Advanced Manufacturing of Antidiabetic Intermediates via Improved Reduction and Cyclization
Advanced Manufacturing of Antidiabetic Intermediates via Improved Reduction and Cyclization
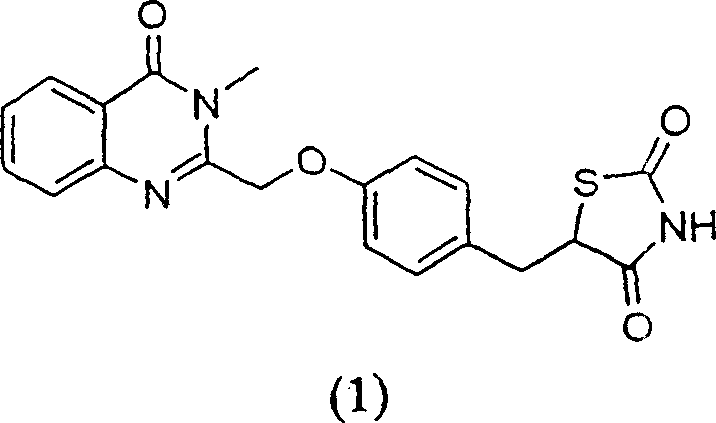
The pharmaceutical industry constantly seeks robust, scalable, and economically viable synthetic routes for high-value active pharmaceutical ingredients (APIs) and their key intermediates. Patent CN1231481C presents a groundbreaking advancement in the synthesis of 5-[4-[[3-methyl-4-oxo-3,4-dihydroquinolin-2-yl]methoxy]benzyl]thiazolidine-2,4-dione, a critical scaffold in the development of antidiabetic therapeutics targeting Type 2 diabetes (NIDDM). This intellectual property addresses the severe limitations of previous methodologies, specifically the reliance on prohibitively expensive stoichiometric catalysts and cumbersome multi-step activation protocols. By transitioning from palladium-based reduction systems to more accessible alternatives like Raney Nickel or Magnesium, this technology offers a pathway to substantial operational efficiency. For R&D directors and procurement specialists, understanding the mechanistic shifts in this patent is essential for evaluating supply chain resilience and long-term cost structures in the production of thiazolidine-2,4-dione derivatives.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art methodologies, such as those described in international publication WO 97/41097, rely heavily on the use of Palladium on Carbon (Pd/C) as a stoichiometric catalyst for the reduction of unsaturated precursors. This approach presents a massive economic bottleneck, as the cost of the palladium catalyst alone can constitute nearly 70% of the total production cost for the intermediate. Furthermore, the reaction kinetics are sluggish, often requiring approximately 40 hours to reach completion, which severely limits throughput in a commercial manufacturing setting. Beyond the catalyst cost, the downstream processing is exceptionally laborious; the hydrolysis of the ethyl ester involves a complex sequence of methanol removal, aqueous dilution, organic extraction, and precise pH adjustments to precipitate the acid, typically yielding only about 80%. The subsequent activation of this acid requires hazardous reagents like pivaloyl chloride or thionyl chloride to form mixed anhydrides or acid chlorides, introducing significant safety risks and moisture sensitivity issues that complicate scale-up operations.
The Novel Approach
The improved process detailed in CN1231481C fundamentally re-engineers the synthetic strategy to eliminate these bottlenecks. Instead of relying on precious metals, the new method employs Raney Nickel under hydrogen pressure or Magnesium in alcoholic solvents to effect the reduction of the olefinic bond. This substitution not only slashes raw material costs but also simplifies the reaction environment, allowing for completion within significantly shorter timeframes, often between 12 to 24 hours depending on the specific conditions. Crucially, the novel approach bypasses the traditional acid activation step entirely. The carboxylic acid intermediate is condensed directly with N-methyl anthranilamide without pre-conversion to an acid chloride or anhydride. This direct thermal cyclization streamlines the workflow, reduces the number of unit operations, and minimizes the generation of chemical waste, thereby offering a reliable pharmaceutical intermediate supplier with a distinct competitive advantage in terms of both environmental compliance and manufacturing economics.
