Industrial Scale Synthesis of 4,6-Diaminopyrimido[5,4-d]pyrimidine for Global Pharma
The pharmaceutical landscape is constantly evolving, driven by the need for more efficient and safer synthesis routes for complex active pharmaceutical ingredients. Patent CN1500091A introduces a groundbreaking preparation method for 4,6-diaminopyrimido[5,4-d]pyrimidines, a class of compounds known for their potent inhibition of tyrosine kinase-mediated signal transduction. This technology represents a significant leap forward in process chemistry, addressing critical bottlenecks that have historically plagued the industrial production of these valuable therapeutic intermediates. By leveraging a novel sequence of protection, reduction, and oxidation steps, the invention enables the transformation of readily available starting materials into high-purity final products with exceptional space-time yields. For R&D directors and procurement specialists, understanding the nuances of this patent is essential, as it offers a pathway to reduce manufacturing costs while enhancing supply chain reliability. The method avoids the use of hazardous sulfur-containing reagents found in prior art, such as WO97/32880, thereby mitigating environmental risks and simplifying waste management protocols. This report delves deep into the technical merits and commercial implications of this synthesis, providing a comprehensive analysis for stakeholders aiming to optimize their production of kinase inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 4,6-diaminopyrimido[5,4-d]pyrimidines has been fraught with significant challenges that hinder large-scale commercial viability. Previous methods, such as those described in WO97/32880, relied heavily on starting materials like 4-anilino-6-methylsulfinylpyrimido[5,4-d]pyrimidine and 4-anilino-6-methylsulfonylpyrimido[5,4-d]pyrimidine. While chemically feasible on a small laboratory scale, these routes present severe drawbacks when translated to industrial manufacturing environments. The primary issue lies in the reaction byproducts; the conversion of sulfinyl or sulfonyl groups often results in the release of toxic thiols, which possess foul odors and pose significant health and safety risks to plant personnel. Furthermore, the removal of these sulfur-containing impurities requires extensive and costly purification steps, often involving chromatography, which is impractical for multi-kilogram or ton-scale production. The starting materials themselves are not only difficult to obtain in bulk quantities but also contribute to a complex impurity profile that complicates regulatory approval processes. Consequently, manufacturers have faced inflated production costs, extended lead times, and increased environmental compliance burdens, creating a pressing need for a cleaner, more robust synthetic alternative.
The Novel Approach
The methodology outlined in patent CN1500091A offers a transformative solution by fundamentally reimagining the synthetic pathway to avoid sulfur-based intermediates entirely. Instead of relying on problematic sulfinyl or sulfonyl precursors, this novel approach utilizes 2,4,6,8-tetrachloropyrimido[5,4-d]pyrimidine, also known as tetrachlorohomopurine, as the foundational building block. This starting material is readily available and amenable to large-scale procurement, immediately addressing supply chain vulnerabilities. The process involves a strategic sequence of amination, protection, and reduction steps that maintain high chemical integrity throughout the synthesis. By employing chloro-substituted intermediates, the reaction avoids the generation of toxic thiol byproducts, thereby eliminating the need for complex odor control systems and hazardous waste treatment associated with sulfur chemistry. Moreover, the intermediates generated in this pathway, such as the protected Formula II compounds, exhibit high purity and can be carried forward to subsequent steps without the need for chromatographic purification. This streamlined workflow not only accelerates the production timeline but also drastically reduces solvent consumption and operational expenses, making it an ideal candidate for cost-effective commercial manufacturing of high-value pharmaceutical intermediates.
Mechanistic Insights into Protected Intermediate Formation and Reduction
The core of this innovative synthesis lies in the precise manipulation of the pyrimido-pyrimidine scaffold through a series of well-controlled chemical transformations. A critical stage in this process involves the formation of the protected intermediate, designated as Formula II in the patent documentation. This structure serves as a pivotal junction where the core heterocyclic system is functionalized with specific amine groups while maintaining stability through protecting groups like acetyl or benzoyl moieties. The introduction of these protecting groups is crucial, as it prevents unwanted side reactions during the subsequent amination steps, ensuring that the nucleophilic attack occurs selectively at the desired positions on the ring system. Following the construction of the protected scaffold, the process employs a rigorous deprotection step, typically using alkaline hydrolysis, to reveal the reactive amine functionalities. This is immediately followed by a reduction phase, which can be achieved via catalytic hydrogenation using palladium on carbon or through chemical reduction with hydriodic acid and red phosphorus. This reduction step is vital for converting the chlorinated or oxidized precursors into the saturated or partially saturated intermediates required for the final oxidation to the target diamino compound.
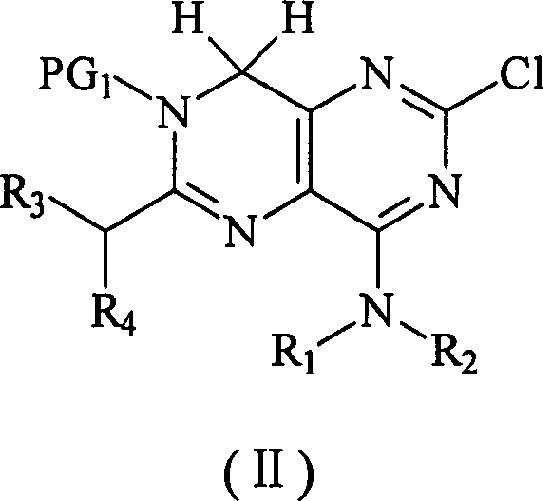
Following the reduction phase, the synthesis culminates in a controlled oxidation step that restores the aromaticity or specific oxidation state required for the final 4,6-diaminopyrimido[5,4-d]pyrimidine product. This oxidation is typically performed using peroxodisulfates or hydrogen peroxide in an acidic aqueous medium, conditions that are both cost-effective and environmentally benign compared to heavy metal oxidants. The mechanistic elegance of this route ensures that impurities are minimized at every stage, as the reaction conditions are optimized to favor the formation of the desired isomer while suppressing side products. For R&D teams, understanding this mechanism is key to troubleshooting potential scale-up issues, as the balance between reduction and oxidation potentials must be carefully managed to prevent over-reduction or degradation of the sensitive heterocyclic core. The ability to perform these transformations without chromatographic purification highlights the robustness of the chemistry, allowing for a telescoped process where crude intermediates can be directly utilized in subsequent steps. This level of process intensification is a hallmark of modern green chemistry and significantly enhances the overall economic feasibility of the manufacturing route.
