Advanced Synthetic Routes for Substituted Dioxazine Intermediates in Fungicide Manufacturing
Advanced Synthetic Routes for Substituted Dioxazine Intermediates in Fungicide Manufacturing
The development of efficient synthetic pathways for heterocyclic intermediates is a cornerstone of modern agrochemical innovation. Patent CN1179956C introduces a groundbreaking methodology for the preparation of novel 3-(1-hydroxyphenyl-1-alkoximinomethyl)dioxazines, compounds that serve as critical precursors for materials with potent antifungal properties. This intellectual property represents a significant leap forward in process chemistry, addressing the longstanding inefficiencies associated with traditional heterocycle construction. By leveraging a versatile array of starting materials including omega-nitro-acetophenones and benzofuranone derivatives, the disclosed technology enables the production of high-purity intermediates through streamlined reaction sequences. For R&D directors and procurement strategists, understanding the mechanistic nuances and commercial implications of this patent is essential for optimizing supply chains and reducing the cost of goods sold in fungicide manufacturing.
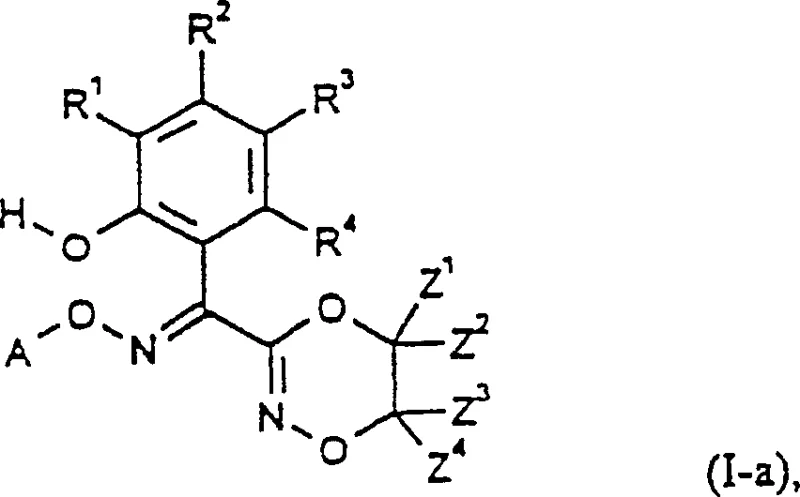
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of related dioxazine structures relied on cumbersome multi-step protocols that severely impacted economic viability and operational efficiency. As referenced in prior art such as WO 95-04728, conventional routes typically commenced with hydroxyphenylacetic acid esters, necessitating a complex sequence involving dihydropyran protection to form ether intermediates. This was followed by nitrosation, methylation, reaction with hydroxylamine, and a cyclization step using ethylene dibromide, culminating in an acid-catalyzed deprotection to remove the tetrahydropyranyl group. Each of these discrete unit operations introduced opportunities for yield loss, increased solvent waste, and extended cycle times. The cumulative effect of these inefficiencies resulted in a low overall cost rate and a substantial environmental footprint, making the legacy process ill-suited for the demands of modern large-scale commercial production where margin compression is a constant pressure.
