Advanced Synthesis of 2-(trans-4-n-propylcyclohexyl) Propane-1,3-diol for High-Performance Liquid Crystal Manufacturing
The rapidly evolving landscape of the electronic chemical sector demands intermediates that not only meet stringent performance metrics but also adhere to rigorous safety and environmental standards. Patent CN113233958A, published in August 2021, introduces a groundbreaking preparation method for 2-(trans-4-n-propylcyclohexyl) propane-1, 3-diol, a critical organic material proven to be suitable for the production of advanced liquid crystal mixtures. This specific diol is essential for formulating liquid crystals that exhibit high resistivity, low power consumption, and low driving threshold voltage, which are paramount for next-generation display technologies. The disclosed technology represents a significant paradigm shift from traditional synthesis routes that often rely on hazardous reagents and extreme conditions. By leveraging a sequential process of bromination, alkylation, and reduction, this method ensures a safer, low-cost, and environmentally friendly pathway that is inherently suitable for large-scale industrial production. For R&D Directors and Supply Chain Heads, understanding the nuances of this patent is crucial for securing a reliable liquid crystal intermediate supplier capable of delivering consistent quality without the baggage of legacy safety risks.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 1, 3-propanediol derivatives like the target compound has been plagued by significant safety and efficiency hurdles that impact both operational costs and supply chain stability. Prior art, such as the process disclosed in patent document CN103524305B, relies heavily on the use of Titanium Tetrachloride (TiCl4) as a catalyst during the coupling reaction. This reagent is notoriously unstable; it generates decomposition reactions and releases intense heat when exposed to moisture or during heating processes, emitting toxic and corrosive smoke that requires specialized containment and scrubbing systems. Furthermore, the subsequent hydrogenation reaction in these conventional routes introduces a substantial risk of physical and chemical explosion due to the high pressures and temperatures involved. These factors not only inflate the capital expenditure for safety infrastructure but also create potential bottlenecks in production continuity, making cost reduction in electronic chemical manufacturing difficult to achieve when relying on such volatile chemistries.
The Novel Approach
In stark contrast, the novel approach detailed in CN113233958A circumvents these dangers by employing a milder, three-step sequence that prioritizes operational safety and chemical efficiency. The process initiates with a bromination reaction using phosphorus tribromide, followed by a highly selective alkylation using a lithium methoxide/ethanol system, and concludes with a reduction using potassium borohydride and lithium chloride. This strategic selection of reagents eliminates the need for hazardous Titanium Tetrachloride and high-pressure hydrogenation, thereby drastically simplifying the reactor requirements and waste treatment protocols. The alkylation step, in particular, utilizes a specific solvent and base combination that minimizes side reactions, ensuring that the commercial scale-up of complex organic intermediates can proceed with higher confidence and lower risk. This transition from hazardous to benign chemistry is not merely a regulatory compliance measure but a strategic advantage for any procurement manager seeking to stabilize their supply chain against safety-related shutdowns.
Mechanistic Insights into Lithium Methoxide-Catalyzed Alkylation and Reduction
The core technical breakthrough of this patent lies in the optimization of the alkylation and reduction steps, which are critical for controlling the impurity profile and stereochemistry of the final product. In the alkylation phase, the reaction between 1-bromo-4-n-propylcyclohexane and diethyl malonate is facilitated by lithium methoxide in an ethanol solution. Comparative data within the patent reveals that this specific system outperforms sodium-based or methanol-based alternatives, which tend to generate higher levels of alkene impurities due to excessive basicity or poor solubility dynamics. The lithium cation appears to offer a balanced basicity that promotes the formation of the necessary enolate without triggering excessive dehydrobromination of the alkyl halide. Following this, the reduction step employs a synergistic system of potassium borohydride and lithium chloride. This combination allows for a controlled reduction of the ester groups to hydroxyl groups while maintaining the integrity of the cyclohexane ring structure.
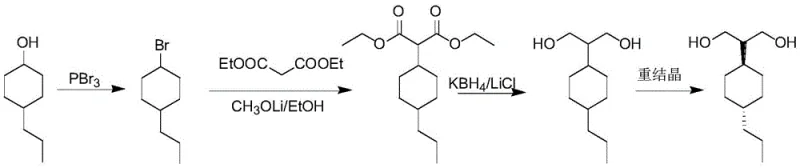
Furthermore, the patent specifies a precise temperature profile for the reduction reaction to maximize yield and stereoselectivity. The process involves an initial low-temperature phase (30-35°C) followed by a higher-temperature phase (42-48°C). This two-stage thermal strategy is essential for managing the reaction kinetics; the initial lower temperature controls the exotherm and prevents rapid, uncontrolled reduction that could lead to byproduct formation, while the subsequent higher temperature ensures the complete conversion of the intermediate ester to the desired diol. The final recrystallization from n-heptane is the key to achieving the high-purity liquid crystal material required for display applications, effectively separating the trans-isomer from the cis-impurity. This level of mechanistic control ensures that the final product meets the rigorous purity specifications demanded by high-end electronic material manufacturers, with the patent examples demonstrating purity levels of ≥99.8% and cis-impurity levels of <0.5%.
