Advanced Synthesis of Selective Sigma Receptor Ligands for Commercial Pharmaceutical Applications
Advanced Synthesis of Selective Sigma Receptor Ligands for Commercial Pharmaceutical Applications
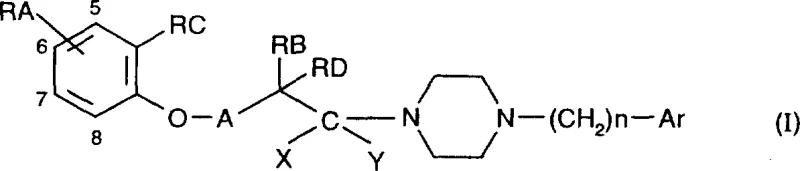
The pharmaceutical landscape is constantly evolving towards higher specificity and reduced toxicity, a trend exemplified by the innovations disclosed in patent CN1132202A. This pivotal document introduces a novel class of benzopyran derivatives containing a piperazine structure, specifically engineered to act as highly selective ligands for sigma receptors. Unlike earlier generations of central nervous system (CNS) agents that suffered from broad-spectrum receptor binding and severe side effects, these new compounds offer a refined pharmacological profile. The core innovation lies in the aralkylpiperazinylalkylbenzopyran scaffold, which decouples therapeutic efficacy from the sedative and extrapyramidal liabilities common in prior art. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, understanding the structural nuances and synthetic accessibility of these molecules is crucial for developing next-generation therapeutics targeting neurodegenerative and immune disorders.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, compounds such as aminomethylchromans and certain phenylpiperazine derivatives were explored for their CNS activity, but they presented significant clinical hurdles. Literature from the early 1980s indicates that while these molecules possessed affinity for 5-hydroxytryptamine (serotonin) and dopamine receptors, this lack of selectivity resulted in profound adverse effects. Patients treated with these non-specific agents frequently experienced severe sedation, drowsiness, and more dangerous acute reactions such as tongue spasms and oculogyric crises. These side effects, stemming from off-target receptor binding, severely limited the therapeutic window and patient compliance. Furthermore, the synthesis of some earlier analogues often involved harsh conditions or difficult-to-source precursors, complicating the commercial scale-up of complex pharmaceutical intermediates. The industry urgently required a structural motif that could retain CNS penetration while bypassing the dopaminergic and serotonergic pathways responsible for these debilitating toxicities.
The Novel Approach
The methodology outlined in CN1132202A represents a paradigm shift by introducing an aralkylpiperazinylalkylbenzopyran structure that demonstrates negligible affinity for serotonin and dopamine receptors. Instead, these derivatives exhibit high selectivity for sigma receptors, a distinct pharmacological target associated with memory modulation, motor control, and immune response. This selectivity profile effectively eliminates the sedative and extrapyramidal side effects plaguing previous generations of drugs. From a manufacturing perspective, the novel approach utilizes versatile synthetic routes that allow for extensive structural diversification at the aromatic rings and the piperazine nitrogen. This flexibility enables the fine-tuning of physicochemical properties such as solubility and metabolic stability without compromising the core biological activity. Consequently, this approach not only solves the safety issues of the past but also opens up new avenues for cost reduction in API manufacturing by utilizing robust and scalable chemical transformations.
Mechanistic Insights into Sigma Receptor Selectivity and Synthetic Pathways
The biological efficacy of these benzopyran derivatives is rooted in their precise three-dimensional arrangement, which facilitates optimal binding to the sigma receptor pocket while sterically hindering interaction with dopamine D2 or serotonin 5-HT2 receptors. Pharmacological data from the patent reveals IC50 values in the nanomolar range (e.g., 9.1 x 10^-9 M for Example 1), confirming potent binding affinity. The presence of the piperazine ring linked via a methylene or ethylene bridge to the benzopyran core appears critical for this selectivity. The substituent Ar, which can be a phenyl or naphthyl group, further modulates lipophilicity and receptor fit. Mechanistically, the synthesis relies on the formation of a stable amide bond followed by a rigorous reduction step. This sequence ensures that the stereochemistry and integrity of the sensitive benzopyran ring system are maintained throughout the process. Understanding these mechanistic details is vital for quality control teams to monitor impurity profiles, particularly ensuring the complete reduction of the carbonyl group to the methylene linkage to avoid residual amide contaminants.
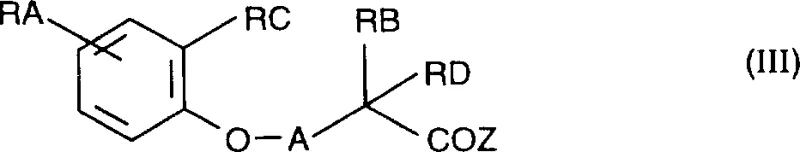
Impurity control is paramount in the production of high-purity pharmaceutical intermediates intended for CNS applications. The synthetic route described involves the reaction of a benzopyran carboxylic acid derivative with a substituted piperazine. A key potential impurity arises from incomplete reduction of the intermediate amide or over-reduction of the aromatic system. However, the patent specifies the use of lithium aluminum hydride under controlled conditions, typically at room temperature or with mild reflux, which provides excellent chemoselectivity. The process allows for the isolation of the free base, which can then be converted into pharmaceutically acceptable salts such as hydrochlorides or tartrates. This salt formation step not only enhances the stability of the final product but also aids in purification through crystallization. By strictly adhering to the stoichiometry and reaction times detailed in the examples, manufacturers can achieve purity levels that meet stringent regulatory specifications, thereby reducing the lead time for high-purity pharmaceutical intermediates entering clinical trials.