The Novel Approach
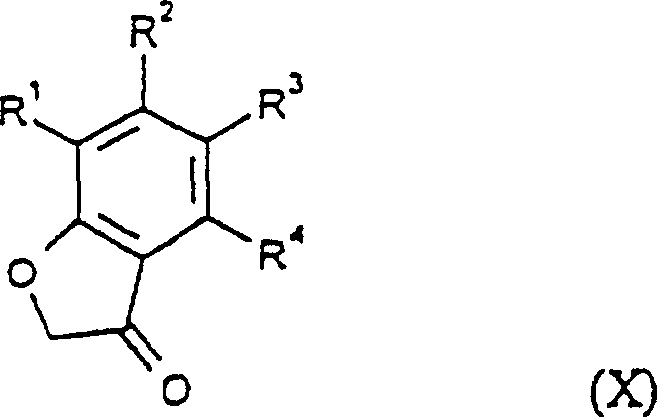
In stark contrast, the methodology disclosed in CN1179956C offers a radically simplified approach that bypasses the need for protecting groups in several key embodiments. The core innovation lies in the direct cyclization of O-hydroxyethyl-O'-alkyl-cumarone dione dioximes (Formula II) or the rearrangement of O-alkyl-cumarone dione dioximes (Formula III) under basic conditions. This strategic pivot eliminates the tedious protection-deprotection cycles inherent in older methods. Furthermore, the patent describes multiple convergent pathways (Processes a through i) to access these key intermediates, providing manufacturers with the flexibility to select the most cost-effective route based on local raw material availability. Whether starting from omega-nitro-2-hydroxy acetophenones or benzofuranones, the new process achieves the target dioxazine scaffold with fewer steps, higher atom economy, and improved safety profiles by avoiding hazardous reagents like ethylene dibromide in the primary cyclization step.
Mechanistic Insights into Base-Catalyzed Cyclization and Isomerization
The heart of this technological advancement is the base-catalyzed cyclization mechanism that transforms the linear oxime precursors into the cyclic dioxazine core. When Formula II intermediates are treated with alkali metal hydroxides in aqueous or alcoholic media, an intramolecular nucleophilic attack occurs, closing the six-membered 1,4,2-dioxazine ring. This reaction is remarkably robust, tolerating a wide range of substituents on the phenyl ring and the oxime ether moiety. The process is further enhanced by the discovery that the reaction can proceed effectively even in the presence of water, which simplifies work-up procedures and reduces the reliance on expensive anhydrous solvents. Additionally, the patent highlights a surprising stability of the intermediate Formula III compounds under alkaline conditions, allowing for a telescoped process where the intermediate is generated and consumed in situ without isolation, thereby maximizing throughput and minimizing handling losses.
Stereochemical control is another critical aspect of this synthesis, particularly given the biological significance of geometric isomers in agrochemicals. The initial cyclization often yields a mixture of E and Z stereoisomers. The patent discloses a highly effective post-synthetic isomerization protocol wherein the crude mixture is treated with inorganic or organic acids, such as hydrochloric acid or methanesulfonic acid, in inert organic solvents. This acid-catalyzed equilibration drives the equilibrium towards the thermodynamically more stable E-isomer, which is often the biologically active form. This capability ensures that the final product meets stringent purity specifications required by downstream formulators. The ability to convert unwanted Z-isomers into the desired E-form significantly boosts the effective yield of the process, turning what would otherwise be waste into valuable product and enhancing the overall sustainability of the manufacturing campaign.
How to Synthesize 3-(1-Hydroxyphenyl-1-alkoximinomethyl)dioxazines Efficiently
Implementing this synthesis requires careful attention to reaction parameters to maximize the benefits of the novel pathway. The process generally begins with the condensation of an appropriate ketone precursor, such as an omega-halo-2-hydroxy acetophenone or a benzofuranone, with an alkoxylamine to form the requisite oxime. Following this, nitrosation or alkylation steps generate the cyclization precursors. The critical cyclization is then effected using bases like sodium hydroxide or potassium carbonate in solvents ranging from water to toluene, depending on the solubility profile of the specific substrate. Temperature control is vital, with reactions typically proceeding between 0°C and 100°C. For a detailed breakdown of the specific reagents, stoichiometry, and work-up procedures validated in the patent examples, please refer to the standardized guide below.
- Prepare the key O-alkyl-benzofuranone oxime or omega-nitro-acetophenone oxime precursor via condensation with alkoxylamine.
- Execute nitrosation or alkylation with ethane derivatives to form the O-hydroxyethyl-O'-alkyl-cumarone dione dioxime intermediate.
