Revolutionizing Hydrazide Manufacturing: A Deep Dive into Triazine-Mediated Carboxylic Acid Activation
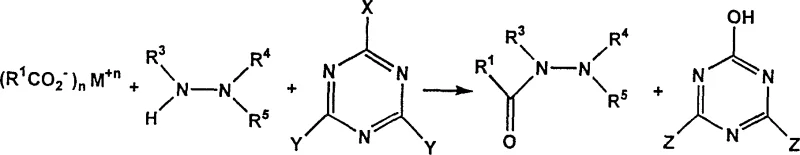
The chemical landscape for synthesizing high-value hydrazide intermediates is undergoing a significant transformation driven by the need for safer, more cost-effective, and environmentally sustainable manufacturing processes. Patent CN1219752C introduces a groundbreaking methodology that fundamentally alters the conventional approach to preparing monoacylhydrazines and diacylhydrazines, which are critical precursors in the production of advanced agrochemicals and pharmaceutical compounds. This innovation bypasses the traditional reliance on highly reactive and corrosive acyl chlorides, opting instead for a sophisticated coupling strategy utilizing chloro- or fluoro-substituted 1,3,5-triazines. By enabling the direct reaction of carboxylic acids with hydrazines in the presence of these triazine activators, the technology offers a robust pathway that mitigates the generation of hazardous hydrogen chloride byproducts and simplifies the overall synthetic workflow. For industry leaders seeking a reliable agrochemical intermediate supplier, understanding the mechanistic advantages of this patent is crucial for optimizing supply chain resilience and reducing long-term operational expenditures associated with waste management and equipment corrosion.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial preparation of acylhydrazines has predominantly relied on the reaction between hydrazines and acyl chlorides derived from parent carboxylic acids. While technically straightforward, this legacy approach is fraught with significant economic and safety drawbacks that hinder efficient large-scale manufacturing. The conversion of carboxylic acids to acyl chlorides typically necessitates the use of aggressive chlorinating agents, which not only increases raw material costs substantially but also generates stoichiometric amounts of hydrogen chloride gas that require complex scrubbing systems to manage. Furthermore, acyl chlorides are inherently unstable in the presence of moisture, posing severe handling risks and requiring stringent anhydrous conditions that drive up energy consumption for solvent drying. The high reactivity of acyl chlorides also often leads to poor regioselectivity, resulting in complex mixtures of mono- and di-acylated products that demand expensive and time-consuming purification steps to isolate the desired intermediate, thereby eroding profit margins and extending production lead times for high-purity specialty chemicals.
The Novel Approach
In stark contrast, the novel triazine-mediated process described in the patent data presents a paradigm shift by utilizing stable and inexpensive carboxylic acid reagents directly, effectively circumventing the need for pre-activation into acid chlorides. This method leverages the electrophilic nature of chloro-triazines, such as cyanuryl chloride or its methoxy-substituted analogs, to activate the carboxylic acid in situ within the reaction mixture. This strategic modification allows for the selective formation of the desired hydrazide product with high specificity, virtually eliminating the formation of unwanted isomers or over-acylated byproducts that plague conventional methods. The operational simplicity of this approach, which proceeds efficiently in common polar solvents like acetonitrile at mild temperatures ranging from 0°C to 50°C, translates directly into enhanced process safety and reduced capital expenditure on specialized corrosion-resistant reactor infrastructure. For procurement managers focused on cost reduction in agrochemical intermediate manufacturing, this technology represents a tangible opportunity to streamline operations and achieve substantial cost savings through simplified raw material sourcing and waste treatment protocols.