Mechanistic Insights into Raney Nickel Reduction and Direct Cyclization
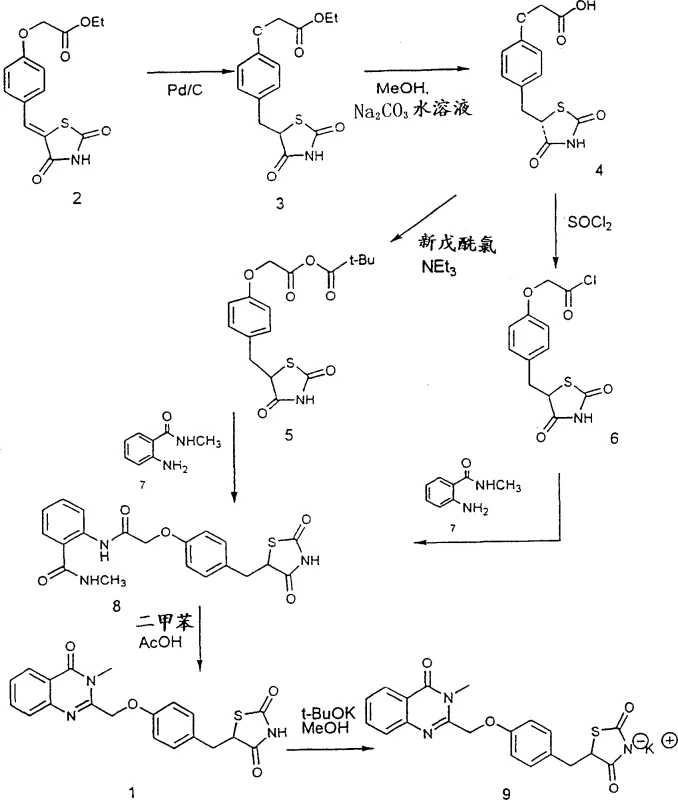
The core innovation lies in the heterogeneous catalytic reduction using Raney Nickel, which facilitates the hydrogenation of the exocyclic double bond in the thiazolidine-2,4-dione ring system. Unlike homogeneous catalysts or stoichiometric hydride donors, Raney Nickel provides a high surface area for hydrogen adsorption, enabling efficient transfer of hydrogen atoms to the substrate under moderate pressures (e.g., 400 psi). The mechanism involves the adsorption of the alkene onto the nickel surface, followed by sequential hydrogen addition to form the saturated benzyl-thiazolidine structure. Alternatively, the use of Magnesium in methanol proceeds via a single-electron transfer mechanism, where magnesium acts as a reducing agent in the alcoholic medium, simultaneously facilitating reduction and potential transesterification. This dual functionality allows for flexible process design, where the ester can be reduced and subsequently hydrolyzed in a telescoped manner, significantly reducing solvent consumption and processing time compared to the stepwise isolation required in older protocols.
Furthermore, the direct condensation and cyclization mechanism represents a significant departure from classical amide coupling strategies. In the absence of activating agents like thionyl chloride, the reaction relies on thermal energy in high-boiling solvents such as xylene or toluene, often aided by acid catalysts like p-toluenesulfonic acid. The mechanism likely proceeds through an initial amidation at elevated temperatures, followed by an intramolecular nucleophilic attack of the amide nitrogen onto the carbonyl carbon of the thiazolidine ring, or vice versa depending on the specific tautomeric forms present. This thermal cyclization avoids the formation of reactive acid chloride intermediates, which are prone to hydrolysis and side reactions in the presence of trace moisture. By maintaining a strictly anhydrous environment during the initial heating phase and utilizing a Dean-Stark apparatus to remove water azeotropically, the process drives the equilibrium towards the desired quinolinone product, achieving purities exceeding 98% without the need for extensive chromatographic purification.
How to Synthesize 5-[4-[[3-methyl-4-oxo-3,4-dihydroquinolin-2-yl]methoxy]benzyl]thiazolidine-2,4-dione Efficiently
Implementing this improved synthesis requires careful control of reaction parameters to maximize the benefits of the new catalytic systems. The process begins with the preparation of the unsaturated precursor, followed by the critical reduction step using either Raney Nickel or Magnesium. Operators must ensure that the catalyst is properly washed and activated before introduction to the reactor to prevent poisoning. Following reduction, the hydrolysis step is simplified to a basic aqueous treatment, avoiding the complex extraction sequences of the past. The final cyclization demands precise temperature control, typically refluxing in aromatic solvents for 10 to 15 hours to ensure complete conversion. For detailed operational parameters, stoichiometry, and specific workup instructions tailored to your facility's capabilities, please refer to the standardized protocol below.
- Reduce the unsaturated precursor using Raney Nickel under hydrogen pressure or Magnesium in alcohol, avoiding expensive stoichiometric Pd/C catalysts.