How to Synthesize 4,6-Diaminopyrimido[5,4-d]pyrimidine Efficiently
Implementing this synthesis requires a detailed understanding of the reaction parameters and safety protocols associated with each step. The process begins with the reaction of tetrachlorohomopurine with specific amines to form the initial chlorinated intermediates, followed by selective reduction using borohydrides to establish the correct oxidation state of the ring system. Subsequent steps involve the introduction of protecting groups and nucleophilic substitution with cyclic amines to construct the core pyrimido-pyrimidine scaffold with high regioselectivity. The final stages execute deprotection, catalytic hydrogenation or hydriodic acid reduction, and controlled oxidation to yield the target diamino compound with pharmaceutical-grade purity. Detailed standardized synthesis steps are provided in the guide below to ensure reproducibility and safety during scale-up operations.
- React tetrachlorohomopurine with specific amines to form chlorinated intermediates, followed by selective reduction using borohydrides.
- Introduce protecting groups and perform nucleophilic substitution with cyclic amines to construct the core pyrimido-pyrimidine scaffold.
- Execute final deprotection, catalytic hydrogenation or hydriodic acid reduction, and controlled oxidation to yield the target diamino compound.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of the technology described in CN1500091A offers substantial strategic advantages that extend beyond mere chemical efficiency. The primary benefit lies in the significant reduction of manufacturing costs driven by the elimination of expensive and hazardous reagents. By avoiding the use of sulfur-containing starting materials that generate toxic waste, manufacturers can drastically lower their expenditure on waste disposal and environmental compliance measures. Furthermore, the ability to bypass chromatographic purification for intermediates translates into reduced solvent usage and shorter production cycles, directly impacting the bottom line through improved asset utilization. This process intensification allows for higher throughput in existing facilities without the need for major capital investment in new equipment, making it an attractive option for optimizing current production capabilities. The use of readily available starting materials like tetrachlorohomopurine also mitigates supply chain risks, ensuring a steady flow of raw materials even in volatile market conditions.
- Cost Reduction in Manufacturing: The elimination of toxic thiol byproducts and the removal of chromatographic purification steps lead to substantial cost savings in both material and operational expenses. By utilizing cheaper, readily available chloro-precursors instead of complex sulfinyl compounds, the raw material cost base is significantly lowered. Additionally, the reduced need for specialized waste treatment infrastructure for sulfur compounds further decreases the overall cost of goods sold, enhancing profit margins for the final pharmaceutical product.
- Enhanced Supply Chain Reliability: The reliance on stable, commercially available starting materials ensures a more robust and predictable supply chain. Unlike specialized sulfur-containing intermediates that may have limited suppliers, tetrachlorohomopurine is a commodity chemical with a broad supply base, reducing the risk of shortages. This stability allows for better long-term planning and inventory management, ensuring that production schedules are met consistently without unexpected delays caused by raw material procurement issues.
- Scalability and Environmental Compliance: The process is inherently designed for industrial scale-up, with reaction conditions that are easily manageable in large reactors. The absence of hazardous sulfur waste simplifies environmental compliance, making it easier to obtain necessary permits and maintain operational licenses. This scalability ensures that the manufacturing process can grow alongside market demand, providing a sustainable solution for the long-term production of high-purity pharmaceutical intermediates without compromising on safety or regulatory standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. These answers are derived directly from the technical specifications and beneficial effects outlined in the patent documentation, providing clarity on process capabilities and limitations. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their specific manufacturing needs. The responses highlight the key differentiators of this method compared to conventional approaches, focusing on purity, safety, and scalability.
Q: How does this patent method avoid toxic byproducts compared to prior art?
A: Unlike previous methods utilizing methylsulfinyl or methylsulfonyl groups that release toxic thiols, this process uses chloro-substituted intermediates and controlled oxidation, eliminating foul-smelling and hazardous sulfur waste streams.
Q: Is chromatographic purification required for the intermediates?
A: No, the patent explicitly states that intermediates possess high purity and can be processed further without chromatographic purification, significantly reducing production time and solvent consumption.
Q: What are the scalability advantages of this synthetic route?
A: The method utilizes readily available starting materials like tetrachlorohomopurine and operates under conditions suitable for industrial scale-up, ensuring consistent supply and cost efficiency for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 4,6-Diaminopyrimido[5,4-d]pyrimidine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic routes in the development of next-generation pharmaceuticals. Our team of experts possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that complex chemistries like the one described in CN1500091A are translated efficiently from the lab to the plant. We are committed to delivering high-purity intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. Our CDMO services are designed to provide end-to-end support, from process optimization to regulatory filing assistance, helping you bring your products to market faster and more cost-effectively.
We invite you to collaborate with us to explore how this advanced synthesis technology can enhance your supply chain and reduce your manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific production volumes. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our expertise can drive value for your organization. Let us be your partner in navigating the complexities of fine chemical manufacturing and achieving your commercial goals.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
