Advanced Metal-Free Synthesis of 6-Benzylphenanthridine Compounds for Commercial Scale-Up
Advanced Metal-Free Synthesis of 6-Benzylphenanthridine Compounds for Commercial Scale-Up
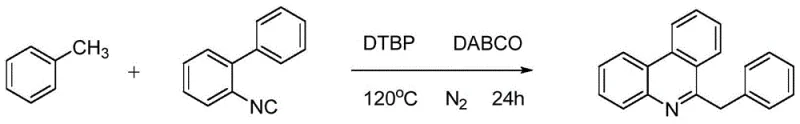
The development of efficient synthetic routes for bioactive heterocyclic scaffolds remains a critical priority for the global pharmaceutical industry. As detailed in the groundbreaking patent CN107235900B, a novel methodology has been established for the synthesis of 6-benzylphenanthridine compounds, a structural motif prevalent in natural products exhibiting potent pharmacological activities such as anti-liver cancer and anti-gastric cancer properties. This technology represents a paradigm shift from traditional transition-metal catalyzed processes to a greener, metal-free radical C-H activation strategy. For R&D directors and procurement specialists seeking reliable pharmaceutical intermediate suppliers, this innovation offers a robust pathway to access complex phenanthridine derivatives with improved safety profiles and reduced operational costs. The method leverages readily available toluene derivatives and 2-isocyanobiphenyl precursors, utilizing di-tert-butyl peroxide (DTBP) as an oxidant to drive the cyclization under mild thermal conditions.

The significance of this technological advancement cannot be overstated when considering the historical challenges associated with phenanthridine synthesis. Conventional approaches often relied on hazardous reagents or expensive catalytic systems that complicated downstream processing. By eliminating the need for heavy metal catalysts, this new route not only aligns with stringent environmental regulations but also simplifies the purification workflow, directly addressing key pain points in cost reduction in API manufacturing. The versatility of the reaction allows for the introduction of diverse functional groups, enabling the rapid generation of analog libraries for drug discovery programs while maintaining high purity standards essential for clinical applications.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of 6-benzylphenanthridine derivatives was predominantly achieved through methods fraught with significant operational and safety drawbacks. The Grignard reagent method, for instance, necessitated the use of 2-cyano-2'-bromobiphenyl and ethyl magnesium bromide in the presence of copper catalysts. This approach demanded harsh reaction conditions involving high temperatures and pressures, which inherently increased energy consumption and equipment stress. Furthermore, the reliance on transition metal catalysis introduced the persistent issue of residual metal contamination, requiring extensive and costly purification steps to meet pharmaceutical grade specifications. Alternatively, the azidotrimethylsilane method utilized dangerous azide raw materials and required substantial amounts of strong acids like trifluoroacetic acid, posing severe safety risks regarding explosion hazards and generating significant acidic waste streams that burdened environmental compliance protocols.
The Novel Approach
In stark contrast, the methodology disclosed in patent CN107235900B introduces a streamlined, metal-free radical reaction that operates under significantly milder conditions. By employing toluene derivatives and 2-isocyanobiphenyl as starting materials in the presence of an oxidant and a catalytic amount of base, the reaction proceeds efficiently at temperatures ranging from 0°C to 120°C. This approach completely circumvents the need for toxic heavy metal catalysts and hazardous azide reagents, thereby drastically improving the safety profile of the manufacturing process. The use of common organic solvents and inexpensive oxidants like DTBP ensures that the process is not only economically viable but also environmentally benign. This novel route facilitates easier post-treatment procedures, as the absence of metal residues eliminates the need for complex chelation or filtration steps, resulting in higher overall throughput and reduced production lead times for high-purity pharmaceutical intermediates.
Mechanistic Insights into Metal-Free Radical C-H Activation
The core of this synthetic breakthrough lies in its elegant mechanistic pathway, which activates the benzylic C-H bond of the toluene derivative through a radical mechanism initiated by the oxidant. Under thermal conditions, the peroxide oxidant decomposes to generate free radicals that abstract a hydrogen atom from the methyl group of the toluene substrate, forming a stabilized benzylic radical species. This reactive intermediate then undergoes addition to the isocyano group of the 2-isocyanobiphenyl, triggering a cascade of intramolecular cyclization events that construct the phenanthridine core. The presence of a base, such as DABCO, plays a crucial role in facilitating proton transfer steps and stabilizing intermediate species, ensuring the reaction proceeds to completion with high selectivity. This metal-free activation strategy avoids the coordination complexities often seen with transition metals, leading to a cleaner reaction profile with fewer side products.
