Advanced Sertraline Ketimine Synthesis: A Commercial Scale-Up Guide for Pharmaceutical Intermediates
The pharmaceutical industry constantly seeks robust, scalable, and environmentally benign pathways for synthesizing critical antidepressant intermediates. Patent CN1390198A introduces a transformative methodology for the preparation of ketimines, specifically targeting the synthesis of sertraline precursors. This innovation addresses long-standing inefficiencies in the production of sertraline-imine isomers, which are pivotal building blocks for the final active pharmaceutical ingredient. Unlike legacy processes that rely on hazardous Lewis acids and energy-intensive cooling, this novel approach leverages a versatile solvent system and a strategic recrystallization protocol at the imine stage. For R&D directors and procurement specialists, understanding this shift is crucial for optimizing supply chains and reducing the total cost of ownership for high-volume API manufacturing. The technology enables the conversion of sertralone isomer mixtures directly into high-purity imines, bypassing the need for extensive purification at the ketone stage.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of ketimines for sertraline production has been plagued by significant operational and environmental drawbacks, as documented in prior art such as USA-4536518 and USA-4855500. Traditional protocols typically necessitate the use of titanium tetrachloride (TiCl4) as a promoter, a reagent that poses severe ecological hazards and requires stringent handling procedures to prevent hydrolysis and corrosion. Furthermore, these reactions often mandate cryogenic conditions or substantial cooling to control exotherms, driving up energy consumption and capital expenditure for refrigeration infrastructure. The reliance on aprotic solvents like tetrahydrofuran (THF) or dichloromethane introduces additional risks regarding flammability and toxicity, complicating waste management and regulatory compliance. Post-reaction workups are equally burdensome, often requiring the addition of anti-solvents like hexane to precipitate the product, followed by the difficult removal of expensive molecular sieves or metal salts, which can trap valuable product and reduce overall yield.
The Novel Approach
In stark contrast, the methodology disclosed in CN1390198A offers a streamlined alternative that eliminates the need for titanium tetrachloride and molecular sieves entirely. The core innovation lies in reacting a sertralone isomer mixture directly with methylamine in a broad spectrum of commercially available solvents, ranging from simple alcohols like ethanol to nitriles and esters. This flexibility allows manufacturers to select solvents based on cost and recovery efficiency rather than strict chemical necessity. Crucially, the process shifts the purification burden to the imine stage; instead of laboriously recrystallizing the starting ketone to high purity, the invention demonstrates that recrystallizing the crude imine mixture yields superior results. This strategic pivot simplifies the workflow, reduces solvent usage, and enables the direct isolation of the desired 3,4-dichloro isomer with exceptional purity, effectively turning a multi-step purification challenge into a single, efficient crystallization event.
Mechanistic Insights into Acid-Catalyzed Imine Formation and Recrystallization
The chemical elegance of this process is rooted in the controlled formation of the imine bond under mild acidic catalysis, followed by a thermodynamically driven purification. The reaction initiates with the nucleophilic attack of methylamine on the carbonyl carbon of the sertralone derivative. While the reaction can proceed without a catalyst at elevated temperatures, the patent highlights the efficacy of protonic acids such as methanesulfonic acid or p-toluenesulfonic acid in accelerating the equilibrium towards imine formation at moderate temperatures (30-100°C). The presence of these catalysts facilitates the dehydration step essential for imine stability without introducing heavy metal contaminants that are difficult to purge later. The versatility of the solvent system is mechanistically significant; protic solvents like ethanol can participate in hydrogen bonding networks that stabilize the transition state, while aprotic solvents like acetonitrile offer different solubility profiles that favor the precipitation of the desired isomer upon cooling.
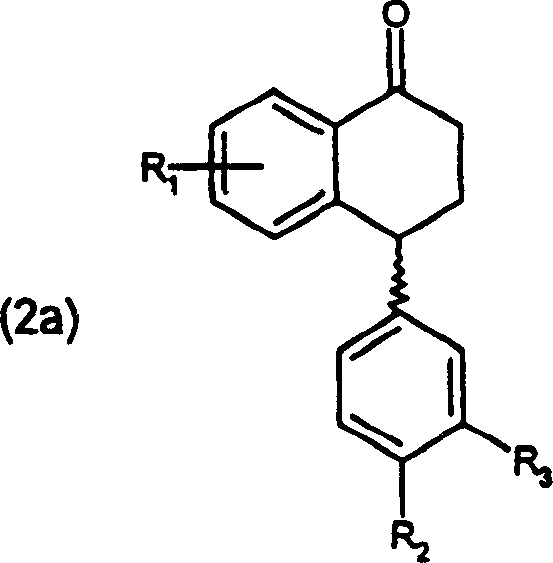
Following the reaction, the purification mechanism relies on the differential solubility of the geometric isomers. The crude product typically contains a mixture of the desired 3,4-dichloro isomer (formula 1a) and the undesired 2,3-dichloro isomer (formula 1b). By carefully selecting a recrystallization solvent and controlling the cooling gradient (0.1-1°C/min), the process exploits the lattice energy differences between these isomers. The desired 3,4-dichloro sertraline-imine crystallizes preferentially, leaving the impurities and residual ketone in the mother liquor. This recrystallization step is so effective that it can upgrade an isomeric mixture from roughly 95% purity to over 99.9% in a single pass. Furthermore, the crystal lattice formed during this process excludes catalyst residues and water, resulting in a dry, free-flowing solid that requires minimal downstream drying, thereby enhancing the overall throughput and energy efficiency of the manufacturing line.