How to Synthesize Benzopyran Piperazine Derivatives Efficiently
The synthesis of these valuable intermediates follows a logical two-step sequence that balances yield with operational simplicity, making it highly attractive for industrial application. The process begins with the acylation of a 1-substituted piperazine with a corresponding benzopyran carboxylic acid chloride. This step is typically conducted in a polar aprotic solvent like dichloromethane or tetrahydrofuran, often in the presence of a base to scavenge the generated acid. Following the formation of the amide intermediate, the second critical step involves reduction using a powerful hydride source. As illustrated in the reaction scheme below, this transformation converts the carbonyl functionality into the requisite methylene bridge, completing the aralkylpiperazinylalkylbenzopyran skeleton.
- React a substituted benzopyran carboxylic acid chloride with a 1-substituted piperazine in a solvent like dichloromethane or tetrahydrofuran to form an amide intermediate.
- Reduce the resulting amide intermediate using a strong reducing agent such as lithium aluminum hydride (LiAlH4) in anhydrous tetrahydrofuran.
- Purify the final product via silica gel column chromatography and convert to the desired salt form (e.g., hydrochloride) if necessary for stability.
The detailed standardized synthesis steps for producing these compounds efficiently are provided in the guide below, outlining specific molar ratios, solvent choices, and workup procedures optimized for maximum recovery.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthetic route offers substantial strategic benefits beyond mere chemical novelty. The primary advantage lies in the accessibility and stability of the starting materials. The patent explicitly notes that the requisite acid chlorides and piperazines are either commercially available or can be readily prepared using established literature methods. This reliance on commodity chemicals significantly mitigates supply chain risks associated with exotic or proprietary reagents. Furthermore, the reaction conditions are relatively mild, often proceeding at room temperature or with simple reflux, which reduces energy consumption and equipment wear compared to processes requiring cryogenic temperatures or high-pressure reactors. These factors collectively contribute to a more resilient and cost-effective supply chain for complex pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the use of straightforward reduction chemistry drastically simplify the downstream processing requirements. Traditional methods often necessitate expensive heavy metal scavenging steps to meet residual metal limits, adding both time and cost to the production cycle. In contrast, the lithium aluminum hydride reduction described here generates inorganic salts that are easily removed during the aqueous workup and filtration stages. This streamlined purification process leads to substantial cost savings in waste treatment and solvent recovery. Additionally, the high yields reported in the patent examples, often exceeding 80%, minimize raw material waste, further enhancing the overall economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The modular nature of the synthesis allows for flexible sourcing of raw materials. Since the Ar group and the substituents on the benzopyran ring can be varied without altering the core reaction mechanism, manufacturers can switch between different grades of starting materials based on market availability without revalidating the entire process. This flexibility is crucial for maintaining continuous production schedules in the face of global supply fluctuations. Moreover, the robustness of the reaction against minor variations in temperature or stoichiometry ensures consistent batch-to-batch quality, reducing the risk of production delays caused by out-of-specification results. This reliability is essential for securing long-term contracts with major pharmaceutical partners.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is facilitated by the absence of hazardous gas evolution or highly exothermic events that are difficult to manage in large reactors. The use of common organic solvents like tetrahydrofuran and dichloromethane allows for the utilization of existing infrastructure in most fine chemical plants. From an environmental perspective, the atom economy of the reaction is favorable, and the waste streams are primarily composed of manageable inorganic salts and recoverable organic solvents. This aligns well with modern green chemistry initiatives and regulatory requirements for sustainable manufacturing practices. The ability to produce these intermediates with a lower environmental footprint enhances the corporate social responsibility profile of the supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of these benzopyran derivatives. The answers are derived directly from the experimental data and background information provided in the patent documentation, ensuring accuracy and relevance for technical decision-makers.
Q: What distinguishes these benzopyran derivatives from traditional CNS agents?
A: Unlike traditional aminomethylchromans which exhibit high affinity for serotonin and dopamine receptors causing severe sedation, these novel derivatives selectively target sigma receptors, eliminating extrapyramidal side effects.
Q: Are the starting materials for this synthesis commercially viable?
A: Yes, the patent indicates that starting materials such as substituted benzopyran carboxylic acids and various piperazines are either commercially available or easily prepared via standard literature procedures, ensuring supply chain stability.
Q: What therapeutic areas do these intermediates target?
A: These compounds are designed for treating central nervous system disorders including memory deficits, Alzheimer's disease, motion sickness, and immune-related conditions like arthritis, due to their high sigma receptor affinity.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benzopyran Derivatives Supplier
As the demand for specialized CNS therapeutics grows, partnering with an experienced CDMO is essential for navigating the complexities of process development and scale-up. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facility is equipped with state-of-the-art rigorous QC labs capable of verifying stringent purity specifications, including detailed impurity profiling and chiral analysis where necessary. We understand the critical nature of sigma receptor ligands and apply our deep expertise in heterocyclic chemistry to optimize every step of the synthesis for yield, safety, and cost-efficiency.
We invite you to collaborate with us to leverage these technological advancements for your pipeline. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized processes can accelerate your time-to-market while maintaining the highest standards of quality and compliance.
