Advanced Manufacturing of Aminophenylsulfonylureas via Novel Rearrangement Chemistry
The global demand for high-efficiency herbicides continues to drive innovation in the synthesis of sulfonylurea derivatives, a class of compounds renowned for their potent plant growth regulating activity. Patent CN1660798A introduces a groundbreaking methodology for the preparation of aminophenylsulfonylureas, addressing critical bottlenecks that have long plagued the agrochemical industry. Traditional synthetic routes often suffer from low overall yields and complex purification requirements due to the presence of multiple reactive functional groups within the molecule. This new technical approach leverages a sophisticated halogenation and rearrangement sequence to construct the core scaffold with exceptional precision. By fundamentally rethinking the order of operations, specifically regarding the introduction of the sulfonyl and urea moieties, the process achieves a level of efficiency previously unattainable. The target molecules, represented generally by formula (I), serve as vital intermediates for next-generation weed control agents.
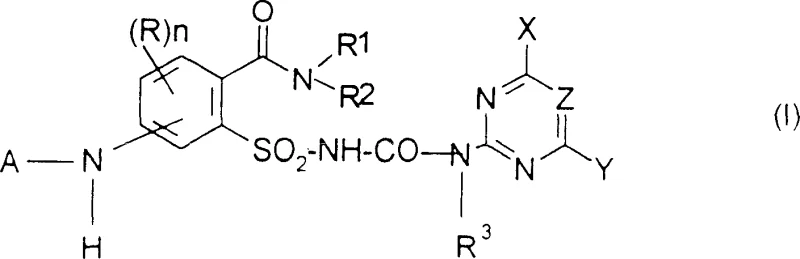
Furthermore, the strategic design of this synthesis pathway allows for significant flexibility in substituent variation, enabling the rapid generation of diverse analogues for biological testing. The ability to introduce various alkyl, alkoxy, and heterocyclic groups at specific positions on the phenyl and pyrimidine rings is crucial for optimizing herbicidal potency and environmental safety profiles. This patent not only discloses the final active ingredients but also claims the novel intermediates formed along the way, highlighting the robustness of the chemical logic employed. For manufacturers, this represents a shift from empirical trial-and-error to a rational, mechanism-driven production strategy. The elimination of unnecessary synthetic steps directly translates to reduced waste generation and lower energy consumption, aligning with modern green chemistry principles.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of amino-substituted phenylsulfonylureas has been fraught with chemical challenges that impede commercial viability. A primary obstacle is the high reactivity of the amino and sulfonamide groups, which often leads to non-selective reactions and the formation of complex impurity profiles. To circumvent this, legacy processes frequently rely on the use of protecting groups, such as the tert-butyl group, to mask the sulfonamide functionality during subsequent transformations. While effective in the laboratory, the removal of these protecting groups on an industrial scale requires harsh reagents like trifluoroacetic acid, which poses significant corrosion risks to equipment and creates difficult waste disposal issues. Moreover, the multi-step nature of these conventional routes inevitably results in moderate total recovery rates, as yield losses compound with each additional isolation and purification stage. The reliance on sensitive intermediates also complicates supply chain logistics, as these materials often have limited shelf stability.
The Novel Approach
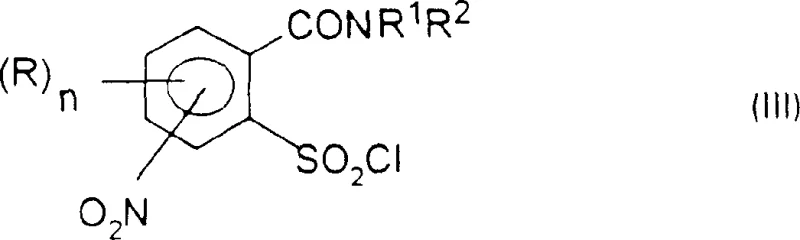
In stark contrast, the methodology described in the patent data utilizes a direct halogenation and rearrangement strategy that bypasses the need for cumbersome protecting group chemistry. The process begins with the conversion of a nitro-substituted benzoic acid derivative into an acid halide, which then undergoes a thermal rearrangement to form a stable sulfonyl chloride intermediate, as depicted in formula (III). This rearrangement is a pivotal step that installs the sulfonyl chloride functionality ortho to the carboxamide group with high regioselectivity. By deferring the reduction of the nitro group until after the sulfonamide and urea linkages are established, the process effectively manages the reactivity of the aromatic ring. This sequential logic prevents side reactions that would otherwise occur if a free amino group were present during the aggressive halogenation conditions. The result is a cleaner reaction profile and a more robust process capable of delivering high-purity intermediates suitable for direct downstream processing.