Mechanistic Insights into Triazine-Mediated Coupling
The core chemical innovation lies in the ability of the 1,3,5-triazine ring system to act as a potent dehydrating coupling agent that facilitates the nucleophilic attack of the hydrazine on the carboxylic acid carbonyl carbon. In this mechanism, the carboxylic acid is first deprotonated by a non-nucleophilic base, such as N-methylmorpholine or triethylamine, to form a reactive carboxylate anion. This anion then attacks the electron-deficient carbon of the chloro-triazine, displacing a chloride ion and forming a mixed anhydride-like intermediate that is highly susceptible to nucleophilic substitution. When the hydrazine reagent is introduced, it selectively attacks this activated carbonyl species, leading to the formation of the monoacylhydrazine bond while releasing a hydroxy-triazine byproduct. This pathway is thermodynamically favorable and kinetically controlled, ensuring that the reaction proceeds to completion without the violent exotherms associated with acid chloride chemistry. The versatility of the triazine reagent allows for fine-tuning of the reaction kinetics by varying the number of halogen substituents, providing chemists with precise control over the activation rate to match the specific steric and electronic properties of diverse carboxylic acid substrates.
Furthermore, the impurity profile of the resulting product is significantly improved due to the mild nature of the reaction conditions and the high chemoselectivity of the triazine activator. Unlike acid chloride routes where residual chlorine can persist and complicate downstream processing, the byproducts generated in this triazine-mediated process, primarily hydroxy-triazines, are chemically distinct and can be readily removed through standard aqueous workup procedures involving acid and base washes. The patent explicitly details that no isomeric products or di-acylates are detected when synthesizing monoacylhydrazines, which is a critical quality attribute for R&D directors concerned with the purity and consistency of API intermediates. This high level of selectivity reduces the burden on purification units, minimizes product loss during crystallization or chromatography, and ensures that the final material meets stringent specifications required for regulatory submission. The ability to produce high-purity OLED material or pharmaceutical precursors with such a clean impurity profile underscores the technical superiority of this method over traditional peptide coupling reagents like DCC, which often leave behind difficult-to-remove urea byproducts.
How to Synthesize Substituted Hydrazide Efficiently
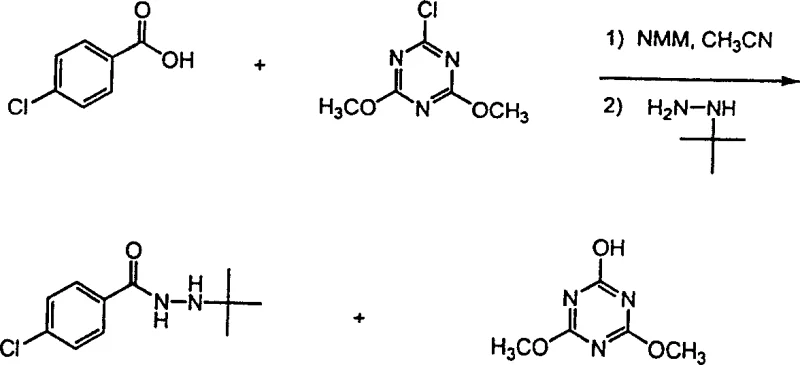
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to reagent stoichiometry and temperature control to maximize yield and minimize byproduct formation. The process begins with the dissolution of the chosen carboxylic acid and the triazine coupling agent in a suitable polar aprotic solvent, followed by the controlled addition of a base to generate the active carboxylate species. Once the activated intermediate is formed, the hydrazine component is added, and the mixture is stirred until analytical monitoring confirms complete consumption of the starting materials. The detailed standardized synthesis steps see the guide below, which outlines the specific molar ratios and workup procedures validated in the patent examples to ensure reproducible results across different batches.
- Dissolve the carboxylic acid and chloro-triazine reagent (e.g., cyanuryl chloride) in a polar aprotic solvent such as acetonitrile under cooling conditions.
- Add a non-nucleophilic base like N-methylmorpholine dropwise to activate the carboxylic acid into a carboxylate anion while maintaining low temperature.
- Introduce the hydrazine or substituted hydrazine reagent to the mixture, stir until reaction completion, and purify the resulting monoacylhydrazine via extraction.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this triazine-based synthesis protocol offers profound advantages for supply chain stability and overall manufacturing economics, addressing several pain points inherent in the traditional production of hydrazide derivatives. By eliminating the requirement for acyl chlorides, manufacturers can source cheaper and more stable carboxylic acid starting materials directly, thereby reducing raw material inventory costs and mitigating the risks associated with transporting hazardous corrosive substances. The simplified reaction workflow also translates into shorter batch cycle times, as the separate step of converting acid to acid chloride is removed entirely, allowing for increased throughput in existing reactor vessels without the need for significant capital investment in new equipment. For supply chain heads concerned with the commercial scale-up of complex polymer additives or agrochemical intermediates, this process offers a more robust and predictable production timeline, reducing the likelihood of delays caused by equipment failure due to corrosion or unexpected safety incidents related to hazardous reagent handling.
