Advanced Synthesis of Selective Dopamine D4 Antagonists for Neuropsychiatric Applications
Advanced Synthesis of Selective Dopamine D4 Antagonists for Neuropsychiatric Applications
The pharmaceutical landscape for treating neuropsychiatric disorders is undergoing a significant paradigm shift, driven by the need for agents that offer efficacy without the debilitating side effects of first-generation neuroleptics. Patent CN1293669A introduces a novel class of substituted 2-(4-benzyl-piperazin-1-yl) and 2-(1-benzyl-piperidin-4-yl) ethanones, specifically designed as selective dopamine D4 receptor antagonists. This technology represents a critical advancement for R&D teams focusing on central nervous system (CNS) therapeutics, particularly for indications such as schizophrenia, depression, and cognitive deficits. By targeting the D4 receptor subtype, which is localized in the limbic brain region, these compounds aim to modulate mood and recognition functions while sparing the striatal D2 receptors responsible for motor control. For procurement and supply chain leaders, understanding the chemical architecture and synthetic accessibility of these molecules is paramount for securing a reliable pipeline of high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional antipsychotic medications, often referred to as typical neuroleptics, primarily function through the blockade of dopamine D2 receptors. While effective in managing positive symptoms of psychosis, this mechanism frequently results in undesirable extrapyramidal side effects (EPS) and tardive dyskinesia due to D2 receptor occupancy in the striatum. Furthermore, conventional synthesis routes for related heterocyclic compounds often suffer from poor regioselectivity and require harsh reaction conditions that complicate purification. The lack of receptor subtype selectivity in older generations of drugs limits their therapeutic window, forcing clinicians to balance efficacy against severe neurological toxicity. From a manufacturing perspective, the production of non-selective analogs often involves complex protection-deprotection sequences and generates significant chemical waste, driving up the cost of goods sold (COGS) and creating supply chain bottlenecks for active pharmaceutical ingredients.
The Novel Approach
The invention disclosed in CN1293669A overcomes these hurdles by introducing a specific structural motif that confers high affinity and selectivity for the D4 receptor subtype. The core innovation lies in the ethanone linker connecting a substituted cyclic amine (such as indoline or tetrahydroquinoline) to a benzylpiperazine or benzylpiperidine moiety. 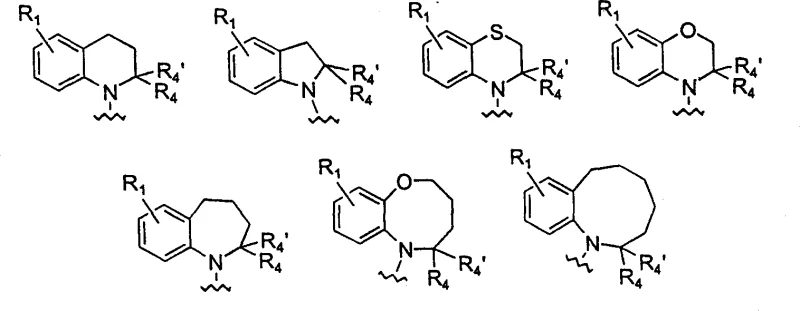 As illustrated in the structural variations, the flexibility to modify substituents at R1 through R11 allows for fine-tuning of pharmacokinetic properties and receptor binding profiles. This modular approach enables chemists to optimize the molecule for oral bioavailability and metabolic stability without compromising potency. The novel synthetic strategies described avoid the use of expensive transition metal catalysts, relying instead on robust nucleophilic substitutions and acylation reactions that are well-suited for industrial scale-up, thereby offering a distinct advantage in cost reduction in pharmaceutical manufacturing.
As illustrated in the structural variations, the flexibility to modify substituents at R1 through R11 allows for fine-tuning of pharmacokinetic properties and receptor binding profiles. This modular approach enables chemists to optimize the molecule for oral bioavailability and metabolic stability without compromising potency. The novel synthetic strategies described avoid the use of expensive transition metal catalysts, relying instead on robust nucleophilic substitutions and acylation reactions that are well-suited for industrial scale-up, thereby offering a distinct advantage in cost reduction in pharmaceutical manufacturing.