Mechanistic Insights into Halogenation-Rearrangement and Selective Urea Formation
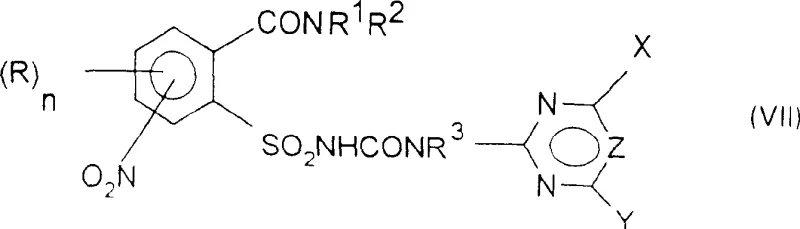
The core of this technological advancement lies in the precise control of reaction conditions during the formation of the sulfonyl chloride and the subsequent urea linkage. The initial halogenation, typically using thionyl chloride or thionyl bromide in an aprotic solvent like chlorobenzene, converts the carboxylic acid to an acyl chloride. Upon heating to temperatures exceeding 100°C, an intramolecular rearrangement occurs, migrating the sulfonyl group to the adjacent position. This transformation is highly dependent on the solvent's boiling point and the absence of protic species that could quench the reactive intermediates. Following this, the sulfonyl chloride is subjected to ammonolysis to generate the sulfonamide. A critical mechanistic advantage is observed in the urea formation step, where the patent specifies the use of carbamate salts rather than neutral carbamates. When a neutral carbamate is used in the presence of a base like DBU, there is a risk of bis-acylation, where both the sulfonamide nitrogen and the aniline nitrogen react. However, by pre-forming the carbamate salt (where M is a metal cation like sodium or potassium), the nucleophilicity is tuned to favor reaction exclusively at the sulfonamide nitrogen or the aniline nitrogen depending on the specific sequence, thereby ensuring the correct connectivity of the final sulfonylurea bridge.
Impurity control is further enhanced by the timing of the nitro group reduction. In one preferred embodiment, the nitro group is reduced to an amine only after the sulfonylurea backbone is fully assembled, or alternatively, the amine is generated prior to urea formation but protected by the specific reaction kinetics of the carbamate salt. Catalytic hydrogenation using standard catalysts like Raney nickel or palladium on carbon is employed for this reduction, operating under moderate hydrogen pressures of 1 to 50 bar. This step is remarkably clean, avoiding the use of stoichiometric metal reductants like iron or zinc which generate substantial solid waste. The selectivity of the hydrogenation is such that other sensitive functional groups, such as the amide or ester moieties potentially present on the ring, remain intact. This chemoselectivity is paramount for maintaining the structural integrity of complex heterocyclic substituents often required for biological activity.
How to Synthesize Aminophenylsulfonylureas Efficiently
The synthesis of these complex agrochemical intermediates requires a disciplined approach to reaction engineering and parameter control. The patented route offers a clear, stepwise protocol that minimizes operational complexity while maximizing yield. Operators must pay close attention to temperature gradients during the rearrangement phase and the stoichiometry of the carbamate salt addition to prevent over-alkylation. The detailed standardized synthesis steps provided below outline the critical parameters for each stage, from the initial halogenation to the final acylation if required. Adhering to these guidelines ensures reproducibility and safety, particularly when handling reactive acid chlorides and hydrogen gas.
- Perform halogenation of nitro-substituted benzoic acid derivatives followed by thermal rearrangement to generate the key sulfonyl chloride intermediate.
- Execute ammonolysis to form the sulfonamide, followed by catalytic hydrogenation to reduce the nitro group to an amine.