How to Synthesize 2-(trans-4-n-propylcyclohexyl) Propane-1,3-diol Efficiently
Implementing this synthesis route requires strict adherence to the inert atmosphere conditions and reagent ratios specified in the patent to replicate the high yields and purity profiles observed in the examples. The process begins with the careful addition of phosphorus tribromide to 4-n-propyl cyclohexanol, followed by the sequential addition of diethyl malonate and lithium methoxide under controlled thermal conditions. The reduction step demands precise batch addition of potassium borohydride to manage gas evolution and heat generation. While the general workflow is outlined here, the specific operational parameters, such as stirring rates, addition speeds, and exact quenching protocols, are critical for success. For a detailed breakdown of the standardized synthesis steps, including specific mass ratios and temperature ramping rates, please refer to the technical guide below.
- Perform bromination of 4-n-propyl cyclohexanol with phosphorus tribromide at 80-90°C to generate 1-bromo-4-n-propyl cyclohexane.
- Conduct alkylation using diethyl malonate and lithium methoxide in ethanol at 80-85°C to form the diethyl malonate intermediate.
- Execute reduction with potassium borohydride and lithium chloride, followed by recrystallization in n-heptane to obtain the final trans-diol product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible benefits that extend beyond mere chemical yield, directly impacting the total cost of ownership and supply reliability. The elimination of Titanium Tetrachloride and high-pressure hydrogenation equipment significantly reduces the capital and operational expenditures associated with safety compliance and hazard management. By shifting to reagents like phosphorus tribromide and potassium borohydride, the process utilizes materials that are more readily available and easier to handle, thereby reducing lead time for high-purity diols and minimizing the risk of supply disruptions caused by regulatory restrictions on hazardous chemicals. This transition supports a more resilient supply chain capable of withstanding market fluctuations and regulatory changes.
- Cost Reduction in Manufacturing: The streamlined process eliminates the need for expensive transition metal catalysts and the complex downstream processing required to remove heavy metal residues, which traditionally adds significant cost to the manufacturing bill of materials. Additionally, the higher selectivity of the lithium methoxide system reduces the volume of waste solvents and byproducts that require disposal, leading to substantial cost savings in waste treatment and environmental compliance. The improved yield profile further contributes to economic efficiency by maximizing the output per batch of raw materials, ensuring that the cost reduction in electronic chemical manufacturing is realized through both input optimization and output maximization.
- Enhanced Supply Chain Reliability: The reliance on common, stable reagents rather than specialized, hazardous catalysts enhances the robustness of the supply chain against external shocks. Since the raw materials such as diethyl malonate and potassium borohydride are commodity chemicals with multiple global sources, the risk of single-source bottlenecks is significantly mitigated. This availability ensures that production schedules can be maintained consistently, providing a reliable liquid crystal intermediate supplier partner who can meet demand spikes without the delays often associated with sourcing restricted or dangerous precursors.
- Scalability and Environmental Compliance: The mild reaction conditions and the absence of high-pressure steps make this process inherently easier to scale from pilot plant to commercial production volumes. The reduced generation of toxic corrosive smoke and the elimination of explosion risks simplify the environmental permitting process and lower the insurance premiums associated with chemical manufacturing. This environmental friendliness aligns with the increasing global demand for green chemistry solutions, positioning the product favorably in markets with strict environmental regulations and ensuring long-term viability for the commercial scale-up of complex organic intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of 2-(trans-4-n-propylcyclohexyl) propane-1, 3-diol based on the disclosed patent technology. These insights are derived directly from the experimental data and comparative examples provided in the intellectual property documentation, offering clarity on yield, purity, and process safety. Understanding these details is vital for technical teams evaluating the feasibility of integrating this intermediate into their own liquid crystal formulations or supply chains.
Q: Why is the Lithium Methoxide/Ethanol system preferred for this alkylation?
A: The patent data indicates that the Lithium Methoxide/Ethanol system significantly reduces side reaction impurities compared to sodium-based or methanol-based systems, leading to higher yields and easier purification.
Q: How does this process improve safety over conventional methods?
A: Unlike conventional routes using Titanium Tetrachloride which release toxic corrosive smoke, or high-pressure hydrogenation which carries explosion risks, this method utilizes milder reagents like Phosphorus Tribromide and Potassium Borohydride under controlled atmospheric conditions.
Q: What purity levels can be achieved with this recrystallization method?
A: Through recrystallization using n-heptane, the process achieves a purity of greater than or equal to 99.8%, with cis-form single impurity content controlled to less than 0.5%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-(trans-4-n-propylcyclohexyl) Propane-1,3-diol Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-performance intermediates play in the advancement of display technologies and electronic materials. As a leading CDMO expert, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory patent to industrial reality is seamless and efficient. Our facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of 2-(trans-4-n-propylcyclohexyl) propane-1, 3-diol meets the exacting standards required for high-resistivity liquid crystal mixtures. We are committed to delivering not just a chemical product, but a comprehensive solution that supports your R&D and manufacturing goals.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific application needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this safer, more efficient route can optimize your budget. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate why we are the preferred partner for high-purity liquid crystal material sourcing in the global market.