From an impurity control perspective, this mechanism offers distinct advantages for process chemistry teams. Traditional metal-catalyzed reactions often suffer from homocoupling side reactions or incomplete conversion due to catalyst deactivation. However, the radical nature of this transformation, driven by the steady decomposition of the peroxide oxidant, ensures a consistent supply of active species throughout the reaction duration of 12 to 24 hours. The high functional group tolerance observed across various substrates, including those with electron-donating methoxy groups and electron-withdrawing chloro or fluoro substituents, indicates that the radical intermediate is sufficiently robust to withstand diverse electronic environments without decomposing. This stability translates to a narrower impurity profile in the crude reaction mixture, simplifying the final crystallization or chromatography steps required to achieve the stringent purity specifications demanded by regulatory bodies for commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize 6-Benzylphenanthridine Efficiently
The practical implementation of this synthesis is designed for ease of operation, making it highly attractive for both laboratory-scale optimization and industrial production. The protocol involves a straightforward sequence where the reaction solvent and catalytic base are charged into a vessel, followed by degassing to establish an inert nitrogen atmosphere. Subsequently, the oxidant, toluene derivative, and isocyanide are added, and the mixture is heated in an oil bath. This simplicity reduces the technical barrier for adoption and minimizes the risk of operator error during batch preparation. For detailed standard operating procedures and specific stoichiometric ratios optimized for different substrates, please refer to the technical guide below.
- Prepare the reaction vessel by adding solvent and catalytic base, followed by three cycles of vacuum-nitrogen purging to ensure an inert atmosphere.
- Add the oxidant (e.g., DTBP), toluene derivative substrate, and 2-isocyanobiphenyl to the vessel and heat to 120°C for 12-24 hours.
- Upon completion, remove the solvent and purify the crude mixture via column chromatography to isolate the high-purity 6-benzylphenanthridine product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this metal-free synthesis route presents compelling economic and logistical benefits. The elimination of expensive transition metal catalysts such as copper or palladium removes a significant cost driver from the raw material bill of materials. Moreover, the avoidance of hazardous reagents like azides reduces the costs associated with specialized storage, handling, and waste disposal, contributing to substantial overall cost savings. The mild reaction conditions also imply lower energy consumption compared to high-pressure alternatives, further enhancing the economic efficiency of the process. These factors collectively position this technology as a superior choice for reducing lead time for high-purity pharmaceutical intermediates while maintaining a competitive price point.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of transition metal catalysts. In traditional processes, the cost of the catalyst itself, combined with the expensive ligands often required and the rigorous purification steps needed to reduce metal residues to ppm levels, constitutes a major portion of the COGS. By switching to an organic radical system using cheap oxidants like DTBP and bases like DABCO, manufacturers can drastically simplify the downstream processing train. There is no need for scavenger resins or complex aqueous workups designed to strip metals, which reduces solvent usage and labor hours. This streamlined workflow directly translates to lower manufacturing costs and improved margin potential for the final API.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of commodity chemicals as starting materials. Toluene derivatives and 2-isocyanobiphenyl are widely available from multiple global suppliers, reducing the risk of single-source bottlenecks. Unlike specialized organometallic reagents that may have long lead times or strict transportation regulations, the reagents used in this process are stable and easy to source. This availability ensures consistent production schedules and minimizes the risk of delays caused by raw material shortages. Furthermore, the robustness of the reaction against moisture and air (after initial purging) allows for more flexible manufacturing windows, enhancing the overall reliability of the supply chain for critical oncology intermediates.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety and environmental challenges, but this methodology mitigates those risks effectively. The absence of high-pressure requirements and explosive azides makes the process inherently safer for large-scale reactors, lowering insurance premiums and safety infrastructure costs. From an environmental standpoint, the generation of non-toxic byproducts and the lack of heavy metal waste simplify effluent treatment, ensuring compliance with increasingly strict global environmental regulations. This 'green' profile not only avoids potential regulatory fines but also aligns with the sustainability goals of major pharmaceutical partners, making the supply of these intermediates more attractive to eco-conscious multinational corporations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield optimization, substrate compatibility, and process safety. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: What are the primary advantages of this metal-free synthesis over traditional Grignard methods?
A: Unlike traditional Grignard methods which require harsh high-temperature/high-pressure conditions and expensive copper catalysts, this novel radical pathway operates under mild conditions (120°C) without transition metals, significantly reducing environmental pollution and post-reaction purification costs.
Q: What is the substrate scope for the toluene component in this reaction?
A: The method demonstrates excellent functional group tolerance, successfully accommodating various substituents on the toluene ring including methyl, methoxy, chloro, fluoro, and even naphthalene groups, yielding products with efficiencies ranging from 68% to 87%.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process utilizes cheap and readily available oxidants like DTBP and common organic bases like DABCO. The absence of sensitive reagents like azides and the mild reaction profile make it highly scalable and safer for industrial manufacturing compared to prior art.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Benzylphenanthridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of this metal-free synthesis technology for the production of next-generation anticancer agents. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from benchtop discovery to market supply is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 6-benzylphenanthridine intermediate delivered meets the highest quality standards required for clinical and commercial use. We are committed to leveraging our technical expertise to optimize this novel route for maximum efficiency and yield.
We invite you to collaborate with us to unlock the full commercial value of this innovative synthesis. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this metal-free approach can improve your bottom line. Please contact our technical procurement team today to request specific COA data for our pilot batches and comprehensive route feasibility assessments. Let us help you secure a sustainable and cost-effective supply chain for your critical phenanthridine-based drug candidates.
