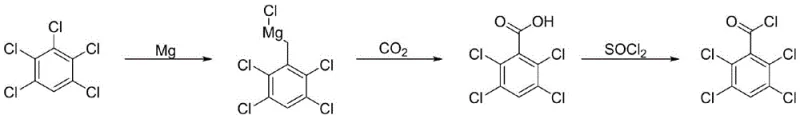
The Novel Approach
In stark contrast, the novel methodology disclosed in the patent employs a Halogen-Magnesium exchange strategy, which offers superior kinetic control and selectivity. Instead of relying on the direct insertion of magnesium metal into the aryl chloride, the process generates an isopropyl Grignard reagent in situ using isopropyl bromide or chloride and magnesium. This reactive species then performs a rapid exchange with the pentachlorobenzene substrate at controlled temperatures (65-70°C), effectively minimizing side reactions. The resulting 1,2,4,5-tetrachlorophenyl magnesium chloride is then trapped with di-tert-butyl dicarbonate (Boc2O) at low temperatures (-25 to -15°C) to form a stable tert-butyl ester. This ester intermediate serves as a protected form of the acid, allowing for easier handling and purification before the final deprotection and chlorination step. This strategic detour through the ester significantly enhances the overall process robustness, making it an ideal candidate for the commercial scale-up of complex pharmaceutical intermediates.
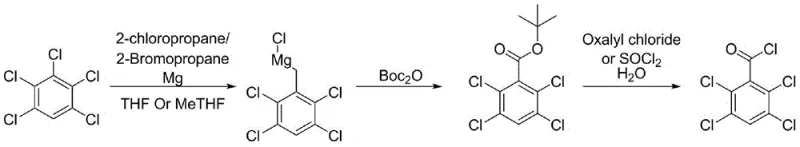
Mechanistic Insights into Grignard-Halogen Exchange and Boc-Trapping
The core of this technological advancement lies in the mechanistic elegance of the Halogen-Magnesium exchange. Unlike direct metal insertion, which can be erratic with poly-halogenated substrates, the exchange reaction is driven by the formation of a more stable Grignard species. The use of stabilizers such as zinc chloride or lithium chloride further facilitates this exchange by coordinating with the magnesium center, enhancing its reactivity towards the specific chlorine atom intended for substitution. This ensures that the Grignard reagent forms predominantly at the desired position, drastically reducing the generation of regio-isomers that would otherwise contaminate the final product. The subsequent reaction with Boc2O is a nucleophilic acyl substitution where the Grignard reagent attacks one of the carbonyl carbons of the anhydride. This step is highly exothermic, necessitating the strict temperature control observed in the patent (-25 to -15°C), which prevents the decomposition of the sensitive Grignard reagent and ensures high conversion to the tert-butyl ester.
From an impurity control perspective, this route offers distinct advantages. In the traditional CO2 route, any unreacted Grignard reagent or side products can lead to complex mixtures upon acidic workup. However, in the new process, the tert-butyl ester acts as a 'mask' for the carboxylic acid. This allows for a quenching step using dilute hydrochloric acid or saturated ammonium chloride that is far more forgiving. The ester can be isolated via crystallization or pulping with n-heptane, as demonstrated in Example 1 where a purity of 96.9% was achieved before the final step. The final conversion to the acid chloride using thionyl chloride or oxalyl chloride in the presence of catalytic water proceeds cleanly, with the volatile byproducts (isobutylene and CO2 from the Boc group, plus SO2/HCl from the chlorination) easily removed under reduced pressure. This mechanism ensures that the final distillation yields a product with purity greater than 99.0%, meeting the stringent specifications required for high-value API synthesis.
How to Synthesize 2,3,5,6-Tetrachlorobenzoyl Chloride Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for laboratory and pilot-scale production, emphasizing safety and reproducibility. The process begins with the careful preparation of the Grignard exchange reagent, followed by the low-temperature trapping with Boc anhydride, and concludes with the acyl chlorination. Each step is optimized to maximize yield while minimizing hazardous waste. For technical teams looking to implement this, the detailed standardized synthesis steps are provided in the guide below, which breaks down the specific molar ratios, solvent choices, and temperature profiles required for success.