- React the resulting amino sulfonamide with a carbamate salt under controlled conditions to form the final sulfonylurea with high chemo-selectivity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits beyond mere technical elegance. The primary advantage lies in the drastic simplification of the manufacturing workflow, which directly correlates to reduced operational expenditures. By eliminating the need for protecting group installation and removal, the process removes two entire unit operations from the production schedule. This reduction in step count not only lowers the consumption of raw materials and solvents but also decreases the labor and utility costs associated with running additional batches. Furthermore, the avoidance of corrosive reagents like trifluoroacetic acid extends the lifespan of reactor vessels and piping, reducing capital expenditure on maintenance and replacement. These efficiencies compound to create a significantly more cost-competitive supply chain for high-volume herbicide intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the atom economy and the reduction of auxiliary chemicals. Traditional routes often consume large molar excesses of protecting group reagents and expensive deprotection acids, none of which end up in the final product. In this new method, the reagents are primarily incorporated into the product or converted into benign byproducts like sulfur dioxide and hydrochloric acid, which are easily scrubbed. Additionally, the use of carbamate salts improves the yield of the critical urea formation step by minimizing byproduct formation, meaning less starting material is wasted. This higher effective yield per batch allows manufacturers to meet demand with fewer production runs, optimizing asset utilization and lowering the cost per kilogram of the active ingredient.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the reliance on specialized or unstable intermediates. This patented method utilizes robust, commercially available starting materials such as nitrobenzoic acids and common heterocyclic amines. The intermediates generated, such as the sulfonyl chloride and the nitro-sulfonamide, are sufficiently stable to be isolated and stored if necessary, providing flexibility in production scheduling. This stability reduces the risk of batch failures due to reagent degradation, a common issue with sensitive protected species. Consequently, suppliers can offer more reliable lead times and maintain consistent inventory levels, shielding downstream formulators from market volatility and raw material shortages.
- Scalability and Environmental Compliance: Scaling chemical processes often reveals hidden inefficiencies, but this route is designed with industrial practicality in mind. The solvents employed, including toluene, chlorobenzene, and THF, are standard in the fine chemical industry with well-established recovery and recycling protocols. The elimination of heavy metal reductants and strong fluorinated acids simplifies wastewater treatment and hazardous waste disposal, facilitating compliance with increasingly stringent environmental regulations. The process operates at pressures and temperatures that are compatible with standard glass-lined or stainless steel reactors, removing the need for specialized high-pressure or cryogenic equipment. This ease of scale-up ensures that production can be ramped from pilot plant to multi-ton commercial capacity without significant process redesign.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating the feasibility of technology transfer and for procurement specialists assessing supplier capabilities.
Q: How does this novel process eliminate the need for protecting groups?
A: Traditional methods often require bulky protecting groups like t-butyl for the sulfonamide moiety, necessitating harsh removal reagents like trifluoroacetic acid. This patented route utilizes a direct halogenation and rearrangement strategy on nitro-substituted precursors, allowing the sulfonamide to be formed directly via ammonolysis without prior protection, thereby simplifying the workflow.
Q: What ensures the chemo-selectivity during the urea formation step?
A: The process achieves superior selectivity by employing carbamate salts (where M is a cation) rather than neutral carbamates. This modification prevents the unwanted double substitution on both the sulfonamide and the aniline nitrogen, ensuring the urea linkage forms exclusively at the desired position with minimal byproduct generation.
Q: Can this synthesis route be scaled for industrial production?
A: Yes, the method is designed for scalability. It utilizes common industrial solvents such as toluene, chlorobenzene, and THF, and avoids exotic catalysts. The reactions, including the thermal rearrangement and catalytic hydrogenation, are conducted under standard pressure and temperature ranges suitable for large-scale reactor operations.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Aminophenylsulfonylureas Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from patent literature to commercial reality requires more than just chemical knowledge; it demands engineering excellence. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel rearrangement chemistry are fully realized in practice. We maintain stringent purity specifications through our rigorous QC labs, utilizing advanced analytical techniques to monitor impurity profiles at every stage of the synthesis. Our commitment to quality ensures that the aminophenylsulfonylureas we deliver meet the exacting standards required for global agrochemical registration and formulation.
We invite you to collaborate with us to optimize your supply chain for these critical herbicide intermediates. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us to request specific COA data and route feasibility assessments, allowing you to make informed decisions based on hard data rather than assumptions. Together, we can drive down costs and secure a sustainable supply of high-performance agrochemical ingredients.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
