Advanced Manufacturing of Vinylimidazole Derivatives: A Safer Route for Global Agrochemical Supply Chains
Introduction to Patent CN1013443B and Process Innovation
The global demand for high-performance antifungal agents in both agriculture and human medicine has necessitated a re-evaluation of traditional synthetic routes for key intermediates. Patent CN1013443B introduces a transformative methodology for producing vinylimidazole derivatives, specifically addressing the critical bottlenecks found in earlier United States Patent 4,328,348. This intellectual property represents a paradigm shift from hazardous, low-yield protocols to a robust, scalable industrial process. By replacing pyrophoric reagents with stable alkali metal hydroxides and optimizing solvent systems to prevent decomposition, this technology offers a reliable agrochemical intermediate supplier pathway that aligns with modern safety and environmental standards. The core innovation lies not just in the final alkylation step, but in the strategic management of reaction byproducts, turning what was once waste into valuable feedstock through acid-catalyzed conversion.
For R&D directors and process engineers, the significance of this patent extends beyond mere yield improvements; it fundamentally alters the risk profile of manufacturing potent antifungal intermediates. The traditional reliance on sodium hydride in dry dimethylformamide (DMF) created a volatile environment prone to thermal runaways and solvent degradation. In contrast, the disclosed method utilizes dimethyl sulfoxide (DMSO) and potassium hydroxide, creating a chemically stable environment that minimizes side reactions. This stability is paramount for maintaining stringent purity specifications required by regulatory bodies for pharmaceutical and agrochemical applications. Furthermore, the ability to recycle the bis-imidazole byproduct back into the main production stream demonstrates a commitment to atom economy that resonates deeply with contemporary green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
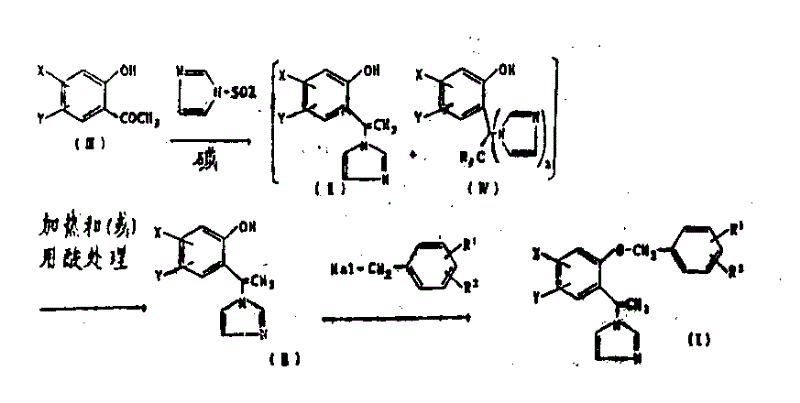
Prior art methodologies, particularly those relying on sodium hydride as a base in DMF solvents, present severe logistical and safety challenges for commercial scale-up. Sodium hydride is notoriously difficult to handle on a multi-ton scale due to its pyrophoric nature, requiring specialized inert atmosphere equipment and rigorous safety protocols that drive up capital expenditure. Moreover, DMF is chemically unstable under the strongly basic conditions required for these reactions, leading to decomposition into dimethylamine. This decomposition product acts as a nucleophile, reacting with benzyl halides to form tertiary amine impurities that are structurally similar to the target molecule and extremely difficult to separate. Consequently, the isolation of the pure vinylimidazole derivative becomes a costly and inefficient process, often resulting in suboptimal yields that fail to meet the economic thresholds for bulk chemical production.
The Novel Approach
The novel approach detailed in the patent data circumvents these issues by substituting sodium hydride with safer, cheaper alkali metal hydroxides such as potassium hydroxide or sodium hydroxide. This substitution alone eliminates the fire hazard associated with hydride reagents, allowing for simpler reactor designs and reduced insurance premiums. More critically, the switch to a DMSO-based solvent system prevents the formation of dimethylamine, thereby eliminating a major source of contamination. The process also introduces a clever chemical strategy where the inevitable formation of the bis-imidazole byproduct is not treated as a loss but as a reversible intermediate. By applying heat or acid treatment, the bis-imidazole species is quantitatively converted back into the desired mono-imidazole precursor. This dual optimization of safety and material efficiency establishes a new benchmark for cost reduction in agrochemical intermediate manufacturing.