Mechanistic Insights into Selective D4 Receptor Binding
The biological efficacy of these benzylpiperazinyl ethanone derivatives is rooted in their precise molecular interaction with the dopamine D4 receptor. The spatial arrangement of the lipophilic aromatic rings and the basic nitrogen atoms within the piperazine or piperidine ring facilitates strong hydrogen bonding and hydrophobic interactions within the receptor binding pocket. Unlike D2 antagonists, these compounds exploit unique amino acid residues present in the D4 subtype, resulting in binding constants (Ki) typically ranging from 0.1 nM to 500 nM. The patent data highlights that preferred embodiments exhibit at least a 10-fold, and often greater than 100-fold, selectivity for D4 over D2 receptors. This selectivity is crucial for minimizing motor side effects, as it ensures that the therapeutic modulation of limbic pathways occurs without disrupting nigrostriatal dopamine transmission. For R&D directors, this mechanistic specificity validates the compound class as a viable candidate for next-generation antipsychotics with an improved safety profile.
From a synthetic chemistry perspective, the formation of the key ethanone linkage is achieved through efficient acylation protocols. 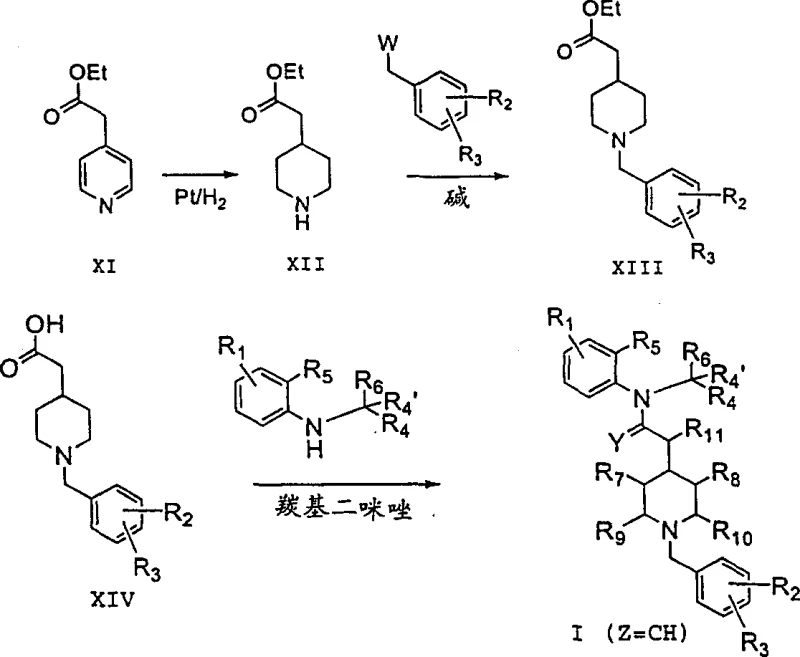 The reaction scheme demonstrates the conversion of pyridine-4-acetic acid esters into piperidine derivatives, followed by N-benzylation and subsequent amidation. This pathway avoids the generation of hazardous byproducts and utilizes mild bases such as sodium carbonate or potassium carbonate. The control of impurities is managed through careful selection of leaving groups (e.g., chloro or sulfonate esters) and optimization of reaction temperatures, typically maintaining conditions between room temperature and reflux. The ability to isolate intermediates as stable salts, such as hydrobromides or hydrochlorides, further enhances the purity profile of the final API. This rigorous control over the synthetic trajectory ensures that the impurity spectrum remains within acceptable limits for clinical development, addressing a primary concern for regulatory compliance.
The reaction scheme demonstrates the conversion of pyridine-4-acetic acid esters into piperidine derivatives, followed by N-benzylation and subsequent amidation. This pathway avoids the generation of hazardous byproducts and utilizes mild bases such as sodium carbonate or potassium carbonate. The control of impurities is managed through careful selection of leaving groups (e.g., chloro or sulfonate esters) and optimization of reaction temperatures, typically maintaining conditions between room temperature and reflux. The ability to isolate intermediates as stable salts, such as hydrobromides or hydrochlorides, further enhances the purity profile of the final API. This rigorous control over the synthetic trajectory ensures that the impurity spectrum remains within acceptable limits for clinical development, addressing a primary concern for regulatory compliance.
How to Synthesize Benzylpiperazinyl Ethanone Efficiently
The preparation of these high-value intermediates follows a logical sequence of organic transformations that prioritize yield and operational simplicity. The process generally begins with the activation of the cyclic amine component, followed by coupling with the piperazine fragment. Detailed standard operating procedures for the acylation, alkylation, and salt formation steps are critical for ensuring batch-to-batch consistency. The following guide outlines the standardized synthesis protocol derived from the patent examples, providing a roadmap for process chemists to replicate the results in a pilot plant environment.