- Cost Reduction in Manufacturing: The elimination of the acyl chloride formation step results in significant cost savings by removing the need for expensive chlorinating agents and the associated waste disposal fees for hydrogen chloride byproducts. Additionally, the use of inexpensive triazine reagents like cyanuryl chloride, which are produced on a massive industrial scale, further drives down the cost of goods sold compared to specialized coupling agents. The reduction in purification complexity also contributes to lower operational expenses, as less solvent and energy are required to achieve the necessary purity levels for commercial distribution. These cumulative efficiencies allow for a more competitive pricing structure while maintaining healthy profit margins, making the technology highly attractive for cost-sensitive markets.
- Enhanced Supply Chain Reliability: Utilizing stable carboxylic acids and solid triazine reagents enhances supply chain reliability by reducing dependence on volatile liquid reagents that may have limited availability or strict transportation regulations. The mild reaction conditions reduce the risk of unplanned shutdowns due to safety incidents, ensuring consistent delivery schedules for downstream customers. Furthermore, the versatility of the method allows for the use of a wide range of commercially available hydrazines and acids, providing flexibility to switch suppliers or raw material grades without compromising the integrity of the final product. This adaptability is crucial for maintaining continuity of supply in a fluctuating global market environment.
- Scalability and Environmental Compliance: The process is inherently scalable, moving seamlessly from gram-scale laboratory optimization to multi-ton commercial production without encountering the heat transfer limitations often seen with highly exothermic acid chloride reactions. The reduction in hazardous waste generation aligns with increasingly stringent environmental regulations, lowering the compliance burden and potential liability for manufacturing facilities. By minimizing the release of corrosive gases and toxic byproducts, companies can improve their sustainability metrics and corporate social responsibility profiles, which are becoming key differentiators in B2B procurement decisions. This environmental advantage also simplifies the permitting process for new production lines, accelerating time-to-market for new products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this triazine-mediated hydrazide synthesis technology, based on the specific embodiments and data provided in the patent documentation. These insights are designed to clarify the operational benefits and address potential concerns regarding process adaptation for existing manufacturing lines. Understanding these details is essential for technical teams evaluating the feasibility of integrating this novel chemistry into their current production portfolios.
Q: Why is the triazine-mediated method superior to traditional acyl chloride routes?
A: The triazine method eliminates the need to convert carboxylic acids into corrosive acyl chlorides, thereby reducing equipment maintenance costs, minimizing hazardous HCl byproduct waste, and improving overall process safety and environmental compliance.
Q: What are the primary purification challenges in this synthesis?
A: The main challenge involves separating the desired hydrazide from the hydroxy-triazine byproduct. However, the patent outlines effective purification strategies using standard extraction and washing protocols with aqueous acid and base solutions to ensure high purity.
Q: Can this process be scaled for commercial production of diacylhydrazides?
A: Yes, the process is highly scalable. The mild reaction conditions (0°C to 50°C) and the use of stable reagents allow for safe scale-up from kilogram to multi-ton production without the severe exothermic risks associated with acyl chloride handling.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Substituted Hydrazide Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the triazine-mediated coupling technology described in CN1219752C and have integrated similar advanced synthetic strategies into our CDMO service offerings to deliver superior value to our global partners. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory discovery to industrial reality is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of substituted hydrazide meets the exacting standards required by the pharmaceutical and agrochemical industries. Our commitment to technical excellence allows us to navigate the complexities of process optimization, ensuring high yields and consistent quality even for the most challenging molecular architectures.
We invite you to collaborate with us to leverage these innovative manufacturing capabilities for your next project. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements and target specifications. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our expertise can enhance your supply chain efficiency and reduce your overall production costs. Let us be your trusted partner in bringing high-quality chemical intermediates to market faster and more economically.