Mechanistic Insights into Acid-Catalyzed Bis-Imidazole Conversion
A deeper mechanistic analysis reveals that the formation of the bis-imidazole derivative (Compound IV) occurs when excess imidazole or its reactive sulfinyl species attacks the intermediate. In conventional processes, this byproduct represents a permanent yield loss, necessitating complex purification steps to remove it from the mono-imidazole product (Compound II). However, the inventors discovered that the bond connecting the second imidazole ring in Compound IV is labile under acidic or thermal conditions. When subjected to acids such as p-toluenesulfonic acid or Lewis acids like boron trifluoride etherate, or simply heated in aromatic solvents like toluene, the bis-imidazole undergoes a cleavage reaction. This reaction releases free imidazole and regenerates the vinylimidazole double bond characteristic of Compound II. This mechanistic insight allows manufacturers to operate the initial condensation step with less stringent stoichiometric control, knowing that any over-reaction can be corrected downstream.
From an impurity control perspective, this mechanism is revolutionary. It implies that the purity of the final intermediate is not solely dependent on the precision of the initial mixing but can be 'polished' in a subsequent step. For example, experimental data shows that treating the crude mixture with 0.1 molar equivalents of p-toluenesulfonic acid in refluxing toluene can convert nearly all bis-imidazole contaminants into the desired product, achieving purity levels exceeding 99%. This reduces the burden on upstream quality control and allows for more flexible batch processing. The ability to tune the ratio of mono- to bis-imidazole through simple thermal or acidic workup provides process chemists with a powerful tool for optimizing throughput without sacrificing quality, ensuring that the final active pharmaceutical ingredient (API) or agrochemical meets rigorous regulatory standards.
How to Synthesize Vinylimidazole Derivatives Efficiently
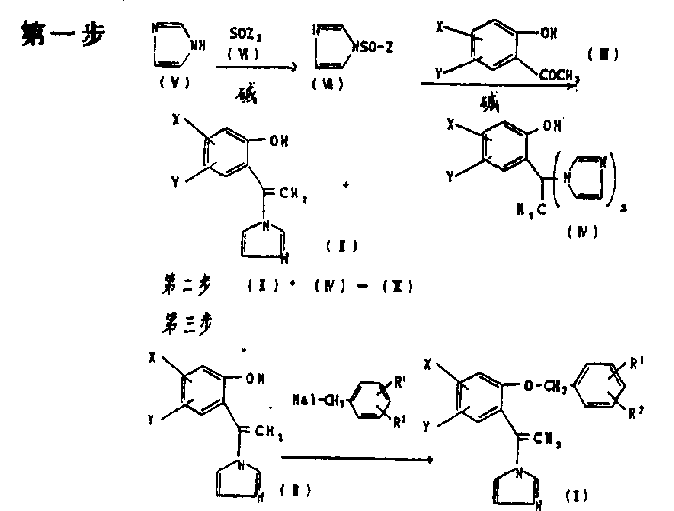
The synthesis protocol outlined in the patent is divided into three distinct operational phases, each designed to maximize yield and safety. The first phase involves the in-situ generation of the reactive imidazole species, typically 1-halosulfinyl-1H-imidazole, using thionyl chloride and a tertiary amine base at controlled low temperatures between 0°C and 10°C. This reagent is then immediately reacted with o-hydroxyacetophenone derivatives to form the crude intermediate mixture. The second phase is the critical conversion step where the crude mixture, containing both mono- and bis-imidazoles, is treated with acid or heated to ensure complete conversion to the mono-form. Finally, the third phase involves the alkylation of the purified mono-imidazole with specific benzyl halides in a DMSO solvent system using potassium hydroxide. Detailed standardized operating procedures for each of these stages are essential for reproducibility.
- Prepare 1-halosulfinyl-1H-imidazole by reacting imidazole with thionyl halide in the presence of an organic base like triethylamine at 0-10°C.