- Perform base-catalyzed cyclization in aqueous or alcoholic media followed by acid-catalyzed isomerization to secure the thermodynamically stable E-isomer.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of the technology described in CN1179956C presents a compelling value proposition centered on cost optimization and supply security. The elimination of protecting group chemistry translates directly into reduced raw material consumption and lower waste disposal costs. By shortening the synthetic route, manufacturers can achieve faster batch cycle times, increasing asset utilization rates in multipurpose plants. Furthermore, the reliance on commodity chemicals such as hydroxylamine derivatives, simple halo-ketones, and common bases mitigates the risk of supply disruptions associated with specialized or exotic reagents. This robustness ensures a reliable agrochemical intermediate supplier can maintain consistent delivery schedules even in volatile market conditions.
- Cost Reduction in Manufacturing: The streamlined process architecture significantly lowers the cost of goods by removing entire unit operations associated with protection and deprotection. The ability to perform reactions in aqueous or alcoholic media reduces the demand for expensive chlorinated solvents, while the high yields reported in the patent examples minimize the cost per kilogram of the active intermediate. Additionally, the avoidance of heavy metal catalysts or toxic alkylating agents like ethylene dibromide in the main cyclization step reduces the regulatory burden and costs associated with environmental compliance and worker safety monitoring.
- Enhanced Supply Chain Reliability: The versatility of the synthetic routes allows for sourcing flexibility. Since the process can commence from multiple distinct starting materials (e.g., benzofuranones or nitro-acetophenones), procurement teams are not locked into a single supply line. If the price or availability of one precursor fluctuates, the manufacturing strategy can be adapted to utilize an alternative pathway described in the patent without compromising the quality of the final dioxazine intermediate. This adaptability is crucial for maintaining business continuity and negotiating favorable terms with upstream vendors.
- Scalability and Environmental Compliance: The reaction conditions described are inherently scalable, utilizing standard reactor configurations and moderate temperatures that do not require specialized cryogenic or high-pressure equipment. The use of water as a solvent or co-solvent in several steps aligns with green chemistry principles, reducing the volume of volatile organic compounds (VOCs) emitted. This environmental compatibility simplifies the permitting process for capacity expansion and supports corporate sustainability goals, making the technology attractive for long-term investment in commercial scale-up of complex agrochemical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this dioxazine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of the new cyclization method over conventional THP-protection routes?
A: The novel process eliminates the need for tetrahydropyranyl (THP) protection and deprotection steps, significantly reducing the total number of synthetic operations. This simplification leads to higher overall yields, reduced solvent consumption, and lower production costs compared to the legacy methods described in WO 95-04728.
Q: How is stereochemical purity controlled during the synthesis of dioxazine intermediates?
A: The process allows for the formation of stereoisomer mixtures which can be efficiently converted to the desired E-isomer through a dedicated acid-catalyzed isomerization step. Using mineral acids like hydrochloric acid in organic solvents ensures high stereochemical purity, which is critical for downstream biological activity.
Q: Are the starting materials for this synthesis commercially scalable?
A: Yes, the route utilizes readily available starting materials such as omega-halo-2-hydroxy acetophenones, benzofuranones, and simple alkoxylamines. These commodities are accessible in bulk quantities, ensuring a robust and continuous supply chain for large-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 3-(1-Hydroxyphenyl-1-alkoximinomethyl)dioxazines Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of high-quality intermediates in the development of next-generation fungicides. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN1179956C are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs to guarantee that every batch of dioxazine intermediate meets the exacting standards required for global agrochemical registration. Our commitment to process excellence means we can deliver materials with consistent stereochemical profiles and minimal impurity burdens.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can drive value in your supply chain. By requesting a Customized Cost-Saving Analysis, you can quantify the potential economic benefits of switching to this more efficient methodology. We are prepared to provide specific COA data and route feasibility assessments tailored to your project requirements, ensuring a seamless transition from laboratory scale to full commercial supply.