How to Synthesize Sertraline Ketimine Efficiently
Implementing this synthesis route requires precise control over reaction parameters to maximize yield and isomeric enrichment. The process begins by charging a reactor with the sertralone isomer mixture and a selected solvent, such as ethanol or acetonitrile, followed by the introduction of methylamine gas or solution. The reaction temperature is maintained between 50°C and 70°C for several hours to ensure complete conversion, often in the presence of a catalytic amount of sulfonic acid. Once the reaction reaches completion, the mixture is cooled to induce crystallization, allowing for the immediate isolation of an enriched imine product. For ultimate purity, the crude solid is redissolved in a hot solvent and subjected to a controlled cooling cycle to precipitate the final high-purity crystals. The detailed standardized synthesis steps, including specific molar ratios and cooling profiles, are outlined in the guide below.
- React sertralone isomer mixture with methylamine in a suitable solvent (e.g., ethanol, acetonitrile) optionally with a protonic acid catalyst at 20-150°C.
- Isolate the crude sertraline-imine mixture by filtration or solvent evaporation, achieving initial enrichment of the desired 3,4-dichloro isomer.
- Purify the crude product via recrystallization in a compatible solvent (e.g., ethanol, ethyl acetate) to obtain >99.9% isomerically pure sertraline-imine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patent-covered technology translates into tangible operational improvements and risk mitigation. The elimination of titanium tetrachloride removes a major hazardous material from the supply chain, reducing the costs associated with specialized storage, handling, and neutralization of acidic waste streams. Additionally, the ability to use common, low-cost solvents like ethanol or ethyl acetate instead of specialized ethers or chlorinated solvents significantly lowers raw material expenditures. The process design inherently supports continuous manufacturing, as the product can be continuously filtered from the reaction mass, allowing for smaller reactor footprints and higher annual output capacities without proportional increases in capital investment. This scalability ensures a reliable supply of critical intermediates, mitigating the risk of production bottlenecks that often plague complex pharmaceutical syntheses.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven primarily by the simplification of the workup and purification stages. By removing the requirement for expensive molecular sieves and the associated filtration and regeneration steps, the operational expenditure per kilogram of product is drastically reduced. Furthermore, the high yield of the recrystallization step (often exceeding 80-90%) minimizes product loss, ensuring that raw material costs are maximized in the final output. The avoidance of cryogenic cooling also leads to substantial energy savings, as the reaction proceeds efficiently at near-ambient or moderately elevated temperatures, reducing the load on plant utilities.
- Enhanced Supply Chain Reliability: The robustness of the solvent system enhances supply chain resilience by allowing for the substitution of solvents based on market availability. If a specific solvent faces shortage, the process flexibility permits switching to alternatives like amines, nitriles, or esters without revalidating the entire chemistry. This adaptability prevents production stoppages due to raw material scarcity. Moreover, the simplified isolation procedure, which often involves straightforward filtration and drying, shortens the batch cycle time, enabling faster turnover and more responsive fulfillment of customer orders for high-purity pharmaceutical intermediates.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this process offers a cleaner profile that aligns with modern green chemistry principles. The absence of heavy metal catalysts simplifies wastewater treatment and reduces the burden on effluent processing facilities. The potential for solvent recycling, particularly when using low-boiling solvents like methylamine or ethanol, further diminishes the environmental footprint. This compliance advantage is critical for maintaining operating licenses in regions with stringent environmental regulations, ensuring long-term business continuity and protecting the corporate reputation of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this ketimine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process evaluation. Understanding these nuances is essential for technical teams assessing the feasibility of technology transfer or scale-up operations.
Q: How does this process improve upon traditional TiCl4 methods?
A: Traditional methods utilize titanium tetrachloride, which is ecologically harmful and requires costly cooling. This patent eliminates TiCl4, using safer solvents and catalysts like sulfonic acids, significantly reducing waste treatment costs and safety risks.
Q: What purity levels can be achieved with this recrystallization method?
A: By performing recrystallization at the imine stage rather than the ketone stage, the process achieves isomeric purity greater than 99.9% for the 3,4-dichloro isomer, with minimal catalyst residues (≤0.001%).
Q: Can this synthesis be adapted for continuous manufacturing?
A: Yes, the patent explicitly describes the suitability of the method for continuous processing. The ability to filter crystallized product continuously and recycle filtrates makes it highly scalable for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sertraline Ketimine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the global pharmaceutical supply chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the innovative processes described in CN1390198A can be seamlessly translated from the laboratory to the plant floor. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of sertraline ketimine meets the exacting standards required for downstream API synthesis. Our commitment to quality assurance means that clients receive materials with consistent isomeric purity and minimal impurity profiles, facilitating smoother regulatory filings and production runs.
We invite potential partners to engage with our technical procurement team to discuss how this advanced synthesis route can optimize your specific manufacturing needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the economic benefits tailored to your volume requirements. We encourage you to contact us today to索取 specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial reliability.