- Hydrolyze the resulting ester to the corresponding carboxylic acid using standard alkaline conditions with simplified workup procedures.
- Condense the acid directly with N-methyl anthranilamide and perform thermal cyclization in high-boiling solvents to form the final quinolinone-thiazolidine scaffold.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to the methodology described in CN1231481C offers profound strategic benefits beyond simple yield improvements. The most immediate impact is the drastic reduction in raw material expenditure associated with eliminating stoichiometric palladium catalysts. In large-scale API manufacturing, the cost of noble metals can fluctuate wildly, creating budgetary uncertainty; replacing Pd/C with Raney Nickel or Magnesium stabilizes the cost base and insulates the supply chain from precious metal market volatility. Additionally, the removal of corrosive activating agents like thionyl chloride reduces the regulatory burden associated with handling hazardous chemicals, lowering compliance costs and improving workplace safety profiles. The simplification of the workup procedure, particularly the elimination of multiple solvent swaps and pH adjustments, translates directly into reduced utility consumption and shorter batch cycle times, enhancing overall plant capacity without the need for capital investment in new equipment.
- Cost Reduction in Manufacturing: The elimination of expensive stoichiometric Pd/C catalysts removes a major cost driver that previously accounted for a dominant portion of the bill of materials. By substituting this with cost-effective Raney Nickel or Magnesium, manufacturers can achieve substantial cost savings in API manufacturing. Furthermore, the avoidance of specialized activating reagents like pivaloyl chloride reduces the complexity of the supply chain, as fewer specialty chemicals need to be sourced, stored, and managed, leading to a leaner and more economical production model.
- Enhanced Supply Chain Reliability: Reliance on palladium introduces supply chain fragility due to the geopolitical concentration of platinum group metal mining. Switching to base metals like nickel or magnesium, which are abundant and widely available, ensures a more robust and continuous supply of critical reagents. The simplified process also reduces the lead time for high-purity pharmaceutical intermediates by shortening the overall reaction and purification timeline, allowing for faster response to market demand fluctuations and more agile inventory management strategies.
- Scalability and Environmental Compliance: The new process significantly reduces the generation of hazardous waste streams associated with heavy metal residues and chlorinated byproducts. This aligns with increasingly stringent environmental regulations and corporate sustainability goals, making the commercial scale-up of complex pharmaceutical intermediates more feasible. The use of simpler solvent systems and the ability to telescope steps reduce the total volume of waste solvent requiring treatment, thereby lowering disposal costs and minimizing the environmental footprint of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved synthesis route. These insights are derived directly from the comparative data and experimental examples provided in the patent documentation, highlighting the practical advantages over legacy methods. Understanding these nuances is vital for technical teams evaluating the feasibility of technology transfer.
Q: Why is replacing Pd/C with Raney Nickel critical for cost reduction?
A: Conventional methods utilize stoichiometric amounts of Palladium on Carbon, which accounts for nearly 70% of the product cost in older processes. Switching to Raney Nickel or Magnesium drastically reduces raw material expenses and eliminates the need for complex heavy metal removal steps.
Q: How does the new process improve safety regarding acid activation?
A: The improved method eliminates the need for converting the carboxylic acid into mixed anhydrides or acid chlorides using corrosive reagents like pivaloyl chloride or thionyl chloride. Direct condensation simplifies the workflow and removes moisture-sensitive hazardous steps.
Q: What are the yield improvements in the cyclization step?
A: While prior art cyclization yields were approximately 50% requiring 20-30 hours of reflux, the optimized thermal cyclization described in the patent achieves significantly higher purity and yield profiles by streamlining the precursor synthesis and reducing side reactions.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 5-[4-[[3-methyl-4-oxo-3,4-dihydroquinolin-2-yl]methoxy]benzyl]thiazolidine-2,4-dione Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of antidiabetic therapies depends on the availability of high-quality, cost-effective intermediates. Our technical team has extensively analyzed the process innovations in CN1231481C and possesses the expertise to implement these improved routes at an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this patented technology are realized in every batch we produce. Our facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that the thiazolidine-2,4-dione derivatives we supply meet the exacting standards required for global pharmaceutical registration.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis can benefit your specific project timelines and budget constraints. By leveraging our manufacturing capabilities, you can access a Customized Cost-Saving Analysis that quantifies the economic impact of switching to this improved method. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of technical excellence and commercial reliability.