- Perform Grignard exchange on pentachlorobenzene using magnesium and isopropyl halide with a stabilizer in THF or 2-MeTHF at 65-70°C.
- React the resulting Grignard reagent with di-tert-butyl dicarbonate (Boc2O) at low temperature (-25 to -15°C) to form the tert-butyl ester.
- Convert the ester to the final acid chloride using thionyl chloride or oxalyl chloride with catalytic water, followed by vacuum distillation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers compelling economic and operational benefits beyond mere chemical yield. The shift away from direct CO2 carboxylation eliminates the need for specialized high-pressure equipment or the handling of large volumes of dry ice, simplifying the infrastructure requirements for production. Furthermore, the use of commodity chemicals like isopropyl bromide and Boc anhydride ensures that raw material sourcing remains stable and cost-effective, mitigating the risks associated with supply chain disruptions for exotic reagents. The ability to purify the intermediate ester via simple pulping and filtration before the final step reduces the load on downstream purification units, leading to significant operational efficiencies.
- Cost Reduction in Manufacturing: The elimination of expensive transition metal catalysts and the use of standard fine chemical unit operations drastically simplify the production process. By avoiding the formation of difficult-to-separate isomers early in the synthesis, the need for costly chromatographic purification or multiple recrystallizations is removed. The high overall yield of 62-64% across three steps translates directly to lower raw material consumption per kilogram of finished product, driving down the cost of goods sold (COGS) substantially.
- Enhanced Supply Chain Reliability: The robustness of the Grignard exchange reaction, supported by stabilizers like ZnCl2 or LiCl, makes the process less sensitive to minor variations in raw material quality or environmental conditions. This reliability ensures consistent batch-to-batch quality, which is critical for maintaining long-term contracts with pharmaceutical clients. Additionally, the final purification via vacuum distillation allows for the production of a uniform, high-purity solid that meets strict regulatory standards, reducing the risk of batch rejection.
- Scalability and Environmental Compliance: The process utilizes solvents like THF and 2-MeTHF, which are well-understood in industrial settings and can be efficiently recovered and recycled. The avoidance of heavy metal catalysts aligns with modern green chemistry principles and simplifies waste treatment protocols. The continuous nature of the reaction steps, particularly the seamless transition from Grignard formation to esterification, facilitates scaling from pilot plants to multi-ton commercial production without the need for fundamental process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of 2,3,5,6-tetrachlorobenzoyl chloride. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: What are the key advantages of the Boc-protection route over direct carbonation?
A: The Boc-protection route avoids the formation of isomeric impurities common in direct CO2 carboxylation and allows for easier purification via distillation of the final acid chloride, achieving purity >99.0%.
Q: Which solvents are compatible with this Grignard exchange process?
A: The patent specifies Tetrahydrofuran (THF) and 2-Methyltetrahydrofuran (2-MeTHF) as effective solvents, with 2-MeTHF offering potential benefits for greener processing and easier recovery.
Q: How is the reaction exotherm managed during the Grignard formation?
A: The process utilizes a controlled initiation at 15-30°C followed by dropwise addition to maintain the reaction temperature between 15-45°C during initiation and 65-70°C for completion, ensuring safety and selectivity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2,3,5,6-Tetrachlorobenzoyl Chloride Supplier
At NINGBO INNO PHARMCHEM, we understand that the successful commercialization of new therapeutic agents depends on the availability of high-quality intermediates. Our team of expert chemists has extensively evaluated the route described in CN111689852A and confirmed its viability for large-scale manufacturing. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with precision and consistency. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, including the detection of trace isomeric impurities that could impact downstream biological activity.
We invite you to collaborate with us to leverage this advanced synthesis technology for your next project. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