- React the prepared reagent with o-hydroxyacetophenone to form a mixture of mono-imidazole and bis-imidazole byproducts, then convert the byproduct to the desired mono-form via acid treatment or heating.
- Perform the final alkylation of the mono-imidazole intermediate with benzyl halides using potassium hydroxide in a DMSO-based solvent system to yield the target vinylimidazole derivative.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process translates directly into tangible operational benefits and risk mitigation. The elimination of sodium hydride removes a high-risk material from the supply chain, reducing the need for specialized hazardous material transport and storage facilities. Furthermore, the use of commodity chemicals like potassium hydroxide and DMSO, which are widely available and inexpensive compared to specialized hydride reagents, ensures a stable and cost-effective raw material base. The process's ability to convert byproducts into main products significantly enhances the effective yield per kilogram of starting material, effectively lowering the cost of goods sold (COGS) without requiring expensive chromatography equipment. This efficiency makes the supply of these critical intermediates more resilient to market fluctuations in raw material pricing.
- Cost Reduction in Manufacturing: The replacement of expensive and hazardous sodium hydride with cheap alkali metal hydroxides drastically lowers reagent costs. Additionally, the prevention of solvent decomposition in DMSO compared to DMF reduces solvent consumption and waste disposal fees. The conversion of bis-imidazole byproducts into saleable product maximizes raw material utilization, ensuring that every kilogram of input contributes to the final output, thereby driving down the unit cost of production significantly.
- Enhanced Supply Chain Reliability: By utilizing widely available commodity reagents such as potassium hydroxide and standard solvents like dichloromethane and toluene, the process avoids dependency on niche suppliers of hazardous materials. The robustness of the reaction conditions, which tolerate a wider range of temperatures and stoichiometries due to the byproduct conversion step, reduces the likelihood of batch failures. This reliability ensures consistent delivery schedules and minimizes the risk of production stoppages due to reagent shortages or safety incidents.
- Scalability and Environmental Compliance: The process is inherently scalable because it avoids exothermic hazards associated with hydride additions, allowing for larger batch sizes in standard reactors. The reduction in hazardous waste generation, particularly the avoidance of DMF decomposition products and hydride residues, simplifies wastewater treatment and aligns with increasingly strict environmental regulations. This compliance facilitates easier permitting for plant expansions and reduces the long-term liability associated with hazardous waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines. The answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making.
Q: Why is the switch from Sodium Hydride to Potassium Hydroxide critical for safety?
A: Sodium hydride is pyrophoric and poses significant explosion risks during large-scale handling. The patented process utilizes alkali metal hydroxides like KOH, which are non-pyrophoric, significantly reducing operational hazards and insurance costs for manufacturing facilities.
Q: How does this process manage the formation of bis-imidazole byproducts?
A: The process uniquely converts the bis-imidazole byproduct back into the desired mono-imidazole intermediate through acid-catalyzed treatment or thermal rearrangement. This eliminates the need for complex chromatographic separation, drastically improving overall material throughput and yield.
Q: What are the solvent advantages of using DMSO over DMF in this synthesis?
A: Unlike DMF, which decomposes under basic conditions to form dimethylamine that reacts with benzyl halides to create difficult-to-remove impurities, DMSO remains stable. This stability ensures higher product purity and simplifies downstream purification processes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vinylimidazole Derivatives Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the development of next-generation antifungal therapies and crop protection agents. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and utilize rigorous QC labs to verify that every batch of vinylimidazole derivative meets the exacting standards required by global regulatory agencies. Our commitment to safety and efficiency mirrors the innovations found in patent CN1013443B, allowing us to offer products that are not only chemically superior but also economically competitive.
We invite potential partners to engage with our technical procurement team to discuss how our manufacturing capabilities can support your specific project needs. Whether you require a Customized Cost-Saving Analysis for your current supply chain or need specific COA data and route feasibility assessments for new analogues, we are equipped to provide comprehensive solutions. By leveraging our advanced synthesis platforms, we can help you reduce lead time for high-purity agrochemical intermediates and secure a stable supply of critical materials for your commercial operations.