- Acylation of the cyclic amine (e.g., indoline or tetrahydroquinoline) with chloroacetyl chloride to form the alpha-chloro ketone intermediate.
- Nucleophilic substitution reaction between the alpha-chloro ketone and a substituted benzylpiperazine derivative in the presence of a base like potassium carbonate.
- Purification of the crude product followed by conversion to a pharmaceutically acceptable acid addition salt, such as the hydrobromide or hydrochloride salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits regarding cost efficiency and supply security. The reliance on commodity chemicals such as indoline, chloroacetyl chloride, and substituted benzyl chlorides means that raw material sourcing is not dependent on exotic or single-source suppliers. This diversification of the supply base significantly mitigates the risk of production stoppages due to raw material shortages. Furthermore, the elimination of precious metal catalysts from the synthesis removes the need for expensive scavenging steps and heavy metal testing, directly contributing to cost reduction in pharmaceutical manufacturing. The streamlined workflow reduces the overall number of unit operations, which translates to lower energy consumption and reduced solvent usage, aligning with modern green chemistry initiatives and environmental compliance standards.
- Cost Reduction in Manufacturing: The synthetic pathway described eliminates the need for costly transition metal catalysts and complex chromatographic purifications, relying instead on crystallization and extraction techniques that are economically favorable at scale. By utilizing widely available starting materials and avoiding specialized reagents, the overall cost of goods is significantly optimized. The process design minimizes waste generation and solvent recovery costs, allowing for a more competitive pricing structure for the final intermediate. This economic efficiency is crucial for maintaining margins in the highly competitive CNS drug market.
- Enhanced Supply Chain Reliability: The robustness of the chemical reactions ensures high reproducibility, which is essential for maintaining consistent supply volumes. The intermediates formed are chemically stable and can be stored for extended periods without degradation, providing a buffer against demand fluctuations. The use of standard equipment and common solvents means that production can be easily transferred between different manufacturing sites if necessary, enhancing the resilience of the supply network. This flexibility ensures that downstream API production schedules are met without delay.
- Scalability and Environmental Compliance: The reaction conditions are mild and do not require extreme pressures or temperatures, making the process inherently safer and easier to scale from kilogram to multi-ton quantities. The waste streams generated are primarily aqueous salts and organic solvents that can be treated using conventional wastewater treatment facilities. This simplifies the permitting process and reduces the environmental footprint of the manufacturing operation. The ability to scale up rapidly without re-engineering the process supports fast-track development timelines for new drug candidates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these dopamine D4 antagonists. The answers are derived directly from the technical specifications and experimental data provided in the patent documentation. Understanding these details helps stakeholders make informed decisions regarding project feasibility and resource allocation.
Q: What is the primary therapeutic advantage of these benzylpiperazinyl ethanone derivatives?
A: These compounds exhibit high selectivity for dopamine D4 receptors over D2 receptors, potentially reducing extrapyramidal side effects (EPS) commonly associated with traditional antipsychotics.
Q: Are these intermediates suitable for large-scale commercial production?
A: Yes, the synthetic routes described utilize commercially available starting materials like indoline and substituted piperazines, employing standard unit operations such as reflux and crystallization that are easily scalable.
Q: How is the stereochemical purity managed in this synthesis?
A: While racemic mixtures are common, the patent describes methods for resolution using chiral HPLC or crystallization with resolving agents to obtain optically active single enantiomers for enhanced efficacy.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzylpiperazinyl Ethanone Supplier
As the demand for targeted CNS therapeutics grows, partnering with an experienced CDMO is essential for translating laboratory innovations into commercial reality. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from clinical trials to market launch. Our facility is equipped with stringent purity specifications and rigorous QC labs capable of analyzing complex impurity profiles, guaranteeing that every batch of benzylpiperazinyl ethanone derivative meets the highest international standards. We understand the critical nature of supply continuity in the pharmaceutical industry and have established robust logistics networks to deliver materials on time, every time.
We invite you to collaborate with our technical team to explore how this patented technology can be integrated into your drug development pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate our capability to support your long-term strategic goals. Let us be your partner in advancing the next generation of neuropsychiatric treatments.
